- Home
- »
- Healthcare IT
- »
-
Clinical Trials Market Size And Share, Industry Report, 2033GVR Report cover
![Clinical Trials Market Size, Share & Trends Report]()
Clinical Trials Market (2026 - 2033) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III, Phase IV), By Study Design, By Indication, Indication By Study Design, By Service, By Sponsor, By Region, And Segment Forecasts
- Report ID: GVR-1-68038-975-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Clinical Trials Market Summary
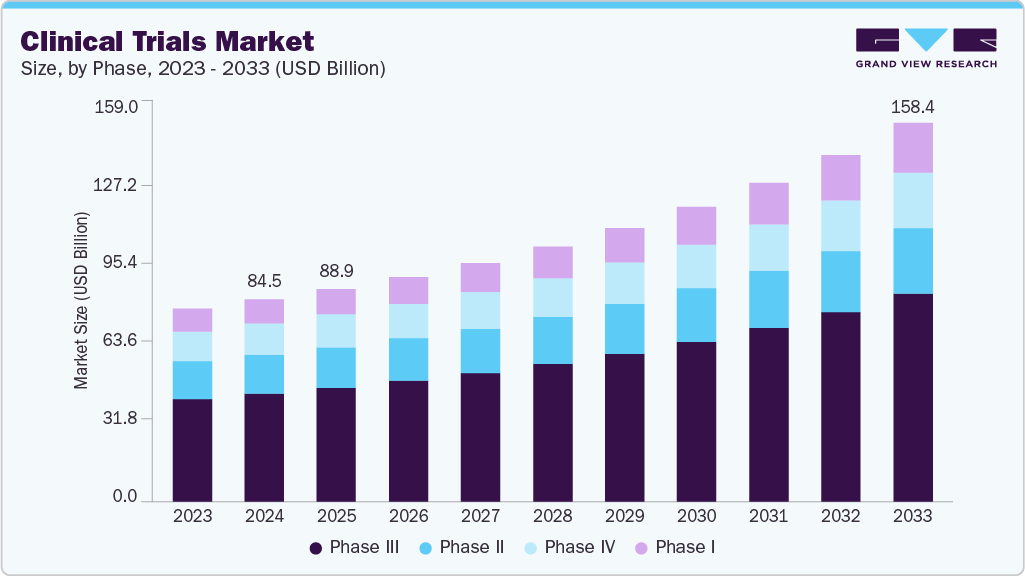
The global clinical trials market size was estimated at USD 89.0 billion in 2025 and is projected to reach USD 158.4 billion by 2033, growing at a CAGR of 7.7% from 2026 to 2033. The market growth is driven by the rising prevalence of chronic and rare diseases, advancements in precision medicine and biotechnology, the expansion of decentralized and virtual trials, and growing R&D investments by pharmaceutical and biotech companies.
Key Market Trends & Insights
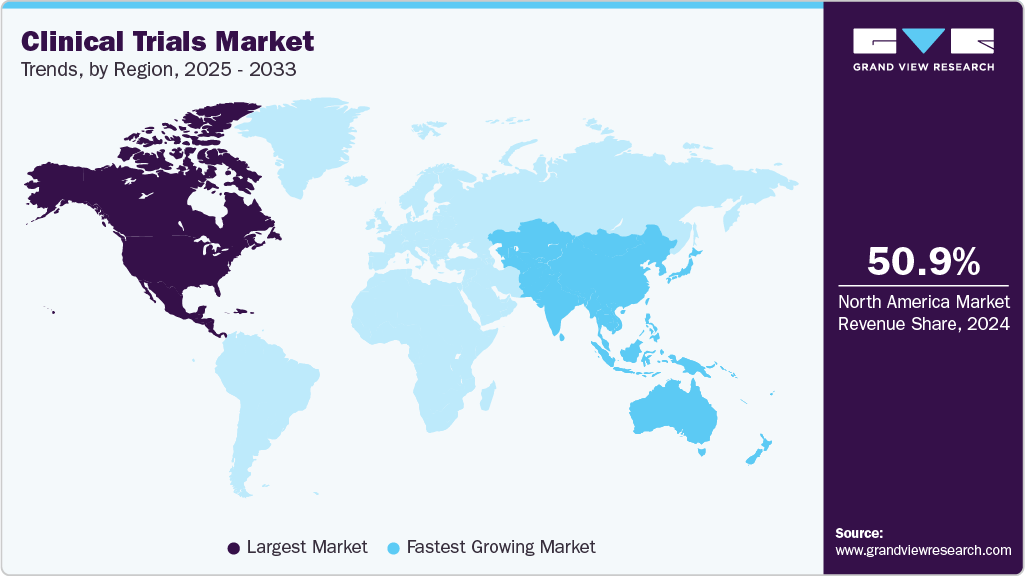
- The North America clinical trials market held the largest global share of 50.7% in 2025.
- The U.S. clinical trials industry is expected to grow lucratively from 2026 to 2033.
