- Home
- »
- Medical Devices
- »
-
Melanoma Therapeutics Market Size & Share Report, 2030GVR Report cover
![Melanoma Therapeutics Market Size, Share & Trends Report]()
Melanoma Therapeutics Market (2025 - 2030) Size, Share & Trends Analysis Report By Product (Chemotherapy, Immunotherapy), By Drug Type (Branded Drugs, Generic Drugs), By Region, And Segment Forecasts
- Report ID: GVR-1-68038-535-9
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Melanoma Therapeutics Market Summary
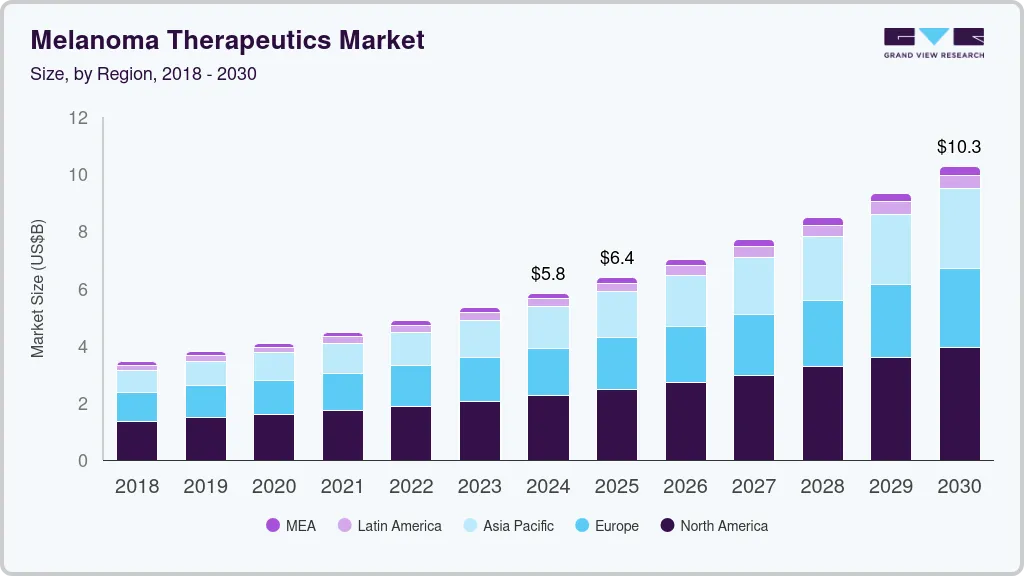
The global melanoma therapeutics market size was estimated at USD 5.83 billion in 2024 and is estimated to reach USD 10.26 billion by 2030, growing at a compound annual growth rate (CAGR) of 9.9% from 2025 to 2030. The increasing occurrence of melanoma and other skin cancers is projected to boost the need for anticancer agents.
Key Market Trends & Insights
- North America dominated the market and accounted for the largest share of 39.9% in 2022.
- By product, the immunotherapy segment accounted for the largest market share of 55.9% in 2022.
- By drug type, the branded drugs segment accounted for the largest market share of 91.3% in 2022.
Market Size & Forecast
- 2024 Market Size: USD 5.83 Billion
- 2030 Projected Market Size: USD 10.26 Billion
- CAGR (2025-2030): 9.9%
- North America: Largest market in 2022
As per the Skin Cancer Foundation, around 97,610 (about 58,120 in men and 39,490 in women) cases of invasive melanoma are expected to be diagnosed in the U.S. in 2023. Melanoma or skin cancer cases are rising rapidly due to the upsurge in skin ailments, allergies, climate change, and excessive exposure to ultraviolet rays.
As per the U.S. Environmental Protection Agency, the depletion of the ozone layer will increase the number of UV rays reaching the earth, which plays a major role in the development of melanoma, causing skin cancer. This will fuel the demand for melanoma therapeutics. The rise in novel technologies and treatments has led to the evolution of melanoma therapies. Early-stage detection treatments with minimum possible side effects are expected to drive the growth. A combination of therapies progresses the survival benefits, and increasing public awareness leads to the upgradation of strategies and increased demand. As per the American Cancer Society, the risk for melanoma increases with age and might affect people over 60 years of age.
