- Home
- »
- Medical Devices
- »
-
Active Pharmaceutical Ingredient CDMO Market Report, 2030GVR Report cover
![Active Pharmaceutical Ingredient CDMO Market Size, Share & Trends Report]()
Active Pharmaceutical Ingredient CDMO Market Size, Share & Trends Analysis Report By Product, By Synthesis, By Drug, By Application, By Workflow, By Region, And Segment Forecasts, 2024 - 2030
- Report ID: GVR-4-68038-282-2
- Number of Pages: 137
- Format: Electronic (PDF)
- Historical Range: 2018 - 2022
- Industry: Healthcare
Market Size & Trends
The global active pharmaceutical ingredient CDMO market size was valued at USD 100.4 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 7.4% from 2024 to 2030. This growth can be attributed to factors such as increased investments in drug R&D, rising incidences of chronic diseases, expanding importance of generics, and an increasing consumption of biopharmaceuticals. Moreover, the rising adoption of development and manufacturing outsourcing services as an effective cost-containment mechanism further fuels market growth.
COVID-19 significantly impacted the supply chain of active pharmaceutical ingredients (API), causing some level of disruption in raw material supply and availability, shipping delays, and costs. However, the demand for new treatment options and an increase in approval rates of new drugs supported market growth during the pandemic. The growing burden of chronic diseases such as cancer, heart disease, and others is boosting the demand for new treatment options. This is expected to drive demand for new API in the coming years and hence support market expansion in the post-pandemic period.
Furthermore, rising demand for generic drugs further propels overall market growth, Generic drugs enter the market after patent expiration of branded drugs and are available at a very economical price, which has led to an increase in their adoption. Countries such as China and India are currently hotspots for development and manufacturing of generic drugs. According to Healthline, China nearly controls the complete supply of generics in the U.S. and has control over critical supplies of drugs & drug companies. Healthline also reported that the U.S. market for generic drugs has moved offshore, especially to China, and nearly no company in the country manufactures generic drugs.
Forinstance, as of 2022, approximately 91% of prescriptions in the U.S were estimated to be fulfilled with generic drugs, and the FDA has approved over 32,000 generic drugs. Similarly, in 2020, generic drug program by the U.S. FDA approved or tentatively approved 948 generic drug applications (ANDAs), out of which 50 were original applications. In total, 72 first generic drugs were approved by the FDA in 2020. A generic drug costs 30-90% less than branded drugs because the cost and time required from development to commercialization of novel drugs are quite high.
Moreover, pharmaceutical companies have been steadily increasing the amount of discovery, development, and manufacturing work they outsource over the last couple of decades. However, the urge to outsource is not uniform in the biopharma industry. Small biotechnology companies frequently rely on a contract development and manufacturing organization (CDMO) to produce their development products as they go through the pipeline.
Market Concentration & Characteristics
The market exhibits a moderate degree of innovation, driven by advancements in synthetic chemistry, process optimization, and the adoption of cutting-edge technologies such as continuous manufacturing and biocatalysis.
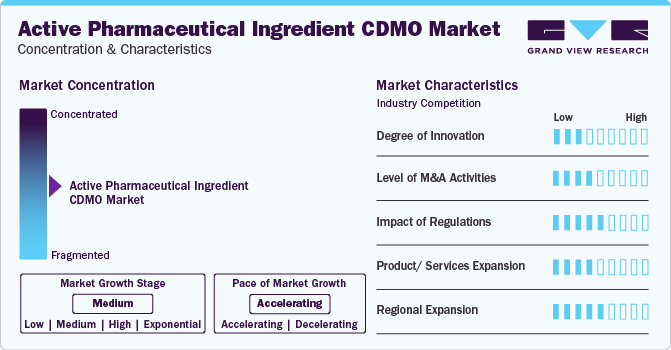
Several market players such as Catalent Inc., Recipharm AB, and CordenPharma International are heavily involved in merger and acquisition activities. Through this strategy, these companies can expand their geographic reach and enter new territories.
