- Home
- »
- Biotechnology
- »
-
Molecular Cytogenetics Market Size & Share Report, 2030GVR Report cover
![Molecular Cytogenetics Market Size, Share & Trends Report]()
Molecular Cytogenetics Market Size, Share & Trends Analysis Report By Application (Oncology, Personalized Medicine), By Technology (FISH, Immunohistochemistry, Karyotyping), By Product, By End-use, By Region, And Segment Forecasts, 2023 - 2030
- Report ID: GVR-1-68038-023-1
- Number of Pages: 236
- Format: Electronic (PDF)
- Historical Range: 2018 - 2021
- Industry: Healthcare
Report Overview
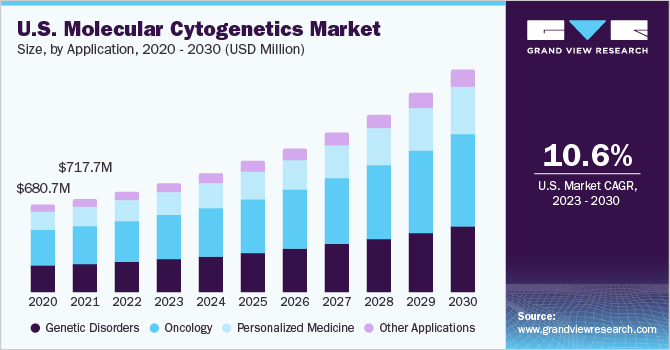
The global molecular cytogenetics market size was estimated at USD 2.02 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 10.6% from 2023 to 2030. The rising number of oncology cases, technological advancements in chromosome analysis tools, and the shift toward personalized medicine are the key factors boosting the market growth of molecular cytogenetics. Moreover, the increasing number of workshops and conferences for cytogenetic analysis is accelerating the molecular cytogenetic market. For instance, in April 2023, the University of Madras conducted a hands-on workshop on genomic techniques in clinical diagnostics, including cytogenetics.
The COVID-19 pandemic has had a positive impact on the molecular cytogenetics industry in several ways. Firstly, the urgent need for rapid and accurate diagnostic testing during the pandemic accelerated the development and implementation of advanced molecular cytogenetic techniques, enabling faster and more efficient detection of genetic abnormalities associated with COVID-19 and other diseases. Additionally, the pandemic has highlighted the value of genomic surveillance, leading to increased investment in molecular cytogenetic research and infrastructure, which has advanced our understanding of viral genomics and transmission dynamics.
With the growing volume of genomic data, the integration of multi-omics data and the development of bioinformatics tools and algorithms are fueling the demand for molecular cytogenetics. These tools facilitate efficient data management, analysis, interpretation, and the discovery of meaningful associations between genetic alterations and disease phenotypes. Thus, the growing genomic pool is anticipated to propel the market for molecular cytogenetics during the forecast period.
Cancer is a primary cause of death across the world, accounting for approximately 10 million deaths in the year 2020. According to a WHO article published in February 2022, every year, nearly 400,000 kids develop cancer. Molecular cytogenetics provides techniques for cytology, epigenetics, and imaging in genetics, as well as the ability to detect chromosomal changes in cancer patients. Thus, the increasing prevalence of cancer is expected to offer lucrative opportunities for the market during the study period.
Furthermore, the growing prevalence of uncommon infections is also expected to propel the demand for the market. The growing knowledge and correlation of features of the human genomic pool have paved the way for various research & development activities for developing personalized medicines. In addition, operating players in the market are introducing novel molecular cytogenetics tools for improved patient care and outcomes. For instance, in October 2021, Phase Genomics, Inc. launched a next-generation cytogenomics platform to advance precision diagnosis and treatment in oncology and reproductive genetics, thereby boosting market growth.
Application Insights
The oncology segment dominated the market for molecular cytogenetics and accounted for the largest revenue share of 39.7% in 2022, owing to the growing number of cancer cases across the world. Molecular cytogenetics enables the genetic characterization of tumors, providing insights into the genomic alterations and chromosomal abnormalities associated with cancer.In addition, the emergence of personalized medicine in oncology has propelled the use of molecular cytogenetics. By analyzing the genetic alterations in tumors, molecular cytogenetics helps identify therapeutic targets and select the most appropriate treatment strategies for individual patients. Thus, the growing oncology patient pool is anticipated to propel segment growth during the forecast period.
The personalized medicine segment is anticipated to witness the fastest CAGR during the forecast period. Rapid advancements in genomic technologies, such as next-generation sequencing (NGS) and microarray analysis, have significantly accelerated the adoption of molecular cytogenetics in personalized medicine. The emergence of targeted therapies, which aim to selectively inhibit specific molecular targets involved in disease pathways, has transformed the field of personalized medicine. Thus, the increasing clinical trials are anticipated to boost the demand for molecular cytogenetics.