The COVID-19 pandemic significantly impacted the market growth. The global focus on managing COVID-19 cases strained healthcare systems, diverting resources from conditions like melanoma and causing disruptions in routine screenings, leading to delayed diagnoses and treatment plan cancellations. Social distancing measures and lockdowns further hindered patient access to healthcare facilities, posing challenges in receiving timely care. These factors indirectly affected the market growth by imposing financial constraints on research, development, and investment in new melanoma treatments.
The demand is expected to increase due to rapid technological advancements, the necessity for innovative therapies, growing R&D efforts, increased funding from both public & private sectors, and the production of cost-effective drugs. For instance, the University of Copenhagen, along with its research team, has successfully treated invasive growth in skin cancer by using synthetic human skin. Improving healthcare infrastructure, growing population, quality services, need for cost-effective treatments, and the expiration of patents of key brands are expected to offer growth opportunities in the market.
Product Insights
The immunotherapy segment accounted for the largest market share of 55.9% in 2022. The market is experiencing growth propelled by advancements in healthcare infrastructure and diagnostics, particularly in immunotherapy. Immunotherapy is crucial in enhancing the individual's immune system to combat and eliminate cancer cells effectively. As per the American Cancer Society, immunotherapy helps improve the person’s immune system to help fight cancer cells more effectively and destroy them. It also helps treat melanoma conditions that can't be removed through surgery, spreading rapidly to all body parts and minimizing the risk of regeneration of cancer cells.
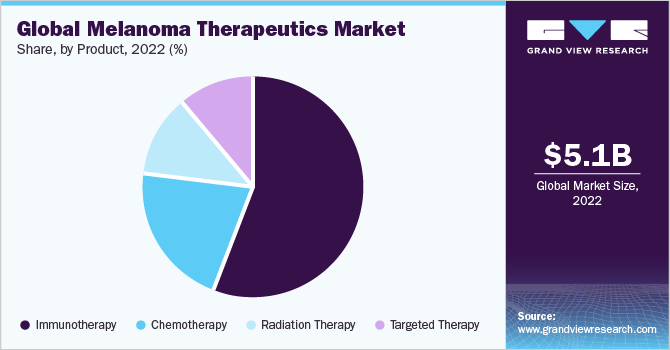
The data from the national clinical trial shows that about 89% of patients with desmoplastic melanoma responded positively after undergoing immunotherapy, reducing the risk of toxicity. The targeted therapy segment is expected to witness the fastest CAGR of 11.9% during the forecast period. The progress in genomics has resulted in the creation of targeted cancer therapies, which offer treatment options for patients who may not respond to traditional intensive cytotoxic chemotherapy. Product development strategies are increasingly gaining favor in the market. For instance, Pluvicto was introduced in March 2022 by Novartis AG, a global pharmaceutical company for individuals diagnosed with mCRPC; Pluvicto is the first Food and Drug Administration (FDA) approved, targeted radioligand therapy (RLT).
Drug Type Insights
The branded drugs segment accounted for the largest market share of 91.3% in 2022. The dominance of the market is attributed to the substantial patient base and increased utilization of biologics. The branded drugs segment is expected to experience growth due to rapid technological advancements. For instance, in February 2023, the FDA approved Bristol Myer’s license application for nivolumab (opdivo) to treat IIB or IIC stage melanoma.
The generic drug segment is expected to witness the fastest CAGR of 10.3% from 2023 to 2030. Generic drugs are cost-effective, easy to develop, and receive approval in less time than biologics, which is expected to fuel the segment growth over the forecast period. There is a strong emerging pipeline of melanoma drugs. Many of these drugs are under clinical trials. As per the National Cancer Institute (NCI), desmoplastic melanoma can be treated with a single immunotherapy drug Keytruda, avoiding the combination of therapies and side effects, according to the Phase II clinical trials.
Regional Insights
North America dominated the market and accounted for the largest share of 39.9% in 2022. According to the National Institutes of Health (NIH), melanoma cases are rapidly rising in the U.S. In addition, according to the American Cancer Society, it is estimated that around 3 million people are diagnosed with melanoma every year and this number will exceed all other cancer cases, which is about 1.9 million. In 2023, it is estimated that around 97,000 individuals might be diagnosed with invasive melanoma and 89000 with melanoma.