Adapting to evolving regulations for data integrity, sustainability, and serialization is crucial, shaping the competitive landscape and operational practices within the API CDMO industry. Potential substitutes in the market include in-house API manufacturing by pharmaceutical companies and alternative sourcing strategies for APIs from multiple suppliers to ensure supply chain resilience and flexibility.
The market is strategically pursuing regional expansion by establishing manufacturing facilities and partnerships to meet growing demand, capitalize on specific market opportunities, and ensure proximity to key pharmaceutical markets, fostering a more responsive and efficient supply chain.
Product Insights
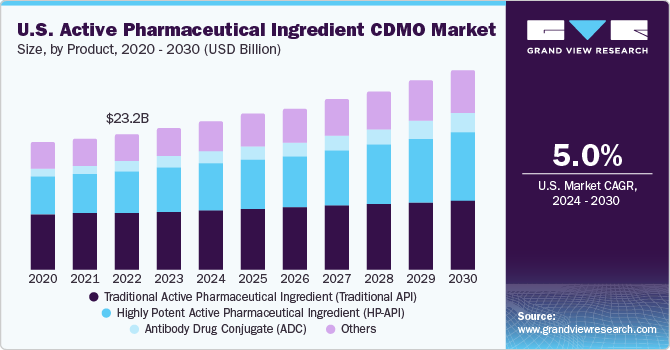
The traditional active pharmaceutical ingredient segment held the largest share of 39.0% in 2023.The market is further anticipated to experience significant growth throughout the forecast period. This growth is attributed to factors such as increased pharmaceutical Research and Development (R&D) activities, a rise in chronic disease occurrence, the growing adoption of generics, and a rising preference for biopharmaceuticals. Moreover, increasing adoption of advanced therapeutics, development of personalized medicines, and the introduction of innovative and novel delivery systems are expected to contribute to market growth.
The growing trend among major pharmaceutical companies to outsource, particularly in the realm of traditional Active Pharmaceutical Ingredient (API) Contract Development and Manufacturing Organizations (CDMO), is poised to drive industry demand. Additionally, rising investments in Research and Development (R&D) that are focused on refining API production processes are anticipated to propel advancement of this market segment.
The antibody-drug conjugate segment is expected to show lucrative growth during the forecast period.The effectiveness of antibody-drug conjugates (ADC) and checkpoint inhibitors in combating cancer is profoundly influencing the research and development landscape of ADCs. Moreover, there is a notable surge in exploring novel and compelling applications of antibody-drug conjugates. The ADC biopharmaceutical sector is witnessing a substantial uptick in R&D efforts, focusing on both combination therapies for cancer treatment and extending the application of ADCs beyond oncology to address various non-cancerous diseases.
Synthesis Insights
The synthetic segment accounted for the largest revenue share in 2023. This is attributed to the heightened accessibility of raw materials and the ease of synthesizing these molecules. The expected expiration of patents for various synthetic molecules in the coming years is poised to stimulate growth in this industry. Moreover, the use of synthetic APIs empowers developers to swiftly create and prototype applications, eliminating the need to wait for the actual APIs to be developed or become available. This expedites the development process, facilitating a faster time-to-market for new products and services.
The biotech segment is estimated to register a faster CAGR over the forecast period. This growth is being propelled by factors such as increased demand for biopharmaceuticals and enhanced molecular efficiency. Moreover, the advancement of this segment can be attributed to substantial investments in the biotechnology and biopharmaceutical industries. While biotech active pharmaceutical ingredients currently represent a limited share of the overall active pharmaceutical ingredient market, the sector is poised for a positive outlook. This stems from the rapid expansion of the biopharmaceutical industry and growing utilization of biotech drugs.
Drug Insights
The innovative segment dominated the market in 2023. Segment growth is propelled by factors such as a rise in FDA approvals for new molecular entities, the elevated cost of innovative active pharmaceutical ingredients compared to generic pharmaceuticals, and a heightened emphasis on research and development. An extensive research in this field has led to a pipeline containing numerous unique molecules, anticipated to be launched during the forecast period. The high share of this segment is majorly due to a growing pace of new drug approvals in recent years. For instance, as per U.S. FDA, around 46 novel drugs were approved as of 2023.