Technology Insights
The comparative genomic hybridization segment dominated the molecular cytogenetics market and accounted for the largest revenue share of 36.5% in 2022. The development of CGH technology has significantly improved the resolution and sensitivity of chromosomal analysis. CGH allows for the detection of submicroscopic chromosomal alterations, including deletions, duplications, amplifications, and copy number variations (CNVs), which may not be visible by conventional cytogenetic techniques.
Moreover, array-based CGH, such as array comparative genomic hybridization (aCGH), has significantly propelled the use of CGH technology. It allows professionals to analyze chromosomes on DNA pulled out from tissue deprived of the need for active dividing cells. Thus, rising technological advancements in molecular cytogenetics technology are anticipated to propel segment growth during the forecast period.
The technology segment consists of comparative genomic hybridization, FISH, immunohistochemistry, karyotyping, and others. The other technologies segment is anticipated to witness the fastest CAGR over the forecast period. Single-cell sequencing technologies have gained significant momentum in recent years. These technologies enable the analysis of individual cells, allowing for the detection of genetic heterogeneity within tissues and tumor samples. Furthermore, bioinformatics plays a crucial role in the analysis and interpretation of large-scale genomic data generated by technologies such as NGS and array-based platforms. The rapid growth of molecular cytogenetics has been accompanied by increased demand for other technologies and data analysis pipelines.
Product Insights
The consumables segment dominated the market for molecular cytogenetics and accounted for the largest revenue share of 39.5% in 2022. The increasing awareness and understanding of genetic diseases, cancer, and personalized medicine have fueled the demand for diagnostic and research tests. This growth in demand directly translates into a higher need for consumables used in molecular cytogenetic testing. According to a CDC article published in April 2022, nearly 21.6%, or around 4 million people in the U.S., screened for cancer in the year 2020. This growing awareness among individuals is raising the demand for molecular cytogenetics, thus promoting segment growth.
The software & services segment is estimated to grow at the fastest rate during the forecast period. The rapid advancements in genomic technologies, such as next-generation sequencing (NGS) and array-based platforms, have helped generate vast amounts of data in molecular cytogenetics. Bioinformatics tools and data analysis algorithms are essential for managing and analyzing these large-scale genomic datasets. Hence, the increasing advancements in software are anticipated to fuel the demand for molecular cytogenetics.
End-use Insights
The clinical & research laboratories segment dominated the market with a revenue share of 32.2% in 2022. Advances in technology have revolutionized molecular cytogenetics. Moreover, it has become an essential tool in clinical diagnostics that enables the identification of chromosomal abnormalities associated with genetic disorders, cancer, and prenatal abnormalities. According to an article published by UC San Diego Health in April 2023, molecular cytogenetics helps patients by providing genetic testing services of chromosomal analysis to various healthcare professionals as well as patients for better individual health.
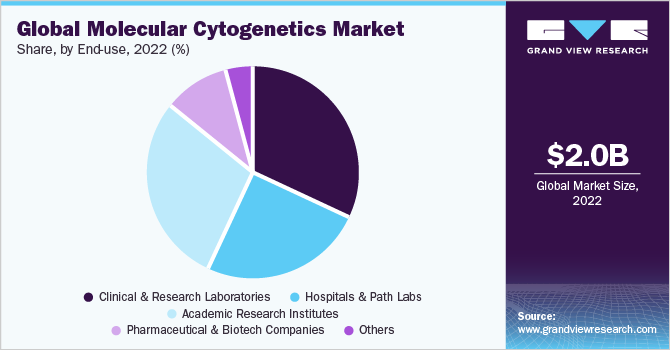
The pharmaceutical & biotech companies segment is projected to witness the fastest CAGR of 12.1% over the forecast period. Molecular cytogenetics helps identify specific genetic alterations and chromosomal abnormalities associated with diseases, including cancer. Pharmaceutical and biotech companies are increasingly focusing on developing targeted therapies that address these specific genetic variations. Moreover, technological advancements in molecular cytogenetics, such as high-resolution imaging, microarray analysis, and next-generation sequencing, are driving its growth in the pharmaceutical and biotech sectors. This is likely to favor segment growth in the market for molecular cytogenetics.
Regional Insights
In 2022, North America dominated the market with a revenue share of 45.97%, owing to the presence of major local entities such as Agilent Technologies, Inc. Presence of advanced healthcare infrastructure, increasing research funding in molecular cytogenetics, growing awareness about advanced laboratory techniques, and high incidence of genetic and chronic diseases are the key factors driving regional growth. For instance, according to the CDC, about 6,000 babies are born with Down syndrome in the U.S. every year. The existence of an efficient regulatory structure for the administration of genetic tests has had a positive impact on market growth.
