EDC Systems Market: Increasing demand for cost-effective eClinical solutions
Electronic data capture (EDC) systems market is predicted to witness lucrative growth in coming years owing to the rising demand for computer-aided software solutions. The attached benefits with its use are the key contributing factors for the increasing demand for EDC systems. These systems improve the information management and analysis, accelerate the timeline for clinical trial approval, and make faster decisions pertaining to product approval procedures.
Medical device manufacturers and biopharmaceutical companies are adopting these solutions to save the cost and time spent on the clinical trial data management.
In addition, increasing competition amongst the majority of pharmaceutical companies is urging them to expand their R&D budgets, thus resulting in many pipeline components. Cumulatively, increasing number of patent expirations is also presumed to be responsible for an extensive indulgence of pharmaceutical corporations in drug development and discovery, which is increasing the competition. This increasing product pipeline demands a streamlining of workflow of all the processes, roles, and activities leading to efficient clinical data management.
eClinical solutions provide a wide range of options for the abovementioned workflow, which presents the market with lucrative growth opportunities. These benefits include recommendations on the medical interpretation of emergent findings, and elimination of data discrepancies. The other benefits of adopting EDC systems include advanced statistical analysis and improved compilation of technical & scientific data required for the regulatory agreement of clinical trials. These are presumed to fasten the overall approval process and, hence, expand the consumer base for EDC systems over the forecast period.
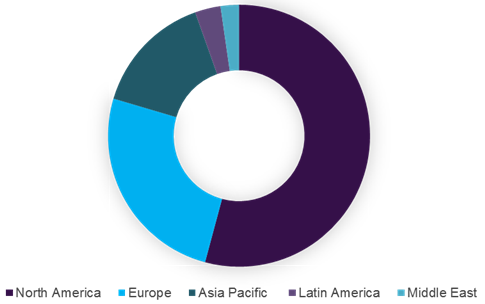
Global EDC systems market, by region, 2016
Moreover, rising trend of outsourcing for the healthcare IT services creates more avenues for contract research organizations to enter into eClinical solutions marketplace. The increasing outsourcing and externalization of clinical trials by a majority of the prominent pharmaceutical and biotechnological companies are anticipated to be responsible for driving the market at an unprecedented rate throughout the forecast period. The growing awareness pertaining to the advantages of EDC services over the paper-based clinical information management is a major factor contributing toward the growth of this market.
Growing adoption of cloud-based services by manufacturers is anticipated to boost the industry growth over the forecast period. In addition, these systems help into minimizing the errors occurred during manual data entry. However, the use of these solutions facilitates considerable saving of time and research-associated costs.
Installation of fully integrated services is an important factor anticipated to contribute to the rising demand for EDC software in the coming years. Furthermore, investment on eClinical services results in better outcomes with improved information management.
 In-depth report on global electronic data capture (EDC) systems market by Grand View Research:
In-depth report on global electronic data capture (EDC) systems market by Grand View Research:
http://www.grandviewresearch.com/industry-analysis/electronic-data-capture-edc-systems-market
To schedule a free market intelligence database demo, please complete the form below:
Service Guarantee
-
Insured Buying
This report has a service guarantee. We stand by our report quality.
-
Confidentiality
Your transaction & personal information is safe and secure.
-
Custom research service
Design an exclusive study to serve your research needs.
-
24/5 Research support
Get your queries resolved from an industry expert.