- Home
- »
- Medical Devices
- »
-
Facial Implants Market Size, Share & Growth Report, 2030GVR Report cover
![Facial Implants Market Size, Share & Trends Report]()
Facial Implants Market Size, Share & Trends Analysis Report By Product (Cheek, Nasal, Injectable), By Procedure (Rhinoplasty, Facelift), By Material (Metal, Biologics, Ceramic, Polymers), By Region, And Segment Forecasts, 2023 - 2030
- Report ID: 978-1-68038-500-7
- Number of Pages: 110
- Format: Electronic (PDF)
- Historical Range: 2018 - 2021
- Industry: Healthcare
Report Overview
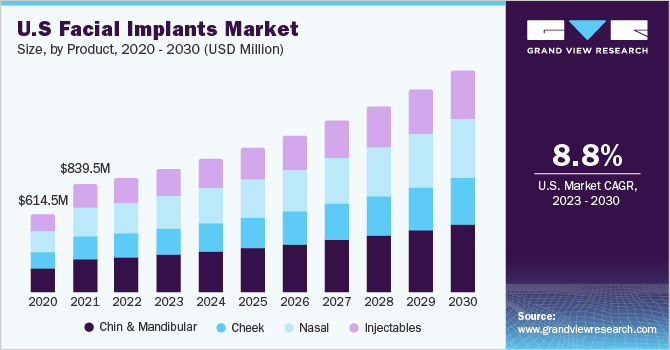
The global facial implants market size was estimated at USD 2.65 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 8.5% from 2023 to 2030. Facial implants are medical devices designed to correct facial structure and enhance facial features, such as the chin, cheeks, and jawline. The market for facial implants is anticipated to witness sustained growth during the forecast period, primarily driven by the increasing prevalence of virtual presence on social media platforms. This virtual representation trend is expected to boost the demand for facial implants as individuals seek to achieve more defined and aesthetically pleasing facial characteristics. In addition, the growing emphasis on personal appearance in both developed and developing countries is projected to contribute to the expansion of the cosmetic surgery industry.
The market is projected to experience a positive trajectory throughout the forecast period, primarily driven by the growing emphasis on physical appearance and aesthetics across all age groups, coupled with the rising adoption of cosmetic procedures on a global scale. This surge in demand for plastic surgeries is expected to fuel market growth. Moreover, the advantages associated with invasive aesthetic surgery, such as painless procedures, enhanced safety and efficacy, shorter recovery times, and reduced hospital stays, are anticipated to further drive the demand for such procedures. In addition, the increasing incidence of facial deformities resulting from congenital disorders and traumatic injuries requiring cosmetic surgery is expected to contribute to the growing demand for facial implants in the coming years.
The medical device industry has faced significant disruptions in its supply chain due to the COVID-19 pandemic. This global outbreak has led to a reduction in the number of annual procedures performed, resulting in an overall market decline. The impact of the coronavirus has been detrimental to the market, with a decrease in procedures observed in 2020 and 2021 due to global restrictions. The American Society for Aesthetic Plastic Surgeons reported a notable 15% decline in the total number of cosmetic procedures in 2020, directly attributed to the pandemic. Despite these challenges, the industry is expected to grow as a result of the increasing adoption of cosmetic surgeries among celebrities and the growing awareness regarding facial implants. However, it is essential to acknowledge potential complications associated with these procedures, such as excessive bleeding and variable success rates, which could impede market growth.
Product Insights
The chin and mandibular segment emerged as the market leader in 2022, capturing the largest revenue share of 30.3%. With a revenue of USD 804.7 million, this segment demonstrated its dominant position in the market. The key factors contributing to its supremacy over the forecast period include the rising demand for chin augmentation procedures to achieve facial rejuvenation and the increasing rate of adoption of such interventions. Chin enhancement surgery, which involves the placement of an implant to enhance the shape and size of the chin, offers a permanent solution for individuals seeking to improve a recessed or weak chin.
However, the injectables segment is anticipated to register a lucrative CAGR of over 8.9% in the coming years owing to the increasing demand for cost-effectiveness, nonsurgical facial rejuvenation procedures, and growing awareness about the use of fillers for augmentation surgery. Facial injectables such as hyaluronic acid and botulinum toxin were among the most popular products among females. The segment is likely to grow due to the increasing use of facial fillers for aesthetic procedures such as wrinkles on the face, facial lines, lip lines, and others.