- By phase, the phase III segment held the largest market share of 53.6% in 2025.
- By study design, the interventional trials segment held the largest market share in 2025.
- By indication, the oncology segment held the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 89.0 Billion
- 2033 Projected Market Size: USD 158.4 Billion
- CAGR (2025-2033): 7.7%
- North America: Largest market in 2025
- Asia-Pacific: Fastest growing market
The market growth is attributed to the growing trend of outsourcing services, the rising prevalence of chronic conditions such as cancer, diabetes, and neurological disorders, and the increasing demand for innovative therapies.In addition, a growing number of pharmaceutical and medical device companies, advancements in digital health, personalized medicine, and global partnerships are expected to drive the growth of the clinical trials industry over the forecast period. In addition, supportive government initiatives, advanced regulatory approvals, and increased funding from pharmaceutical and biotech companies further contribute to the global market. Furthermore, global collaborations, increased patient awareness, and improved access to trials are expected to drive recruitment, data quality, and overall success rates across trial populations and regions.
Similarly, significant changes in the conduct of ongoing or upcoming clinical trials are among the factors contributing to market growth. Regulatory agencies, including the U.S. FDA, the European Medicines Agency (EMA), the National Institutes of Health (NIH), and China’s National Medical Products Administration, among several others, issued various guidelines for conducting trials to support the implementation of decentralized clinical trials and virtual services. The current scenario for research and development activities across the globe and the need for several new treatment options have also led to the adoption of fast-track clinical trials. Thus, the aforementioned factors are expected to open new opportunities for growth in the clinical trial market.
Furthermore, the utilization of CRO services enables manufacturers and sponsors to focus their resources on strengthening core production capabilities and optimizing in-house operational processes. The availability of a vast array of services, from drug discovery to post-marketing surveillance, has further simplified processes for mid-size & small-scale pharmaceutical and biotechnological organizations by providing them with the option to outsource research and development activities to reduce infrastructure investment. For instance, in May 2025, IQVIA entered into a strategic partnership with the Sarah Cannon Research Institute, known as SCRI Development Innovations. This collaboration is focused on advancing oncology trials for biopharma partners around the globe. By leveraging the strengths of both IQVIA and SCRI Development Innovations, the partnership aims to improve clinical trial processes, overcome operational challenges, enhance data delivery, and expedite patient access to innovative therapies.
Opportunity Analysis
Sponsors of clinical trials are gradually shifting their focus toward novel locations, including India and China, which have large populations of naïve patients for treatments and fast recruitment processes that lead to shorter trial times, while remaining economically viable. Besides, decentralization of clinical trials through the use of telemedicine, mobile technology, and other remote means of monitoring is making clinical trials more convenient for patients who are suffering from chronic diseases and genetic disorders, who used to be reluctant to attend frequent site visits. Moreover, there is an increased interest among sponsors in conducting biomarker and genotype-specific trials in the fields of immunology and oncology, and this scenario opens opportunities for innovative services of CROs.
The incorporation of AI in the development of protocols and evaluating the feasibility of conducting studies allows sponsors to recognize high performers within their sites and target potential patients more accurately, which will decrease the likelihood of screening failures. Moreover, mid-sized biotech companies will increase outsourcing due to a lack of infrastructure for conducting clinical research. As a result, there will be an increased need for full-service and functional service providers across phases I through IV of clinical trials.
In addition, the number of rare disease drugs in clinical pipelines is increasing, and many have been designated as orphan drugs. This will necessitate conducting multinational clinical trials, and sponsors will need regulatory experts who can navigate the drug approval process efficiently and endpoint measurement specialists to conduct the trials successfully.
Moreover, growing cooperation among pharmaceutical players, universities, and digital health companies is helping build a data ecosystem and accelerating innovation across fields such as cell and gene therapy. Thus, the above trends are setting the stage for an extremely specialized, technology-focused field of clinical trials.
Impact of U.S. Tariffs on Clinical Trials Market
The U.S. tariffs on pharmaceutical imports are exerting cost pressure on the global clinical trial industry, with the escalation in duties on investigational products, comparators, and lab consumables resulting in rising costs for trials and operations. Additionally, tariffs on APIs and other materials used in clinical development from Asia and Europe are driving up sourcing costs, which are subsequently passed on to sponsors and CROs conducting clinical trials. Moreover, U.S.-based clinical trials are witnessing inflated site-level costs due to the inclusion of higher medical consumable costs in trial budgets, thereby impacting sponsor budgets. In addition, supply chain disruptions caused by tariffs and onshoring efforts have led to a shortage of clinical materials required for trials. Additionally, firms are increasingly adopting new global trial strategies by moving their clinical trials to cheaper regions such as the Asia Pacific and Eastern Europe.
Also, the demand for manufacturing and packaging to be localized in the United States is adding costs due to increased regulation and compliance requirements. In addition, the impact of tariffs is forcing companies to seek strategic alliances with local suppliers and CROs, thus changing the dynamics of outsourcing and vendor selection. Thus, the overall impact of tariffs is prompting more focus on cost-cutting measures, digital clinical trials, and diversified supply chains.
