- Home
- »
- Medical Devices
- »
-
Biopharmaceuticals Contract Manufacturing Market Report, 2030GVR Report cover
![Biopharmaceuticals Contract Manufacturing Market Size, Share, & Trend Report]()
Biopharmaceuticals Contract Manufacturing Market Size, Share, & Trend Analysis Report By Source, By Service (Upstream, Downstream, Fill/Finish), By Product (Biologics), By Region And Segment Forecasts, 2022 - 2030
- Report ID: GVR-2-68038-698-1
- Number of Pages: 193
- Format: Electronic (PDF)
- Historical Range: 2017 - 2020
- Industry: Healthcare
Report Overview
The global biopharmaceuticals contract manufacturing market was valued at USD 17.0 billion in 2021 and is projected to grow at a compound annual growth rate (CAGR) of 5.2% during the forecast period. The success of the biopharmaceutical market can be majorly attributed to the contract manufacturers. Reduction in the overall investment required to bring a new drug product to market, providing access to expensive technologies, quick entry of products into the markets, and greater flexibility are some advantages offered by contract manufacturing organizations (CMOs), which have prompted the companies to outsource their biopharmaceutical manufacturing.
Due to the disruptions from COVID-19, many companies have had to bring on new contract manufacturers or ingredient suppliers due to increasing demands. Furthermore, with a perpetual expansion of the biopharmaceutical industry, the companies are facing production issues, such as lack of expertise and sophisticated equipment, while practicing in-house manufacturing. The maturity of biotechnology and the availability of external funding have resulted in the growth in a number of early-stage bio/pharma companies. These companies are recognized as core customers of CMOs, as these organizations lack the capabilities for the development of robust manufacturing operations.
In order to fulfill the growing demand in the market, key players are going for capacity expansion. For instance, in 2020, Thermo Fisher Scientific announced an investment of USD 180.0 million for the construction of a new commercial manufacturing site in Plainville, Mass for viral vector development and manufacturing services. Also, in 2019, Boehringer Ingelheim invested USD 84 million for expansion in Mexico for increasing production. The customers and CMOs are engaged in standardizing agreement terms for making contract negotiations easy. This is to address the issues pertaining to the regulatory landscape and complexity of service delivery. IP rights, warranty & liabilities, prices & timelines are major issues cited by CMOs and clients which are making negotiations innately difficult.
Breakthrough technological advancements and innovations in bioprocessing have played a pivotal role in the progress of contract service providers by overcoming the manufacturing issues such as high production costs and the need for changeover with every batch. Single-use bioprocessing systems are one of the most significant innovations as it helps in reducing the overall production and scale-up costs. Furthermore, the fast turnaround offered by single-use products while limiting allied activities, such as changeover and cleaning validation, has supported the growth of CMOs to a major extent.
Mergers, acquisitions, and joint ventures are one of the common trends observed across the industry, as it helps CMOs offer integrated bioprocessing services to their clients, which, in turn, makes them a more reliable option for a rapid product launch for commercial use. However, large firms consider outsourcing perilous due to loss of strategic control and limited management oversight. As a result, large pharma companies opt to maintain their manufacturing operations in-house. This is expected to challenge the growth of CMOs to a certain extent.
Source Insights
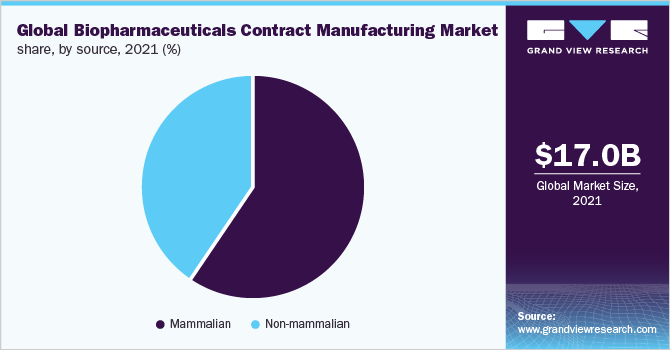
The mammalian source segment led the market in 2021 and accounted for the highest share of 59%. Based on the source, the market is dived into mammalian and non-mammalian-based contract manufacturing. The intensive investigation that is being carried out on the usage of various sources and species for biopharmaceutical development is expected to drive market growth. The main objective of these investigations is to enhance the productivity and efficiency of biopharmaceutical manufacturing. Mammalian-based biomanufacturing contributed to a higher revenue share owing to the higher cost associated with obtaining products from these sources. Companies engaged in providing contract services using mammalian cell culture include AbbVie Contract Manufacturing, AMRI, Avid Bioservices, Boehringer Ingelheim Biopharmaceuticals Gmbh, and Catalent Pharma Solutions.