The generic segment has been anticipated to showcase lucrative growth over the forecast period. The expansion of the generic pharmaceutical sector is primarily attributed to the patent expiration of branded drugs and cost-effectiveness associated with generics. The growing number of patents set to expire, with approximately 190 drugs expected to lose exclusivity by 2030 according to the U.S. FDA, presents a substantial market opportunity for this segment. Among these, 69 are considered blockbuster drugs, contributing to the anticipated growth in this segment.
Application Insights
The oncology segment dominated the market in 2023. The market is growing due to the rising number of cancer cases globally. Thus, a rising demand for new cancer therapies is anticipated to be the outcome of this steady increase in incidence rates. The growing presence of novel drugs in cancer is expected to create further opportunities for this segment. For instance, as per the U.S. FDA, as of 2023, the global oncology pipeline includes 16 oral medications and 6 injectable drugs, with 6 drugs dedicated to breast cancer and 3 aimed at addressing non-small cell lung cancer (NSCLC).
The glaucoma segment has been anticipated to experience lucrative growth over the forecast period. This is largely attributed to the rising prevalence of diabetes, the growing elderly population, and an increase in healthcare spending globally. According to the WHO, glaucoma is the world's second-largest cause of blindness. Thus, a rapidly growing need for lowering the disease burden of glaucoma is likely to support market growth of this segment in the near future.
Workflow Insights
The commercial segment held a dominant market share in 2023. The rising global disease burden supports segment growth, boosting demand for active pharmaceutical ingredients. For instance, an article published by the Europa Group states that, as of September 2023, cardiovascular disease (CVD) continues to remain the primary cause of death in both men and women. Approximately 620 million individuals worldwide are affected by heart and circulatory conditions.
Every year, approximately 60 million people globally are diagnosed with heart or circulatory diseases. Thus, the increasing prevalence of chronic diseases and mortality rates is driving the global demand for CDMO operations in the production of Active Pharmaceutical Ingredients (APIs). Furthermore, key market players are focusing on expanding their presence by improving their API manufacturing capabilities. For instance, in June 2022, Merck & Co., Inc. announced a capacity expansion at its High Potency Active Pharmaceutical Ingredient production facility in Wisconsin, the U.S., to enhance its ability to produce cancer medication.
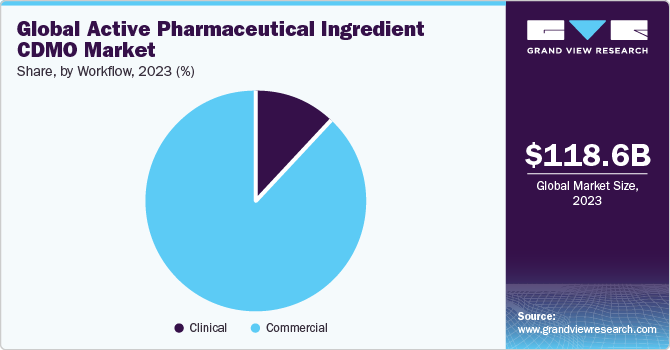
The clinical segment has been anticipated to showcase lucrative growth over the forecast period.A substantial rise in the volume of molecules advancing through preclinical and clinical development pipelines, accompanied by increased investment in research and development activities, stands as a significant contributing factor. This surge reflects the global demand for novel treatment options, leading to more frequent engagement in clinical research activities and thereby fostering growth within this segment.
Regional Insights
Asia Pacific dominated the overall Active Pharmaceutical Ingredient CDMO market with a revenue share of 42.3% in 2023. The regional market is anticipated to be driven by factors such as an enhanced regulatory framework, substantial opportunities for cost savings, heightened complexity, and robust drug pipelines. Additionally, the availability of a skilled workforce at a lower cost compared to developed economies, such as the U.S., is expected to further propel market expansion.