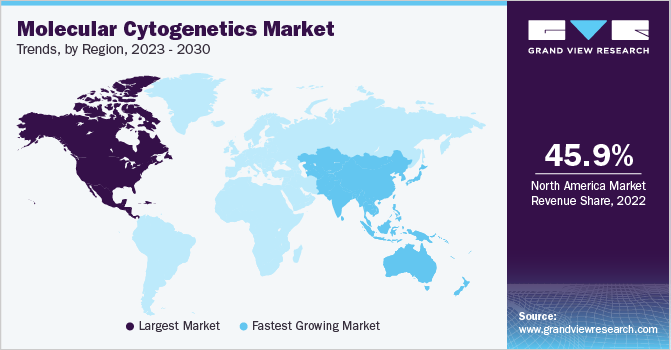
Asia Pacific is projected to witness the fastest CAGR over the forecast period. Continuous GDP growth of countries such as China & India is a factor anticipated to enhance consumer buying power and drive regional growth. Various workshops and interventions in the region, as well as advancements in disease management and human genetics, are propelling the Asian market. These scenarios are anticipated to improve awareness about the significance of advanced therapies and diagnostics among the regional population, thereby boosting market growth in the coming years.
Key Companies & Market Share Insights
Companies are focusing on expansions, the development of innovative medical devices, and technological advances. Moreover, partnerships, mergers, and acquisitions for novel product development constitute some of the strategic initiatives implemented by major players. For instance, in June 2023, Oxford Gene Technology (OTG) entered into a partnership with Applied Spectral Imaging (ASI) to strengthen OTG’s cytogenetic imaging and analysis solutions in Great Britain. The initiative is predicted to offer enhancements in workflow automation and overall efficacy to enable quicker diagnostic decision-making. Some of the key players operating in the global molecular cytogenetics market include:
-
BIOVIEW
-
Danaher
-
MetaSystems
-
Agilent Technologies, Inc.
-
Abbott
-
Bio-Rad Laboratories, Inc.
-
Illumina, Inc.
-
Oxford Gene Technology
-
F. Hoffmann-La Roche Ltd
-
PerkinElmer Inc.
Molecular Cytogenetics Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 2.19 billion
Revenue forecast in 2030
USD 4.46 billion
Growth Rate
CAGR of 10.6% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Report Updated
September 2023
Quantitative units
Revenue in USD Billion/Million and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Application, technology, product, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; Japan; China; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
BIOVIEW; Danaher; MetaSystems; Agilent Technologies, Inc.; Abbott; Bio-Rad Laboratories, Inc.; Illumina, Inc.; Oxford Gene Technology; F. Hoffmann-La Roche Ltd.; PerkinElmer Inc.
Customization scope
Free report customization (equivalent up to 8 analysts’ working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Molecular Cytogenetics Market Report Segmentation
This report forecasts revenue growth at the global, regional & country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global report based on application, technology, product, end-use, and region:
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Genetic Disorders
-
Oncology
-
Personalized Medicine
-
Other Applications
-
-
Technology Outlook (Revenue, USD Million, 2018 - 2030)
-
Comparative Genomic Hybridization
-
aCGH
-
sCGH
-
-
FISH
-
Immunohistochemistry
-
Karyotyping
-
Spectral
-
Virtual
-
-
Others
-
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Instruments
-
Consumables
-
Software & Services
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Clinical & Research Laboratories
-
Hospitals & Path Labs
-
Academic Research Institutes
-
Pharmaceutical & Biotech Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global molecular cytogenetics market size was estimated at USD 2.02 billion in 2022 and is expected to reach USD 2.19 billion in 2023.
b. The global molecular cytogenetics market is expected to grow at a compound annual growth rate of 10.6% from 2023 to 2030 to reach USD 4.46 billion by 2030.
b. Comparative genomic hybridization dominated the molecular cytogenetics market with a share of 36.49% in 2022. This is attributable to its wide adoption across various applications such as diagnostics, research activities, and medicine development.
b. Some key players operating in the molecular cytogenetics market include Agilent Technologies, Inc.; Abbott; Bio-Rad Laboratories, Inc.; Illumina, Inc.; Oxford Gene Technology; F. Hoffmann-La Roche Ltd.; PerkinElmer Inc.; Quest Diagnostics Incorporated; Applied Spectral Imaging; and Biological Industries USA, Inc.
b. Key factors that are driving the market growth include growth in utilization of genetic information in predictive models for disease traits coupled with increasing incidence of cancer and related growth in mortality per year.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