In addition to this, a major percentage of biopharmaceutical products that are in pipeline are mammalian expressed, hence expecting to maintain the dominance over the forecast period as well. Consequently, companies like Lonza and Charles River are making significant investments in the expansion of their mammalian cell culture manufacturing facilities for biologics and biosimilar development. For instance, in October 2017 Lonza acquired a clinical-stage mammalian manufacturing site in the United States from Shire plc. The plant is equipped with single-use bioreactors of 1,000L and 2,000L capacities, coupled with downstream purification capabilities.
Non-mammalian cell line -E. Coli is recognized as the widely adopted non-mammalian cell culture for biopharmaceutical production owing to its rapid access and cost-effective cultivation. The development of a transgenic non-mammalian expression system holds a great promise for the significant growth of this segment throughout the forecast period. Abbott Bioresearch, Avecia Biotechnology, BioReliance, Biovitrum AB, Dow Pharmaceutical, and Celltrion are a few companies working as CMOs using microbial cultures.
Service Insights
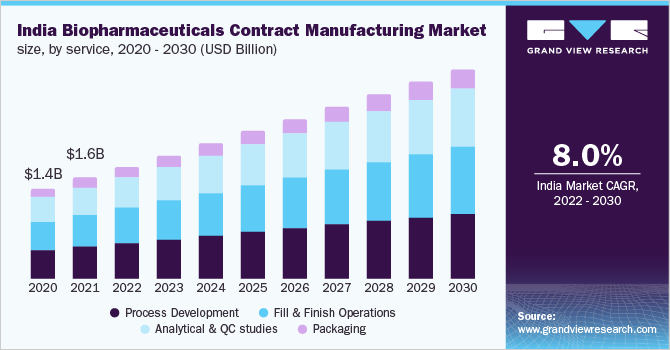
Process development services led the global market in 2021 and accounted for a share of about 33%. This is due to high capital expenditure in downstream processing. Moreover, downstream operations demand vigorous attention for final product recovery and purification steps to maintain product quality and prevent wastage. CMOs have provided biopharma players with a wide array of services ranging from cell cultivation to the final packaging of the product. The manufacturing services offered by CMOs are process development, fill & finish operations, analytical & QC studies, and packaging.
Process development services dominated in 2021 with respect to revenue with downstream processing leading the market. This is due to high capital expenditure in downstream processing. Moreover, downstream operations demand vigorous attention for final product recovery and purification steps, to maintain product quality and prevent wastage. With growing quality concerns and regulatory changes for biopharmaceutical development, analytical & QC studies are expected to grow lucratively during the forecast period. New regulations are being introduced for the production of biologics by CMOs in compliance with regulatory standards to maintain product purity and safety.
Product Insights
The biologics product segment led the market in 2021 with a share of over 81%. Contract manufacturers have played a vital role in the success of both biologics and biosimilars. This is majorly due to the huge commercial success of biologics, which is depicted through the presence of a large number of FDA-approved biologics in the market.
Among all the biologics, Monoclonal Antibodies (MAb) have captured the largest share in 2021. A high capital requirement for the construction of a MAb plant has accelerated the uptake of contract services for Mab production, thereby contributing to the major share of this segment.
However, biosimilars production is considered one of the key strategies for business expansion in comparison with biologics because investment in biosimilars manufacturing helps in the fast market reach of the biopharmaceuticals. Moreover, biosimilars have supported the biopharmaceutical CMO industry with respect to cost-saving advantages.
Regional Insights
With a high number of biomanufacturing facilities in the U.S., North America dominated with the largest revenue share of 34% in 2021. Furthermore, revenue from biopharmaceutical-based R&D activities held a major percentage of the total R&D carried out in various sectors in the U.S. Furthermore, interventions & conferences conducted in the region for pertinent technologies and issues in the application of biologics for the treatment of various chronic conditions are anticipated to positively influence regional growth.
However, Asian countries are expected to emerge as the attractive outsourcing location for the biomanufacturing of large molecules. Low manufacturing and operating cost offered by countries, like China & India, is one key factor driving the Asian market. Faster growth in the Korean market is expected to boost revenue generation from the APAC region.
Key Companies and Market Share Insights
The market service providers are focused on expanding their manufacturing capabilities as well as establishing new services to meet the growing demand of biopharmaceutical companies. Along with small players, these entities are also engaged in a partnership with established biopharma companies.
All the major biopharmaceutical firms have a wide-ranging product pipeline and are investing in developing new products. For instance, more than 60,000 clinical trials were registered globally in 2018. It is challenging for biopharmaceutical players to carry out regulatory-compliant manufacturing processes solely depending on in-house capacities. This, in turn, is driving the demand for biopharmaceutical contract manufacturing of potential drug candidates all across the globe. Some prominent key players in the global biopharmaceuticals contract manufacturing market include:
-
Boehringer Ingelheim GmbH
-
Lonza
-
Inno Biologics Sdn Bhd
-
Rentschler Biotechnologie GmbH
-
JRS PHARMA
-
AGC Biologics
-
ProBioGen
-
FUJIFILM Diosynth Biotechnologies U.S.A., Inc.