Technological Advancements
The advent of new technology in clinical trials is being fueled by the complexity of data generated during drug development and the need for faster timelines, in which artificial intelligence is making protocols and patient identification more efficient, and decentralized technologies are increasing patient access and reducing dependence on sites. Besides, wearable technologies are facilitating constant and real-time data collection, which makes the data collected more accurate in long-term trials. In addition, cloud computing platforms are making data integration easier through global clinical trial networks, enabling stakeholders to use and analyze data more efficiently.

Besides, these technologies are collectively reducing operational bottlenecks and improving trial execution quality. Further, sponsors are increasingly investing in digital infrastructure to support hybrid and virtual trial models. Moreover, integration of multiple digital tools enables end-to-end visibility across trial phases. In addition, regulatory bodies are gradually adapting to these advancements by issuing guidance on digital endpoints and remote monitoring. Furthermore, these developments are supporting more patient-centric trial designs, thereby improving engagement and compliance. Also, data-driven decision-making is becoming central to trial success as analytics capabilities expand. Further, technology adoption is also enabling scalability in global multi-site trials. Collectively, the technological landscape is evolving toward a more efficient, secure, and patient-focused clinical trial ecosystem.
Pricing Model Analysis
The global clinical trials market uses various pricing models, including fee-for-service, full-time equivalent, and milestone-based pricing, that fit the specific needs of clinical trials and the strategies adopted by sponsors. Fee-for-service models are generally employed for defined clinical services, as budgets are more predictable in large, well-defined protocols and late-phase trials. Also, under this model, there is less scientific flexibility, as changes to the protocol and scope expansion require additional payments, which pose a high risk for sponsors. On the other hand, full-time equivalent models are resource-based, in which personnel are used for a period of time. Additionally, this model provides flexibility for protocol changes and design adjustments, making it ideal for early-phase clinical trials. Nevertheless, budget predictability under this arrangement is moderate.
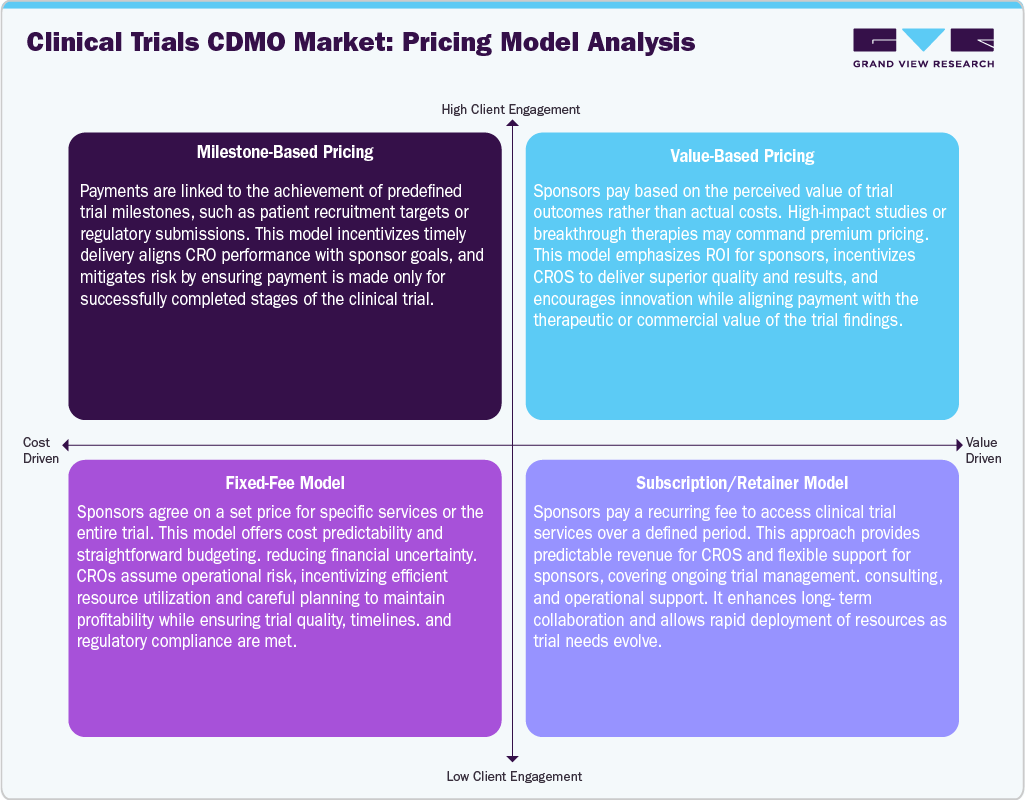
The milestone pricing aligns payments with predefined milestones, such as enrollment goals and regulatory filings, enabling phased budgeting. This pricing approach incorporates joint responsibility for the sponsor and the service provider, ensuring that the total financial risk is minimized through payments based on performance. The use of milestone-based pricing provides some flexibility between milestones, though significant alterations may require changes to the terms of engagement. Thus, all of the above pricing models are often combined in hybrid pricing models.
