U.S. Laboratory Developed Tests Market Size & Outlook
Related Markets
U.S. laboratory developed tests market highlights
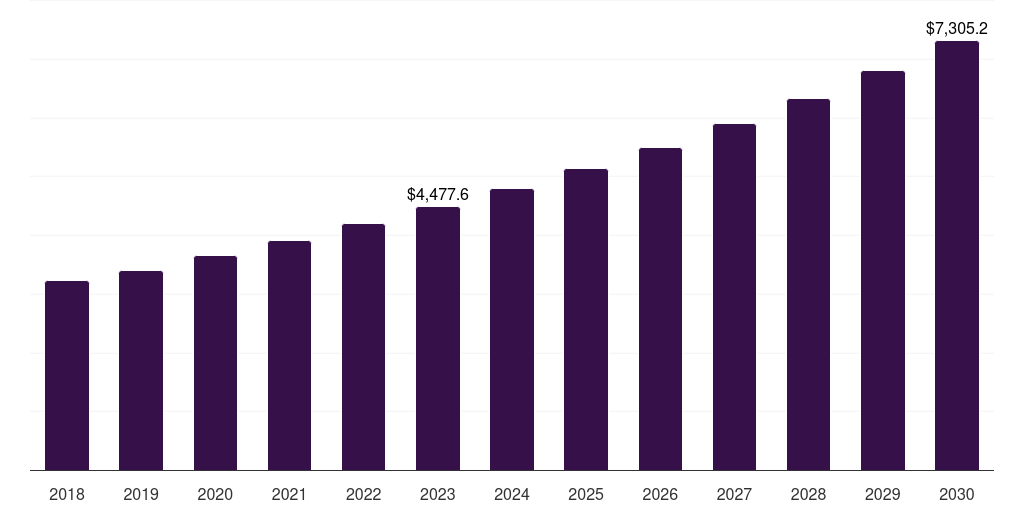
- The U.S. laboratory developed tests market generated a revenue of USD 4,477.6 million in 2023 and is expected to reach USD 7,305.2 million by 2030.
- The U.S. market is expected to grow at a CAGR of 7.2% from 2024 to 2030.
- In terms of segment, molecular diagnostics was the largest revenue generating technology in 2023.
- Immunoassays is the most lucrative technology segment registering the fastest growth during the forecast period.
Laboratory developed tests market data book summary
| Market revenue in 2023 | USD 4,477.6 million |
| Market revenue in 2030 | USD 7,305.2 million |
| Growth rate | 7.2% (CAGR from 2023 to 2030) |
| Largest segment | Molecular diagnostics |
| Fastest growing segment | Immunoassays |
| Historical data | 2018 - 2022 |
| Base year | 2023 |
| Forecast period | 2024 - 2030 |
| Quantitative units | Revenue in USD million |
| Market segmentation | Immunoassays, Hematology and Coagulation, Molecular Diagnostics, Microbiology, Clinical Chemistry, Histology/Cytology, Flow Cytometry, Mass Spectroscopy |
| Key market players worldwide | Quest Diagnostics Inc, 23andMe Holding Co Class A, Abbott Laboratories, Guardant Health Inc, NeoGenomics, Siemens Healthineers AG ADR, Qiagen NV, Illumina Inc, Roche Holding AG ADR |
Other key industry trends
- In terms of revenue, U.S. accounted for 36.2% of the global laboratory developed tests market in 2023.
- Country-wise, U.S. is expected to lead the global market in terms of revenue in 2030.
- In North America, U.S. laboratory developed tests market is projected to lead the regional market in terms of revenue in 2030.
- U.S. is the fastest growing regional market in North America and is projected to reach USD 7,305.2 million by 2030.
