- Home
- »
- Clinical Diagnostics
- »
-
Point of Care Infectious Disease Testing Market Size Report, 2025GVR Report cover
![Point of Care Infectious Disease Testing Market Size, Share & Trends Report]()
Point of Care Infectious Disease Testing Market Size, Share & Trends Analysis Report By Disease (HIV POC, HBV POC, Influenza/Flu POC, TB & drug-resistant TB POC), By End-use, And Segment Forecasts, 2018 - 2025
- Report ID: GVR-1-68038-569-4
- Number of Pages: 145
- Format: Electronic (PDF)
- Historical Range: 2014 - 2016
- Industry: Healthcare
Industry Insights
The global point of care infectious disease testing market size was estimated at USD 1.05 billion in 2016. Driven by the latest trends of consumer-centric services coupled with high demand for portable and rapid testing devices this market is expected to witness lucrative growth. Moreover, continuous rise in number cases of infectious diseases has boosted the advancements in point of care instruments sector thereby driving progress.
Rising incidences & prevalence rates of communicable diseases coupled with increasing geriatric population that have lower immunity are attributive to upsurge the demand for better diagnostics in the market. Demand for these tests is specific in nature and pronounces the need for intensive R&D in order to treat several infections.
Japan POC infectious diseases market, 2014 - 2025 by end-use (USD Million)
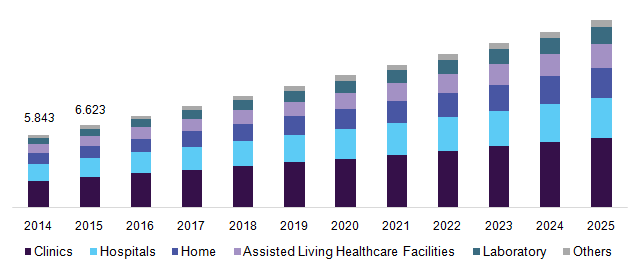
The emerging space of point of care instruments is significantly in advancing and achieving the objectives of the healthcare realm. Launch of mobile & self-testing devices has increased access to testing and monitoring systems to the remote places where the provision of timely medical care is a challenge.
The emerging market players other than established firms are engaged in the design of a significant number of novel and innovative algorithms and solutions thus fueling growth in the diagnostic sector. The presence of several products in the pipeline stage is expected to add to revenue in the coming years.
The decreasing capacity of central labs to meet the growing demand for healthcare is one of the major factors responsible for escalating the demand for decentralized testing. Such factors resultantly increase the demand for self-testing devices thereby leading to revenue generation.
Furthermore, rising adoption of companion diagnostics, the virulence of infectious diseases, and the need for better and regular monitoring systems in near-patient settings are expected to drive the point of care infectious disease diagnostics/testing market with potential avenues. As the trend and advancements in personalized medicine continue to rise, bringing molecular testing to the point of care in the patient’s home, doctor’s clinics and hospitals will continue to be in demand.
Disease Insights
Human Immunodeficiency Virus (HIV) POC tests accounted for the largest revenue share in 2016. Efforts taken up by various entities such as World Health Organization (WHO), Centers for Disease Control and Prevention (CDC), and major companies for contributing towards advancing and delivering sensitive and accurate devices are anticipated to enhance penetration of these devices in the coming years.
Demand for POC testing of influenza, TB and MDR-TB is expected to grow at a lucrative pace in the coming years as a consequence of rising incidence rates of these diseases. Moreover, the development of rapid RT-PCR tests for influenza that are better in the detection of the microorganisms than the conventional tests are anticipated to boost progress in this segment.
End-use Insights
Clinics accounted for the largest market share owing to the higher adoption of these devices and platforms in clinics. Moreover, increasing penetration of these devices in the clinics and associated facilities is attributive to the estimated share.
Assisted living healthcare facilities are anticipated to register the fastest growth in the coming years due to higher usage in this segment as a consequence of rising demand by the geriatric population base.