Market Characteristics & Characteristics
The market growth stage is high, and the pace of the market growth is accelerating. The clinical trials market is characterized by evolving technologies and therapeutics, regulatory considerations, the growing need for advanced therapeutics, the increasing demand for personalized medicine, and the outsourcing of development processes to leverage cost advantages and specialized capabilities.
Advancements in drug development, the adoption of advanced therapies such as cell and gene therapy, and the rising demand for personalized medicine are likely to influence market growth. The clinical trials industry has seen a surge in innovative technologies such as AI, big data analytics, and remote monitoring. Companies that invest heavily in advanced technologies and effectively utilize them often gain a competitive edge, driving market growth.
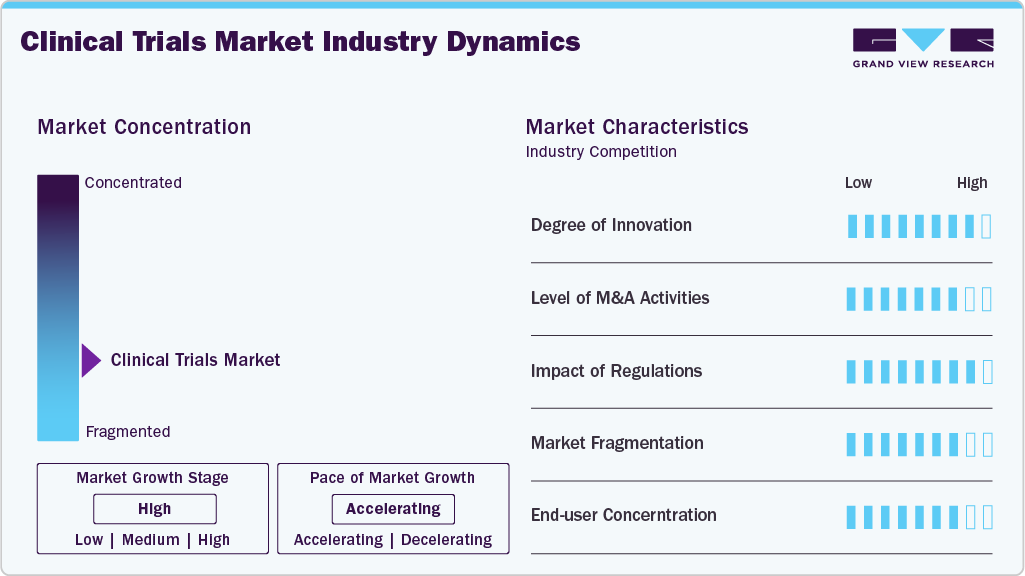
Stringent quality protocols and regulatory norms by several nations to ensure patient safety and data integrity, which highly impact operational capabilities in the clinical trials industry. Market participants with robust compliance measures and a track record of fulfilling regulatory standards gain credibility and preference from biopharmaceutical sponsors. Compliance with these regulations demands substantial resources, leading to barriers for smaller or newer firms.
Mergers and acquisitions in the clinical trials market are increasing and are expected to continue growing during the analysis timeframe. Several companies are acquiring development-stage companies to expand their service portfolios and serve large patient populations. Moreover, these firms are integrating advanced facilities and forming strategic alliances to achieve synergies in capabilities and resources, enhancing their competitiveness.
The market comprises a large number of biopharmaceutical and medical device manufacturers, and CROS specializes in drug development, leading to a fragmented market.
Pharmaceutical, biotech, medical device firms, and academic research centers are major consumers of clinical trial services. Their preferences, bargaining power, and concentration can significantly influence the market. Concentration among these end users might favor specific service providers or trial conductors.
Phase Insights
The phase III segment led the market, accounting for a revenue share of 53.6% in 2025. Phase III trials are the most expensive and involve a large number of subjects. The median cost for a single-phase III trial is over USD 19.0 million. In addition, phase III requires a higher number of patients and often a longer treatment period. According to a clinical trial logistics survey by Nice Insights, 35.0% of phase III clinical trials are outsourced, and the number is likely to increase owing to the growing number of investigational drugs advancing to the next phase.
The Phase I segment is expected to grow significantly over the analysis period. These trials serve as the cornerstone of drug development and medical device innovation, focusing on safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) in small groups of healthy volunteers or patients. Moreover, progress in genomics and biomarker research has led to more advanced and sophisticated Phase I trials. Furthermore, the shift towards digitalization, automation, and artificial intelligence is improving clinical trial operations, reducing costs, and speeding up drug development timelines. Innovations such as AI, adaptive clinical trial designs, and decentralized methods are making Phase I trials safer, faster, and more efficient, ultimately improving patient outcomes and accelerating the development of new therapies.