No credit card required*
Horizon in a snapshot
- 30K+ Global Market Reports
- 120K+ Country Reports
- 1.2M+ Market Statistics
- 200K+ Company Profiles
- Industry insights and more
Laboratory Developed Tests Market Scope
Laboratory Developed Tests Market Companies
| Name | Profile | # Employees | HQ | Website |
|---|---|---|---|---|
| NeoGenomics | View profile | 501-1000 | Fort Myers, Florida, United States, North America | http://www.neogenomics.com |
| 23andMe Holding Co Class A | View profile | 816 | 349 Oyster Point Boulevard, South San Francisco, CA, United States, 94080 | https://www.23andme.com |
| Guardant Health Inc | View profile | 1779 | 3100 Hanover Street, Palo Alto, CA, United States, 94304 | https://www.guardanthealth.com |
| Qiagen NV | View profile | 5900 | Hulsterweg 82, Venlo, LI, Netherlands, 5912 PL | https://www.qiagen.com |
| Illumina Inc | View profile | 9300 | 5200 Illumina Way, San Diego, CA, United States, 92122 | https://www.illumina.com |
| Quest Diagnostics Inc | View profile | 48000 | 500 Plaza Drive, Secaucus, NJ, United States, 07094 | https://www.questdiagnostics.com |
| Siemens Healthineers AG ADR | View profile | 71400 | Siemensstr. 3, Forchheim, BY, Germany, 91301 | https://www.siemens-healthineers.com |
| Abbott Laboratories | View profile | 114000 | 100 Abbott Park Road, Abbott Park, IL, United States, 60064-6400 | https://www.abbottinvestor.com |
| Roche Holding AG ADR | View profile | 103605 | Grenzacherstrasse 124, Basel, Switzerland, 4070 | https://www.roche.com |
U.S. laboratory developed tests market outlook
The databook is designed to serve as a comprehensive guide to navigating this sector. The databook focuses on market statistics denoted in the form of revenue and y-o-y growth and CAGR across the globe and regions. A detailed competitive and opportunity analyses related to laboratory developed tests market will help companies and investors design strategic landscapes.
Molecular diagnostics was the largest segment with a revenue share of 27.32% in 2024. Horizon Databook has segmented the U.S. laboratory developed tests market based on immunoassays, hematology and coagulation, molecular diagnostics, microbiology, clinical chemistry, histology/cytology, flow cytometry, mass spectroscopy covering the revenue growth of each sub-segment from 2018 to 2030.
Several key market players operating in the LDT market are introducing new products and entering into several strategic alliances, which is contributing to the country’s market growth. For instance, in May 2022, Guardant Health launched its first blood-based test, Sheild, in the U.S., for Colorectal Cancer (CRC) detection at an early stage.
The test is intended for individuals aged 45 and older who are not updated with screening guidelines, exhibit no symptoms, and are at average risk for CRC. Similarly, in March 2020, LabCorp announced the availability of a coronavirus test that was expected to accelerate the screening process in the U.S.
Furthermore, in August 2018, Enzo Biochem announced the acquisition of a manufacturing & distribution unit to advance its therapeutic and diagnostic growth strategy. This expansion would facilitate the company’s efforts to obtain CE marking and FDA approval for its LDTs.
Reasons to subscribe to U.S. laboratory developed tests market databook:
-
Access to comprehensive data: Horizon Databook provides over 1 million market statistics and 20,000+ reports, offering extensive coverage across various industries and regions.
-
Informed decision making: Subscribers gain insights into market trends, customer preferences, and competitor strategies, empowering informed business decisions.
-
Cost-Effective solution: It's recognized as the world's most cost-effective market research database, offering high ROI through its vast repository of data and reports.
-
Customizable reports: Tailored reports and analytics allow companies to drill down into specific markets, demographics, or product segments, adapting to unique business needs.
-
Strategic advantage: By staying updated with the latest market intelligence, companies can stay ahead of competitors, anticipate industry shifts, and capitalize on emerging opportunities.
Target buyers of U.S. laboratory developed tests market databook
-
Our clientele includes a mix of laboratory developed tests market companies, investment firms, advisory firms & academic institutions.
-
30% of our revenue is generated working with investment firms and helping them identify viable opportunity areas.
-
Approximately 65% of our revenue is generated working with competitive intelligence & market intelligence teams of market participants (manufacturers, service providers, etc.).
-
The rest of the revenue is generated working with academic and research not-for-profit institutes. We do our bit of pro-bono by working with these institutions at subsidized rates.
Horizon Databook provides a detailed overview of country-level data and insights on the U.S. laboratory developed tests market , including forecasts for subscribers. This country databook contains high-level insights into U.S. laboratory developed tests market from 2018 to 2030, including revenue numbers, major trends, and company profiles.
Partial client list
U.S. laboratory developed tests market size, by technology, 2018-2030 (US$M)
U.S. Laboratory Developed Tests Market Outlook Share, 2024 & 2030 (US$M)
Related industry reports
Related statistics
Sign up - it's easy, and free!
Sign up and get instant basic access to databook, upgrade
when ready, or enjoy our
free plan indefinitely.
Included in Horizon account
- 30K+ Global Market Reports
- 120K+ Country Reports
- 1.2M+ Market Statistics
- 200K+ Company Profiles
- Industry insights and more