Point-of-care infectious disease diagnostics/testing market, by region, 2016 (%)
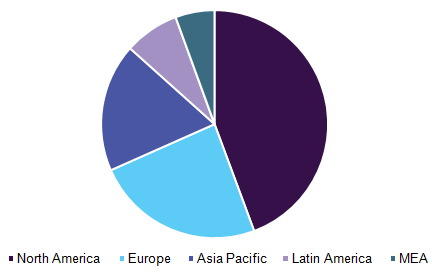
Regional Insights
North America dominated the point of care infectious disease testing market, particularly the U.S. holds the highest revenue share in the field of rapid and easy to use diagnostics, owing to factors such as the presence of substantial players, well-established healthcare framework. Moreover, the introduction of technological advances in this region supports the estimated revenue share.
However, Asia Pacific is expected to emerge as the fastest-growing region due to increment in R&D expenditure in patient-centric facilities in Japan and China. Furthermore, the dramatically increasing burden of chronic infectious diseases coupled with the presence of government initiatives aimed to promote early diagnosis are factors expected to fuel regional progress.
Companies in Asia are focusing on the development of novel assays, for instance, in May 2017, One BioMed and A*Star’s Genome Institute of Singapore established a joint lab venture for molecular diagnostic assay production. This collaboration is intended for the development of assays for the Asian clinical infectious disease testing market. These assays include tests for pediatric respiratory infections, mosquito-borne illnesses, and drug-resistant tuberculosis.
Point of Care Infectious Disease Testing Market Share Insights
The major players in the sector include Alere, Thermo Fisher Scientific Inc, F. Hoffmann-La Roche Ltd, Siemens Healthineers, BD & Company, Chembio Diagnostics Inc., Trinity Biotech, Cardinal Health, Quest Diagnostics Inc., Bio-Rad Labs Inc., bioMérieux SA, Sight Diagnostics Ltd., Gene POC, Trivitron Healthcare, OJ-Bio Ltd., and Ortho-Clinical Diagnostics.
Currently, major players in the sector are forming strategic collaborations with other established and developing entities. The objectives behind the collaborations are enhancement of market presence by building a strong product portfolio that aids in accurate diagnosis.
Report Scope
Attribute
Details
Base year for estimation
2016
Actual estimates/Historical data
2014 - 2016
Forecast period
2017 - 2025
Market representation
Revenue in USD Million & CAGR from 2017 to 2025
Regional scope
North America, Europe, Asia Pacific, Latin America, Middle East & Africa
Country scope
U.S., Canada, U.K., Germany, Japan, China, Brazil, South Africa.
Report coverage
Revenue forecast, company share, competitive landscape, growth factors and trends
15% free customization scope (equivalent to 5 analyst working days)
If you need specific market information, which is not currently within the scope of the report, we will provide it to you as a part of customization
Segments Covered in the ReportThis report forecasts revenue growth at the regional & country level and provides an analysis of the latest trends and opportunities in each of the sub-segments from 2014 to 2025. For the purpose of this report, Grand View Research has segmented the point of care infectious disease testing market on the basis of diseases, end-use, and regions:
-
Disease Outlook (Revenue, USD Million; 2014-2025)
-
HIV POC
-
Clostridium difficile POC
-
HBV POC
-
Pneumonia or Streptococcus associated infections
-
Respiratory syncytial virus (RSV) POC
-
HPV POC
-
Influenza/Flu POC
-
HCV POC
-
MRSA POC
-
TB and drug-resistant TB POC
-
HSV (Herpes Simplex Virus) POC
-
Other infectious disease POC
-
-
End-Use Outlook (Revenue, USD Million; 2014-2025)
-
Hospitals
-
Clinics
-
Home
-
Assisted Living Healthcare Facilities
-
Laboratories
-
Others
-
-
Regional Outlook (Revenue, USD Million; 2014-2025)
-
North America
-
The U.S.
-
Canada
-
-
Europe
-
Germany
-
The U.K.
-
-
Asia Pacific
-
Japan
-
China
-
-
Latin America
-
Brazil
-
-
The Middle East and Africa (MEA)
-
South Africa
-
-
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