Study Design Insights
The interventional studies segment dominated the clinical trials market in 2025. It is one of the most widely used methods in clinical trials. Interventional studies accounted for over 75.0% of the total registered studies in 2022, of which most were drug or biologics, followed by clinical procedures, behavioral, and device interventional studies. These studies account for 94.0% of the total studies that posted results, with drug or biologics contributing the most, followed by behavioral, devices, and Clinical procedure intervention studies.
The observational segment is anticipated to register a notable CAGR during the forecast period. Observational studies focus on analyzing health outcomes in groups of patients without changing the treatment they receive. These studies are essential for outlining robust protocols that specify the objectives, methods, and procedures for conducting a clinical trial. It serves as a roadmap for researchers to gain insights into the natural progression of diseases, identify potential risk factors, and assess the effectiveness of treatments in real-world data (RWD), patient-reported outcomes, and epidemiological endpoints contexts. Besides, these studies address ethical considerations, regulatory requirements, and potential challenges that may arise during the trial. In addition, as new drugs emerge from the development pipeline, they are expected to drive the growth of the observational studies segment.
Indication Insights
The oncology segment accounted for the largest revenue share of the clinical trials industry in 2025. According to the U.S. FDA and other sources, the pharmaceutical industry currently spends more than USD 38.0 billion on the preclinical and clinical development of oncology therapy products. Thus, growing investment in this field is accelerating the research and development activities in this segment, further contributing to market growth.
The autoimmune/inflammation segment is also anticipated to witness lucrative growth over the forecast period. In autoimmune/inflammation, the immune system erroneously targets healthy cells & tissues; these diseases affect approximately one in 10 individuals. Among autoimmune conditions like Rheumatoid Arthritis (RA), Multiple Sclerosis (MS), lupus, and type 1 diabetes, a shared aspect is the immune system’s malfunction. The misdirected immune response in autoimmune disease leads to persistent inflammation and harm to various organs & systems. Hence, clinical trials represent a key milestone in the development of new devices and drugs for autoimmune diseases. Moreover, the ongoing innovative approaches for autoimmune disease drug development are expected to drive the segment growth.
Service Insights
The laboratory services segment garnered significant market share in 2025. The growing complexity of clinical trials, coupled with the adoption of biomarker-based research, has created a need for high-quality diagnostic and analytical laboratories at all stages. Additionally, the focus on personalized medicines and treatments requires testing and analysis of samples, making standardized, high-throughput labs indispensable. Moreover, the strong emphasis on regulatory requirements for accurate data and its validation leads sponsors to rely heavily on the provision of laboratory services to avoid non-compliance. Further, the expansion of multi-regional trials and the growth of genomics and molecular testing are creating additional demand for laboratory services.
Patient recruitment represents the fastest-growing segment, owing to an increasingly difficult process of identifying qualified participants for clinical trials within a certain period of time, especially in the case of studies related to rare diseases and oncology, due to stringent inclusion criteria. The growing popularity of the use of advanced technologies for recruiting subjects for clinical trials, such as social media campaigns, data mining from electronic health records, and artificial intelligence-driven recruitment, is boosting recruitment efficiency and scope. Decentralized trials also allow reaching a larger number of potential participants.
Sponsor Insights
The pharmaceutical & biopharmaceutical companies accounted for the largest revenue share of the clinical trials industry in 2025. This can be attributed to the greater interest of the pharmaceutical industry in the research field. In addition, the number of clinical trials funded by pharmaceutical & biopharmaceutical companies has increased. The pharmaceutical industry plays a vital role in financing the research for the development of new drugs.
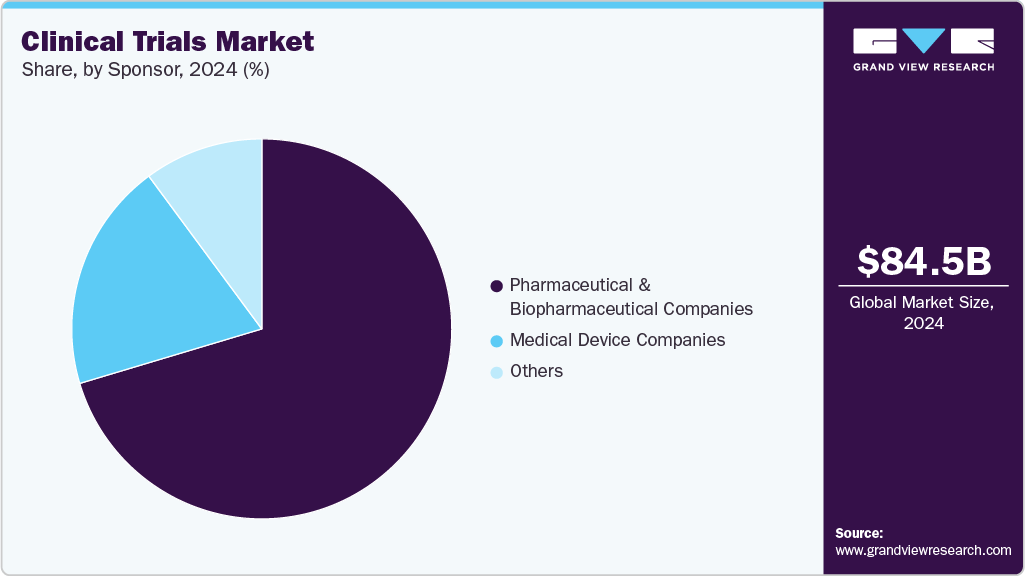
The medical device companies segment is anticipated to grow at a considerable CAGR over the forecast period. The segment growth is driven by the increasing demand for advanced diagnostic and therapeutic devices, growing investment in R&D activities, and continuous technological innovations aimed at enhancing device accuracy, safety, and patient outcomes. In addition, the rising prevalence of chronic diseases, favorable regulatory reforms, and the growing adoption of digital health technologies such as remote monitoring and AI-based diagnostics are further supporting market expansion.
