- Home
- »
- Pharmaceuticals
- »
-
Vaccine Market Size, Share & Trends Analysis Report, 2030GVR Report cover
![Vaccine Market Size, Share & Trends Report]()
Vaccine Market Size, Share & Trends Analysis Report By Type (Subunit, mRNA), By Route of Administration (Oral, Nasal), By Disease Indication (HPV, MMR), By Age Group, By Distribution Channel, And Segment Forecasts, 2023 - 2030
- Report ID: 978-1-68038-990-6
- Number of Pages: 180
- Format: Electronic (PDF)
- Historical Range: 2018 - 2021
- Industry: Healthcare
Report Overview
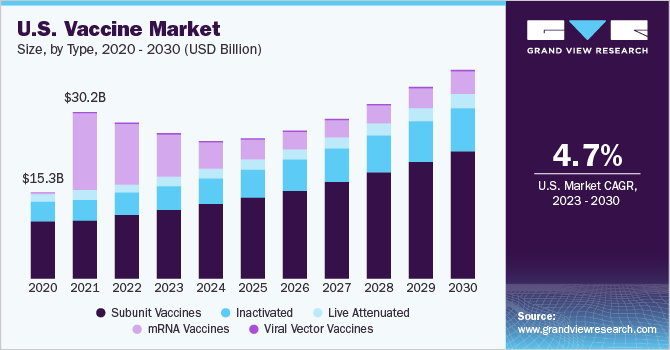
The global vaccine market size was estimated at USD 124.2 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 1.4% from 2023 to 2030. Excluding the impact of COVID-19, the vaccine market is expected to grow at a CAGR of 8.6% from 2023 to 2030. Over time, the range of vaccines available worldwide has been growing steadily, with the creation of new vaccines aimed at diseases that typically impact lower-income countries. In the U.S., the COVID vaccine market is anticipated to go commercial by the second half of 2023 as the stock purchased by the federal government will be exhausted.
The opening up of commercial markets will lead to price increases. For instance, in March 2023, Moderna announced that its COVID vaccine market price will increase to around USD 110 to USD 130 per dose. The privatization of the market will also lead to intensified competition.
The COVID-19 pandemic has highlighted that simply relying on significant public investment, efficient processes, new technologies, collective financing, and procurement strategies is not enough to achieve the best public outcomes. Despite the global delivery of roughly 15 billion doses through different mechanisms as of October 2022, only 12% of this total came from COVAX, a worldwide global alliance for equitable access to Covid vaccines. This indicates that achieving equitable access for all populations and preventing future pandemics necessitates more than just financing and procurement initiatives.
Regulatory agencies and regional networks played a key role in enhancing regulatory capacity and promoting coordinated regulatory efforts across countries. This resulted in wider accessibility of newly developed vaccines across different regions. WHO has been providing regulatory assistance to countries procuring through United Nations agencies by means of its prequalification program.
Additionally, WHO has assisted countries in developing efficient, stable, and integrated regulatory systems with predictable timelines and streamlined processes. As a result of these efforts, WHO has classified the national regulatory authority in 35 vaccine-producing countries as having sufficient maturity levels to oversee vaccine development, manufacturing, and release.
Type Insights
The mRNA segment dominated the vaccine market with a share of 39.12% in 2022. Pfizer/BioNTech and Moderna are the two companies that have launched mRNA COVID vaccines in the market. The advantages of these vaccines over traditional vaccines such as their ability to adjust antigen design and even integrate sequences from multiple variants to tackle new mutations in the virus genome are a major factor for the segments’ dominance. The success of mRNA-based COVID-19 vaccines has given impetus to the development of mRNA platforms for the prevention of various infectious diseases such as flu and RSV. For instance, in February 2023, Moderna announced interim results from its phase 3 trial for mRNA-1010, which is a seasonal flu shot under development. The vaccines showed superior results against influenza A strains but fell short against certain influenza B strains.
The subunit vaccines segment is estimated to have the fastest CAGR during the forecast period, owing to the increasing incidence of infectious diseases, the rising demand for safe & effective vaccines, and the growing focus on preventive healthcare. The growing need for more effective vaccines against diseases such as cancer, autoimmune disorders, and allergies is also driving the demand for subunit vaccines. For instance, in November 2022, Curevo Vaccine (Curevo), a clinical-stage biotechnology company that aims to develop safe and effective vaccines to reduce the burden of infectious diseases, announced the completion of a Series A1 funding round worth $26 million. The proceeds from the financing will be used to support the development of CRV-101, a clinical-stage adjuvanted sub-unit vaccine that is being investigated for its potential in preventing shingles in older adults.