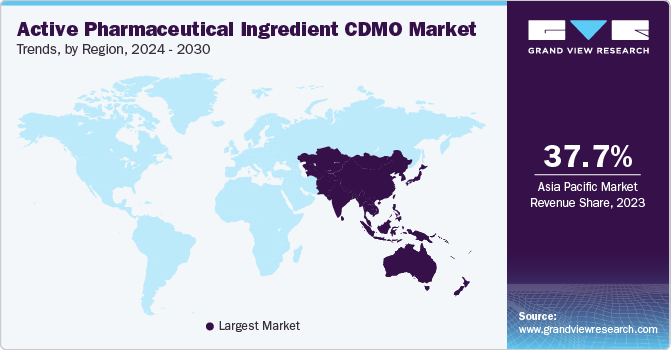
Countries such as India and China are the largest manufacturers of APIs globally, primarily due to the high pharmaceutical R&D investment and well-established CDMOs across Asian countries. Furthermore, India is anticipated to witness the fastest CAGR during the analysis timeframe in this region, with the COVID-19 pandemic highlighting the importance of strengthening the supply chain of pharmaceuticals. The Indian government is taking several initiatives to eliminate its dependencies on China for APIs by augmenting production facilities and investing in R&D activities to bolster domestic production of APIs. These aforementioned factors are responsible for this regional dominance.
China accounted for the largest revenue share in the Active Pharmaceutical Ingredient (API) CDMO market in Asia Pacific region in 2023. The country offers a robust infrastructure, cost advantages, and a skilled workforce, making it an attractive destination for pharmaceutical manufacturing. Additionally, China's vast chemical and pharmaceutical manufacturing capabilities, coupled with its ability to produce a wide range of APIs, have positioned it as a key player in the regional and global API market.
North America is expected to hold a substantial market share due to the presence of several established biotechnology and pharmaceutical companies. Rising R&D investments made by life sciences and pharmaceutical companies are anticipated to fuel the demand for API contract manufacturing in this region. Stringent regulations governing manufacturing and product quality are likely to create growth opportunities for domestic contract manufacturing services. The rising regulatory emphasis on quality control in manufacturing stands as another crucial factor expected to propel market growth during the forecast period.
The U.S. accounted for a dominant revenue share in the API CDMO market in North America in 2023. The U.S. pharmaceutical industry is characterized by significant research and development activities, a robust regulatory framework, and a high concentration of major pharmaceutical companies. This environment fosters a strong demand for outsourced API development and manufacturing services, driving the growth of the API CDMO market.
Key Active Pharmaceutical Ingredient CDMO Company Insights
Some key players operating in this market include Cambrex Corporation, CordenPharma International, and Recipharm AB
-
Cambrex Corporation offers a wide range of technologies and capabilities, such as highly potent APIs, generic API manufacturing, custom API manufacturing, clinical API manufacturing, and analytical services.
-
Recipharm AB offer a range of services, including pharmaceutical development and manufacturing, sterile fill and finish development and manufacturing, and development and GMP manufacture of ATMPs (Plasmid, xRNA, Viral vectors, Microbiome)
-
Siegfried Holding AG and Piramal Pharma Solutions are some notable emerging market players in the Active Pharmaceutical Ingredient CDMO industry.
- Siegfried Holding AG offers a range of API CDMO services, including the development and manufacturing of active pharmaceutical ingredients (APIs), intermediates, and finished dosage forms.
Key Active Pharmaceutical Ingredient CDMO Companies:
The following are the leading companies in the active pharmaceutical ingredient CDMO market. These companies collectively hold the largest market share and dictate industry trends. Financials, strategy maps & products of these active pharmaceutical ingredient CDMO companies are analyzed to map the supply network.
- Cambrex Corporation
- Recipharm AB
- Thermo Fisher Scientific Inc. (Pantheon)
- CordenPharma International
- Samsung Biologics
- Lonza
- Catalent, Inc.