Regional Insights
North America accounted for 50.7% of the global clinical trials industry in 2025 and is expected to continue its dominance over the forecast period. This can be attributed to increased R&D investments and the adoption of new technologies in clinical trials in this region. For instance, the implementation of virtual services in various stages of clinical trials by market players, such as IQVIA and PRA Health Sciences, is anticipated to further propel the North America’s virtual clinical trials market growth.
U.S. Clinical Trials Market Trends
The U.S. clinical trials industry’s growth is driven by the country’s favorable government support in the U.S. for clinical trials. For instance, in February 2024, the National Cancer Institute (NCI) launched the Virtual Clinical Trials Office, a centralized remote staffing initiative to address clinical trial workforce shortages, enhance patient enrollment, and support cancer research across NCI-designated centers and community programs nationwide. This initiative is expected to strengthen trial efficiency, expand access in underserved regions, and accelerate progress in cancer research across the U.S.
The clinical trials market in Canada is advancing, fueled by strong government funding and innovation-focused initiatives. For instance, in August 2024, the Government of Canada invested over USD 43 million to support 14 new clinical trials addressing major health threats. This strengthened national R&D capabilities, bolstered the life sciences sector, and enhanced preparedness for future health emergencies.
Europe Clinical Trials Market Trends
The Europe clinical trials industry is expanding steadily, driven by strong regulatory support, rising investments in rare disease research, and the adoption of decentralized and digital trial models. For instance, in July 2025, European clinical trial organizations urged the urgent implementation of the Life Science Strategy to address regulatory fragmentation, recruitment challenges, and administrative burdens that threaten Europe’s clinical research ecosystem and competitiveness. Such initiatives are crucial to restoring efficiency, fostering innovation, and ensuring that Europe remains a competitive and attractive hub for global clinical research.
The clinical trials market in the UK is expected to grow significantly over the forecast period, with the highest CAGR. The country’s growth is influenced by regulatory reforms and streamlined approval processes, as well as the adoption of artificial intelligence and digital platforms. According to the UK report from October 2025, clinical trials in the UK strengthened with faster approval timelines driven by MHRA reforms and AI integration, reducing approval times from 91 to 41 days. These advancements enhance patient access, accelerate trial setup, and boost research efficiency. The UK positions itself as a global leader in modernized, AI-enabled clinical research, supported by the 10-Year Health Plan.
Germany's clinical trials market held the largest share in 2025. Strong R&D infrastructure, supportive regulatory frameworks, and high investment in neurodegenerative research fuel the growth. For instance, in August 2025, Eisai and Biogen launched LEQEMBI, the first anti-amyloid Alzheimer’s therapy in Germany, following EU approval. Such advancements emphasize Germany’s leadership in clinical innovation and early-stage therapeutic development.
Asia Pacific Clinical Trials Market Trends
Asia Pacific is the fastest-growing market, as many developed economies are outsourcing clinical trials to countries such as India, China, and South Korea. The evolving business model of outsourcing and R&D among key global companies is expected to increase demand for clinical trial services in the region, owing to the cost-efficiency offered by CROs in countries such as India and China. Furthermore, large & diverse patient pools, clinical trial recruitment, established clinical infrastructure, and the availability of skilled medical practitioners are supporting market growth.
In addition, the presence of multinational pharmaceutical/biopharmaceutical companies & CROs in the region, along with the higher cost-efficiency of conducting clinical trials compared to the U.S. and European countries, is creating growth opportunities in the market. Governments are undertaking efforts to drive the market booth. In addition, APAC hosts a growing pool of scientific & development expertise, enabling rapid adoption of advanced technologies & quality clinical outsourcing services.
The China clinical trials market dominated Asia Pacific, accounting for the largest revenue share in 2025. The market witnessed rapid expansion driven by regulatory reforms, government funding, and digital innovation. The 2025 Outsourcing in Clinical Trials China and Clinical Trial Supply China events in Suzhou highlighted the nation’s growing global influence. Initiatives like the 2015 policy and major deals, such as AstraZeneca’s USD 5.2 billion collaboration with CSPC, fueled innovation, global partnerships, and growth of the biotech ecosystem.