Material Insights
The polymer segment dominated the market and accounted for the largest revenue share of 38.2% in 2022. Polymer material dominated the market in terms of revenue as of 2022 due to the increasing usage for deformity correction surgeries and the rise in demand for polymer materials. Polymeric materials have considerable tensile strength and are best suited for facial implants. Furthermore, other benefits associated with it including flexibility, rigid support, and biocompatibility are anticipated to fuel the market growth. The most frequently used implants are solid silicone implants that are used for augmentation surgery.
The biologicals segment is expected to witness the most lucrative CAGR of 8.9% during the forecast period owing to the greater resemblance to the body tissues enabling easy adoption. Furthermore, the increasing investments in the R&D of biocompatible implant manufacturing are also projected to propel market growth in the future.
Procedure Insights
The eyelid segment dominated the market and accounted for the largest revenue share of 39.5% in 2022. The number of procedural implants is expected to rise due to the rising demand for facial surgeries globally. Eyelid surgery held the largest share in 2022 owing to the increasing number of aesthetic procedures for reducing bagginess, correcting vision, and removing extra skin from eyelids. Eyelid surgery or blepharoplasty is a long-term procedure, the outcomes of which the patient can enjoy for many years, hence the number of procedures performed globally constantly growing.
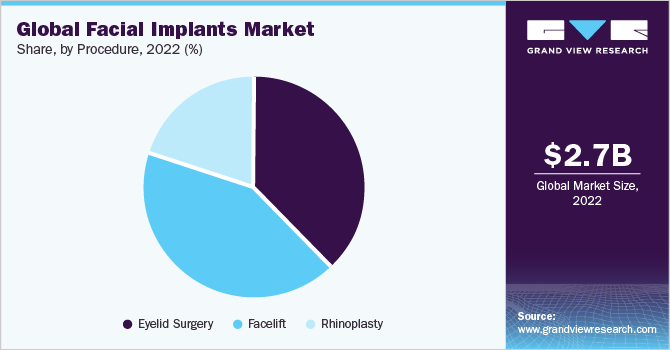
Eyelid surgery is painless, performed under local anesthesia, and lasts up to an hour. Immediately after the procedure, the patient is free to go home. All these factors are also boosting the eyelid surgery segment market growth. According to ASPS, about 352,112 eyelids and 234,374 facelift procedures were performed in the U.S. in 2020 and are expected to grow over the forecast period. Facelift surgery is projected to gain pace in the coming years due to the growing awareness of aesthetics, especially among baby boomers. According to the stats published by The American Society of Plastic Surgeons (ASPS), nose reshaping facelift and eyelid surgery were the topmost cosmetic surgeries performed in the U.S. in 2020.
Regional Insights
North America emerged as the dominant market in 2022, capturing a significant revenue share of more than 35.5%. This market dominance can be attributed to the increasing incidence of facial distortion resulting from extensive commercialization. Additionally, the region benefits from factors such as strong patient awareness, growing demand for minimally invasive surgeries, the introduction of technologically advanced facial implants, and the presence of advanced healthcare infrastructure tailored to facial procedures. The rising emphasis on facial aesthetics and the desire to maintain a youthful appearance among the population is also expected to drive the demand for facial surgeries in the future.
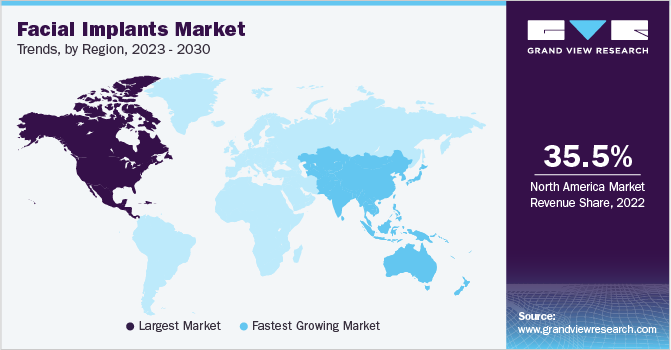
In Asia Pacific, the market is expected to witness the highest CAGR during the forecast period. The presence of high untapped opportunities, economic development, rising patient awareness levels, and constantly improving healthcare infrastructure are some key factors driving the Asia Pacific market. In addition, increasing medical tourism activities supported by low procedural costs in the emerging economies of India and China are expected to drive industry growth over the next seven years.