- Siegfried Holding AG
- Piramal Pharma Solutions
- Boehringer Ingelheim International GmbH
Recent Developments
-
In November 2023, Hovione expanded its nasal drug delivery through a partnership with IDC (Industrial Design Consultancy), introducing innovative nasal powder devices for local, systemic, and nose-to-brain drug delivery. The partnership enhances Hovione's integrated nasal drug development and manufacturing services
-
In May 2023, GHO Capital and Partners Group announced an investment in Sterling Pharma Solutions, a leading CDMO. The investment supports Sterling's global growth, production capacity expansion, and strategic acquisitions, with GHO remaining the majority shareholder
-
In April 2023, Cognizant announced a strategic partnership with Boehringer Ingelheim, a prominent research-focused biopharmaceutical firm, with an aim to accelerate the pace and enhance the quality of medicinal therapy developments
-
In March 2023, NAGASE and HALIX partnered to offer comprehensive contract development and manufacturing services for biopharmaceuticals and APIs in Japan. The collaboration leverages HALIX's expertise in biomanufacturing, expanding NAGASE's presence in the high-molecular pharmaceuticals space
-
In November 2022, Exelixis, Inc. signed a new license agreement with Catalent’s subsidiary, Redwood Bioscience. As per the agreement, Exelixis, Inc. paid USD 30 million to Catalent for developing 3 ADC programs for the former
-
In May 2022, Piramal Pharma Solutions announced the successful launch of its new active pharmaceutical ingredient (API) facility in Aurora, Ontario, which at that time had already commenced operations and completed its initial production runs
Active Pharmaceutical Ingredient CDMO Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 107.2 billion
Revenue forecast in 2030
USD 164.5 billion
Growth rate
CAGR of 7.4% from 2024 to 2030
Base year for estimation
2023
Historical data
2018 - 2022
Forecast period
2024 - 2030
Quantitative units
Revenue in USD million/billion and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, synthesis, drug, application, workflow, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; U.K.; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; Colombia; Chile; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Cambrex Corporation; Recipharm AB; Thermo Fisher Scientific Inc. (Pantheon); CordenPharma International; Samsung Biologics; Lonza; Catalent, Inc.; Siegfried Holding AG; Piramal Pharma Solutions; Boehringer Ingelheim International GmbH
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope. Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options Global Active Pharmaceutical Ingredient CDMO Market Report Segmentation
This report forecasts revenue growth at the global, regional & country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For the purpose of this study, Grand View Research has segmented the global Active Pharmaceutical Ingredient CDMO market report on the basis of product, synthesis, drug, application, workflow, and region:
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Traditional Active Pharmaceutical Ingredient (Traditional API)
-
Highly Potent Active Pharmaceutical Ingredient (HP-API)
-
Antibody Drug Conjugate (ADC)
-
Others
-
-
Synthesis Outlook (Revenue, USD Million, 2018 - 2030)
-
Synthetic
-
Biotech
-
-
Drug Outlook (Revenue, USD Million, 2018 - 2030)
-
Innovative
-
Generics
-
-
Workflow Outlook (Revenue, USD Million, 2018 - 2030)
-
Clinical
-
Commercial
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Oncology
-
Hormonal
-
Glaucoma
-
Cardiovascular disease
-
Diabetes
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
U.K.
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
India
-
Japan
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
Colombia
-
Chile
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The synthetic segment dominated the API CDMO market and accounted for the largest revenue share of 73.0% in 2023.
b. The innovative drugs segment dominated the API CDMO market and accounted for the largest revenue share of 73.3% in 2023.
b. The oncology segment dominated the active pharmaceutical ingredient CDMO market and accounted for the largest revenue share of 35.7% in 2023.
b. Based on workflow, the commercial segment led the API CDMO market and accounted for the largest revenue share of 87.9% in 2023.
b. Asia Pacific dominated the active pharmaceutical ingredient CDMO market and accounted for the largest revenue share of 42.25% in 2023.
b. The global active pharmaceutical ingredient CDMO market size was estimated at USD 100.4 billion in 2023 and is expected to reach USD 107.2 billion in 2024.
b. The global active pharmaceutical ingredient CDMO market is expected to grow at a compound annual growth rate of 7.4% from 2024 to 2030 to reach USD 164.5 billion by 2030.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