-
Toyobo Co. Ltd.
-
Samsung BioLogics
-
Thermo Fisher Scientific, Inc.
-
Binex Co., Ltd.
-
WuXi Biologics
-
AbbVie, Inc.
-
Novartis AG
-
ADMA Biologics, Inc.
-
Catalent, Inc
-
Cambrex Corporation
-
Pfizer Inc.
-
Siegfried Holding AG
Biopharmaceuticals Contract Manufacturing Market Report Scope
Report Attribute
Details
Market size value in 2022
USD 18.18 billion
Revenue forecast in 2030
USD 27.3 billion
Growth rate
CAGR of 5.2% from 2022 to 2030
Base year for estimation
2021
Actual estimates/Historical data
2017 - 2020
Forecast period
2022 - 2030
Quantitative units
Revenue in USD Million and CAGR from 2022 to 2030
Report coverage
Revenue forecast, company share, competitive landscape, growth factors, and trends
Segments covered
Source, service, product, therapeutic area, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S., Canada, UK, Germany, France, Italy, Spain, India, China, Japan, Australia, South Korea, Brazil, Colombia, Mexico, Argentina, South Africa, Saudi Arabia, UAE
Key companies profiled
Boehringer Ingelheim GmbH; Lonza; Inno Biologics Sdn Bhd; Rentschler Biotechnologie GmbH; JRS Pharma; AGC Biologics; ProBioGen; Fujifilm Diosynth Biotechnologies U.S.A., Inc.; Toyobo Co., Ltd.; Samsung BioLogics; Thermo Fisher Scientific, Inc.; Binex Co., Ltd.; WuXi Biologics; AbbVie, Inc; Novartis AG; ADMA Biologics, Inc.; Catalent, Inc; Cambrex Corporation; Pfizer Inc.; Siegfried Holding AG
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Segments Covered in the Report
This report forecasts revenue growth at global, regional, & country levels and provides an analysis of industry trends in each of the sub-segments from 2017 to 2030. For this study, Grand View Research has segmented the global biopharmaceuticals contract manufacturing market based on source, service, product, therapeutic area and region:
-
Source Outlook (Revenue, USD Million, 2017 - 2030)
-
Mammalian
-
Non-mammalian
-
-
Service Outlook (Revenue, USD Million, 2017 - 2030)
-
Process Development
-
Downstream
-
Upstream
-
-
Fill & Finish Operations
-
Analytical & QC studies
-
Packaging
-
-
Product Outlook (Revenue, USD Million, 2017 - 2030)
-
Biologics
-
Monoclonal antibodies (MABs)
-
Recombinant Proteins
-
Vaccines
-
Antisense, RNAi, & Molecular Therapy
-
Others
-
-
Biosimilars
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2017 - 2030)
-
Oncology
-
Autoimmune Diseases
-
Metabolic Diseases
-
Cardiovascular Diseases
-
Neurology
-
Infectious Diseases
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2017 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
-
Asia Pacific
-
China
-
India
-
Japan
-
Australia
-
South Korea
-
-
Latin America
-
Brazil
-
Colombia
-
Mexico
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
-
Frequently Asked Questions About This Report
b. The global biopharmaceuticals contract manufacturing market size was estimated at USD 17.0 billion in 2021 and is expected to reach USD 18.18 billion in 2022.
b. The global biopharmaceuticals contract manufacturing market is expected to grow at a compound annual growth rate of 5.2% from 2022 to 2030 to reach USD 27.3 billion by 2030.
b. North America dominated the biopharmaceuticals contract manufacturing market with a share of 34% in 2021. This is attributable to a reduction in overall investment required to bring a new drug product to market, providing access to expensive technologies, and quick entry of products in markets.
b. Some key players operating in the biopharmaceuticals contract manufacturing market include Boehringer Ingelheim GmbH; Lonza; Inno Biologics Sdn Bhd; Rentschler Biotechnologie GmbH; JRS PHARMA; AGC Biologics; ProBioGen; FUJIFILM Diosynth Biotechnologies U.S.A., Inc.; TOYOBO CO., LTD; Samsung BioLogics; Thermo Fisher Scientific, Inc.; Binex Co., Ltd.; WuXi Biologics; and AbbVie, Inc.
b. Key factors that are driving the biopharmaceuticals contract manufacturing market growth include growing quality concerns and regulatory changes for biopharmaceutical development, and analytical and QC studies.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