Key Companies & Market Share Insights
Key companies have established key business strategies such as strategic partnerships and collaborations, product innovation, joint ventures, new product launches, new service launches, and contracts to build their market position along with gaining popularity in the market. The market is competitive as key players are engaged in rigorous R&D to develop efficient and cost-effective products for reconstructive surgery and trauma treatment.
For instance, in February 2022, Zimmer Biomet and Bactiguard agreed to expand the global license agreement that was started in 2019, to cover a range of implant product segments to prevent post-operative infections. The exclusive license partnership includes implants for sports medicine, joint reconstruction (hips and knees), cranial, and maxillofacial and thoracic applications. Some prominent players in the global facial implants market include:
-
Zimmer Biomet
-
Depuy Synthes
-
Stryker Corp.
-
Medartis AG
-
EUROS
-
Xilloc
-
KLS Martin Group
-
Hanson Medical, Inc.
-
Integra LifeSciences
-
OsteoMed
Facial Implants Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 2.86 billion
Revenue forecast in 2030
USD 5.06 billion
Growth rate
CAGR of 8.5% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Report updated
July 2023
Quantitative units
Revenue in USD million/billion and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Product, material, procedure, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Germany; UK; France; Italy; Spain; Norway; Denmark; Sweden; China; Japan; India; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Zimmer Biomet; Depuy Synthes; Stryker; Medartis AG; EUROS; Xilloc; KLS Martin Group; Hanson Medical, Inc.; Integra LifeSciences; OsteoMed
Customization scope
Free report customization (equivalent up to 8 analyst working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Facial Implants Market Report Segmentation
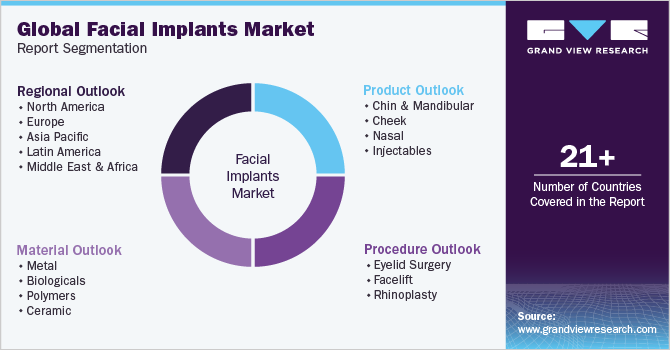
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global facial implants market report based on product, material, procedure, and region:
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Chin and Mandibular
-
Cheek
-
Nasal
-
Injectables
-
-
Material Outlook (Revenue, USD Million, 2018 - 2030)
-
Metal
-
Biologicals
-
Polymers
-
Ceramic
-
-
Procedure Outlook (Revenue, USD Million, 2018 - 2030)
-
Eyelid Surgery
-
Facelift
-
Rhinoplasty
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global facial implants market size was estimated at USD 2.65 billion in 2022 and is expected to reach USD 2.86 billion in 2023
b. The global facial implants market is expected to grow at a compound annual growth rate of 8.5% from 2023 to 2030 to reach USD 5.06 billion by 2030.
b. North America dominated the facial Implant market with a share of 35.5% in 2022. This is attributable to rising incidences of facial deformities due to extensive industrialization, introduction of technologically advanced facial implants, high patient awareness levels, rising demand for minimally invasive surgeries, and presence of sophisticated healthcare infrastructure
b. Some key players operating in the facial implants market include Zimmer Biomet; Depuy Synthes; Stryker; Medartis AG; EUROS; Xilloc; KLS Martin Group; Hanson Medical, Inc.; Integra LifeSciences; OsteoMed
b. Key factors that are driving the facial Implant market growth include rising adoption of minimally invasive procedures, increasing incidences of facial injuries, technological advancements in facial implant manufacturing and rise in medical tourism.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