The clinical trials market in Australia is growing, driven by strong government support, advanced research infrastructure, and a focus on innovative studies. For instance, in June 2025, studies focused trials reflected Australia’s dominance in neurological research and drug repurposing. The country’s streamlined regulatory framework, skilled clinical workforce, and initiatives like MS Trial Screen enhanced patient recruitment and positioned Australia as a preferred destination for complex, high-impact clinical research.
Latin America Clinical Trials Market Trends
Latin America is anticipated to showcase considerable growth over the forecast period, owing to the growing expansion of CRO with the regional pharmaceutical and biotechnological companies, a huge patient pool, and a clinical trial success rate.
In Latin America, 80% of the population lives in metropolitan cities. The three megacities of São Paulo, Buenos Aires, and Mexico City have large patient pools. Although the regulatory timeline in Latin America is a drawback, the time saved by patient recruitment compensates for it. In addition to patient recruitment, patient retention is high due to the healthy patient-physician relationship. Low costs and study coordinators with GCP-compliant knowledge contribute to the country's clinical trial success rate. Mexico and Brazil are expected to witness lucrative growth in the CRO market due to the cost advantage over developed economies, such as the U.S. and Germany.
Furthermore, clinical studies conducted in Latin America adhere to international standards, owing to the growing interest of international players in conducting clinical trials in this region. A major contributor is the investigators' access to thousands of potential treatment candidates. This factor helps in patient recruitment and retention, thereby reducing trial costs.
The clinical trials market in Brazil is gaining momentum, supported by a large patient base, strong investigator expertise, and rising demand for biopharma innovations. For instance, in April 2025, Valneva’s single-dose chikungunya vaccine, IXCHIQ, received ANVISA approval in Brazil, marking the first authorization in an endemic country and enabling Phase 4 clinical trials, supported by CEPI, the EU, and Instituto Butantan. This strengthened the country’s role in global vaccine development and expanded access to critical chikungunya prevention in endemic regions.
Middle East & Africa Clinical Trials Market Trends
The Middle East and Africa clinical trials industry experienced significant growth, driven by rising investments in oncology research, favorable regulatory reforms, and increasing collaborations between local and global pharma companies. Several companies in the region are innovating in patient diversity, cost advantage, infrastructure, and world-class medical facilities. The region has much to offer, with an untapped clinical-trials market and expanding pharmaceutical, biopharmaceutical, and medical device companies. In addition, the number of CROs in the region has been increasing in recent years. The region’s attractive incentive policies have pulled big pharmaceutical companies, such as Pfizer, AstraZeneca, and Amgen. CROs, such as Ergomed and ClinTec International, were the first to establish CRO offices in the UAE.
South Africa’s clinical trials market witnessed robust growth, driven by strong government support, advanced research infrastructure, and a rising prevalence of chronic and infectious diseases. Some of the initiatives, such as IAVI’s Phase 1 HIV vaccine trial (GRAdHIVNE1) in August 2025, were conducted at the Desmond Tutu Health Foundation in Cape Town and the Africa Health Research Institute in Durban. Collaboration with global partners such as ReiThera and the Ragon Institute, and funding from the Gates Foundation, enhanced trial quality. Local expertise and state-of-the-art facilities strengthened South Africa’s position as a key hub for clinical research in sub-Saharan Africa.
The UAE clinical trials market expanded significantly, due to government-backed funding, strategic partnerships, and investments in advanced healthcare infrastructure. For instance, in February 2025, Cardiogeni PLC signed a USD 26.72M MoU with the Private Office of Sheikh Al Qassimi to fund heart failure clinical trials via a joint venture. Strong regulatory support, international collaborations, and rising demand for innovative therapies enhanced the UAE’s position as a regional clinical research hub.
Key Clinical Trials Company Insights
The key players operating across the market are adopting strategic initiatives such as service launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in February 2025, Summit Therapeutics Inc. announced a collaboration with Pfizer Inc. to conduct a clinical trial evaluating ivonescimab, an innovative investigational PD-1/VEGF bispecific antibody. This study will explore the use of ivonescimab in combination with various antibody drug conjugates (ADCs) developed by Pfizer, targeting multiple solid tumor indications.
Key Clinical Trials Companies:
The following key companies have been profiled for this study on the clinical trials market.
- IQVIA Inc.
- Parexel International Corporation
- Thermo Fisher Scientific Inc.
- ICON Plc.
- LabCorp
- Syneos Health
- Novotech
- Avance Clinical
- George Clinical
- Veritus Research
- Eli Lilly and Company
- F. Hoffmann-La Roche Ltd.
- Novo Nordisk A/S
- Pfizer, Inc.