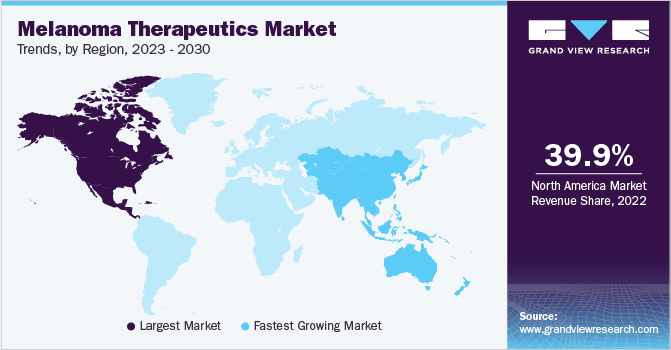
These factors are expected to drive the market growth. Asia Pacific is projected to grow at the fastest CAGR of 11.6% during the forecast period. A constant rise in the incidence of skin cancer combined with an increasing number of government initiatives is likely to fuel regional market growth. National Cancer Grid, an initiative by the government of India, has established a new center for digital oncology to improve cancer treatment using digital technologies.
Key Companies & Market Share Insights
The market is witnessing substantial growth with new product developments, partnerships, and technological development. For instance, in December 2022, Moderna, Inc., a biotechnological company, announced the personalized mRNA cancer vaccine, in combination with Merck’s Keytruda, which has proven to be useful in reducing the risk of death by 44% in stage 3 & 4 melanoma patients, with low risk of recurrence. Some of the key players in the global melanoma therapeutics market include:
-
Bristol-Myers Squibb Company
-
AstraZeneca
-
Novartis AG
-
Merck & Co. Inc.
-
Amgen Inc.
-
F. Hoffman-La Roche Ltd.
-
Abbott Laboratories
Melanoma Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 6.39 billion
Revenue forecast in 2030
USD 10.26 billion
Growth rate
CAGR of 9.9% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Report updated
September 2023
Quantitative units
Revenue in USD million/billion, and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, drug type, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Bristol-Myers Squibb Company; AstraZeneca; Novartis AG; Merck & Co, Inc.; Amgen Inc.; F. Hoffman-La Roche Ltd.; Abbott Laboratories
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
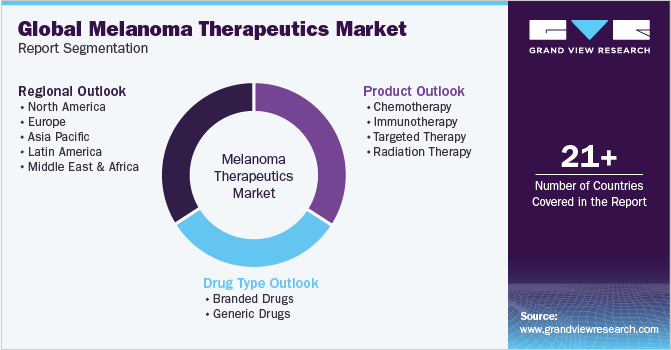
Global Melanoma Therapeutics Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global melanoma therapeutics market report on the basis of product, drug type, and region.
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Chemotherapy
-
Immunotherapy
-
Targeted Therapy
-
Radiation Therapy
-
-
Drug Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Branded Drugs
-
Generic Drugs
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Norway
-
Denmark
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East and Africa
-
Saudi Arabia
-
South Africa
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global melanoma therapeutics market size was estimated at USD 5.08 billion in 2022 and is expected to reach USD 5.55 billion in 2023.
b. The global melanoma therapeutics market is expected to grow at a compound annual growth rate of 10.3% from 2023 to 2030 to reach USD 10.98 billion by 2030.
b. North America dominated the melanoma therapeutics market with a share of 39.8% in 2022. This is attributable to well-established healthcare infrastructure, high healthcare spending, favorable reimbursement policies for therapies, and a high prevalence of melanoma.
b. Some key players operating in the melanoma therapeutics market include Bristol-Myers Squibb Company; Novartis AG; Amgen, Inc.; F. Hoffman-La Roche Ltd; Merck & Co., Inc.; Array Biopharma; AB Science; and Astrazeneca Plc.
b. Key factors that are driving the market growth include rising incidence of melanoma, increasing government initiatives, and the introduction of novel melanoma therapies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