- Actinogen Medical
- Telix Pharmaceuticals
- PYC Therapeutics
- Neuren Pharmaceuticals
- AbbVie
- Merck Sharp & Dohme LLC
Recent Developments
-
In October 2025, Precision BioSciences initiated its first U.S. clinical trial site at Massachusetts General Hospital for the Phase 1 ELIMINATE-B study of PBGENE-HBV. The trial evaluates in vivo gene-editing therapies in patients with chronic hepatitis B. This expansion strengthens clinical development capabilities and accelerates patient access to innovative gene-editing treatments.
-
In October 2025, SeaBeLife secured USD 2.16 million in pre-Series A funding led by iXLife and new investors, supporting the development of drug candidates for dry AMD and severe acute hepatitis. The first clinical trial is set to begin in 2026.
-
In May 2025, ImmunityBio partnered with Saudi Arabia’s Ministry of Investment, KFSHRC, and KAIMRC to launch the FDA-approved Cancer BioShield platform, introducing immune-restorative therapies targeting NK and T cells. The initiative aimed to enhance cancer care and foster regional collaboration.
Clinical Trials Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 94.0 billion
Revenue forecast in 2033
USD 158.4 billion
Growth rate
CAGR of 7.7% from 2026 to 2033
Actual data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends, clinical trials outlook, volume analysis
Segments covered
Phase, study design, indication, indication by study design, service, sponsor, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country Scope
U.S.; Canada; Mexico; UK; Germany; Italy; France; Spain; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Columbia; Saudi Arabia; UAE; Kuwait; Qatar; Oman
Key companies profiled
IQVIA Inc.; Parexel International Corporation; Thermo Fisher Scientific Inc.; ICON Plc.; LabCorp; Syneos Health; Novotech; Avance Clinical; George Clinical; Veritus Research; Eli Lilly and Company; F. Hoffmann-La Roche Ltd.; Novo Nordisk A/S; Pfizer, Inc.; Actinogen Medical; Telix Pharmaceuticals; PYC Therapeutics; Merck Sharp & Dohme LLC; Neuren Pharmaceuticals; AbbVie
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to the country & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Clinical Trials Market Report Segmentation
This report forecasts revenue growth at the global, regional & country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global clinical trials market report based on phase, study design, indication, indication by study design, service, sponsor, and region:
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Phase IV
-
-
Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Indication Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Rheumatoid Arthritis
-
Multiple Sclerosis
-
Osteoarthritis
-
Irritable Bowel Syndrome (IBS)
-
Others
-
-
Pain Management
-
Chronic Pain
-
Acute Pain
-
-
Oncology
-
Blood Cancer
-
Solid Tumors
-
Others
-
-
CNS Conditions
-
Epilepsy
-
Parkinson's Disease (PD)
-
Huntington's Disease
-
Stroke
-
Traumatic Brain Injury (TBI)
-
Amyotrophic Lateral Sclerosis (ALS)
-
Muscle Regeneration
-
Others
-
-
Diabetes
-
Obesity
-
Cardiovascular Diseases
-
Others
-
-
Indication by Study Design Outlook (Revenue, USD Million, 2021 - 2033)
-
Autoimmune/Inflammation
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Pain Management
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Oncology
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
CNS Conditions
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Diabetes
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Obesity
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Cardiovascular Diseases
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
Others
-
Interventional Trials
-
Observational Trials
-
Expanded Access Trials
-
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Protocol Designing
-
Site Identification
-
Patient Recruitment
-
Laboratory Services
-
Analytical Testing Services
-
Clinical Trial Data Management Services
-
Others
-
-
Sponsor Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical & Biopharmaceutical Companies
-
Medical Device Companies
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
India
-
China
-
Japan
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
Columbia
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global clinical trials market size was estimated at USD 89.0 billion in 2025 and is expected to reach USD 94.0 billion in 2026.
b. The global clinical trials market is expected to grow at a compound annual growth rate of 7.73% from 2026 to 2033 to reach USD 158.4 billion by 2033.
b. North America dominated the market for clinical trials and accounted for the largest revenue share of 50.66% in 2025. The dominance is due to the strong presence of leading pharmaceutical and biotechnology companies, robust regulatory frameworks, and well-established clinical research infrastructure.
b. Some key players operating in the clinical trials market include IQVIA Inc., Parexel International Corporation, Thermo Fisher Scientific Inc., ICON Plc., LabCorp, Syneos Health, Novotech, Avance Clinical, George Clinical, Veritus Research, Eli Lilly and Company, F. Hoffmann-La Roche Ltd., Novo Nordisk A/S, Pfizer, Inc., Actinogen Medical, Telix Pharmaceuticals, PYC Therapeutics, Neuren Pharmaceuticals, AbbVie, Merck Sharp & Dohme LLC among others
b. Key factors that are driving the clinical trials market growth includes rising prevalence of chronic and rare diseases, advancements in precision medicine and biotechnology, the expansion of decentralized and virtual trials, and growing R&D investments by pharmaceutical and biotech companies
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