Route of Administration Insights
Parenteral administration is highly preferred for administering vaccines and hence the segment dominated the vaccine market with a share of 97.09% in 2022. Parenteral administration provides faster absorption, is more effective, and reduces the risk of contamination and vaccine degradation. Amongst the currently marketed vaccines, a wide majority are administered as either intramuscular or subcutaneous injections, which contributes to the segments’ dominance.
The oral administration segment is expected to grow at a CAGR of 4.6% during the forecast period. The advantages of orally administered vaccines such as elicitation of both mucosal and systemic immunities and elimination of injection-associated adverse events are a few factors driving market growth. Polio vaccine, rotavirus vaccine, and other such pediatric vaccines are generally administered via the oral route.
Disease Indication Insights
This segment dominated the vaccine market with a share of 85.13%, majorly attributed to COVID-19 vaccines. The market is driven by factors such as increasing the incidence of viral diseases, growing awareness about the benefits of vaccination, and government initiatives to promote immunization programs. The market is highly competitive and is dominated by major players such as Pfizer, GSK, AstraZeneca, and Serum Institute amongst others. The viral diseases segment is further segmented into Hepatitis, Influenza, HPV, MMR, Rotavirus, Herpes Zoster, COVID-19, and others.
The market for bacterial diseases is expected to grow at a significant CAGR during the forecast period. The growing demand for vaccination that can prevent bacterial infections, particularly in regions with high incidences of bacterial diseases is one of the major factors driving segment growth. This market includes the development and distribution of vaccination for a range of bacterial infections, such as pneumonia, meningitis, DPT, and others. In February 2023, BactiVac, the Bacterial Vaccines Network, received USD 1.25 million in funding from Wellcome to accelerate the development of bacterial vaccines and counteract the threat of antimicrobial resistance (AMR). The funding will be utilized to amplify the impact of the BactiVac Network during the next four years.
Age Group Insights
The adult segment accounted for a share of 74.35% of the market in 2022. In 2021, adult vaccination, including those for COVID-19, comprised 75% in terms of volume globally, while pediatric vaccines accounted for 20%. In comparison to 2019, adult vaccine volumes had a nine-fold increase primarily due to COVID-19 vaccination. Additionally, non-COVID-19 adult vaccine volumes increased by 15% because of the widespread use of seasonal influenza vaccination in high-income countries (HICs).
The pediatric segment is expected to grow at the fastest CAGR over the forecast period. During the COVID-19 pandemic, overall pediatric vaccines volumes decreased by 14% compared to the pre-pandemic baseline of 2019, which was driven by a decreased use of oral polio vaccine (OPV) and measles-rubella vaccine (MR) in supplemental immunization activities (SIAs). The approval of COVID vaccines for pediatric use will further boost segment growth. For instance, in June 2022, Pfizer/BioNTech COVID vaccine received emergency use approval (EUA) for use in children aged 6 months to 4 years of age.
Distribution Channel Insights
The government suppliers segment dominated the vaccine market in 2022 with a share of 64.36%. In contrast to other pharmaceutical products, vaccines are primarily financed by public funds through government and pooled procurement arrangements, with private sector procurement playing a minor role. Concentrated demand can assist in planning the necessary supply investments. However, the predictability of demand remains a significant factor affecting access to vaccinations. For instance, the U.S. government has recently announced that it plans to purchase more than 1.5 million Novavax Inc.'s COVID-19 vaccine doses in February 2023. As per the recent agreement, funds have been allocated for the development of an updated vaccine, expected to be ready by the fall of the same year.
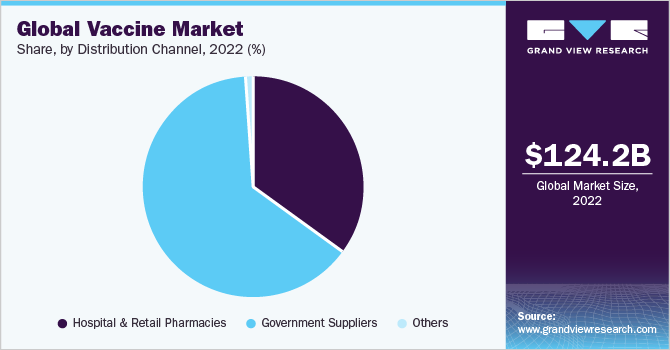
Hospital and retail pharmacies supply channel is estimated to be the fastest growing distribution channel in the vaccine market. Moreover, pharmacies have become an increasingly important source of vaccine supply in many developed countries. They provide convenient access to public vaccination and help increase vaccination rates. Furthermore, the opening of the private market for COVID vaccines in 2023 in the U.S. will also contribute to the market growth.
Regional Insights
The Asia Pacific region dominated the vaccine market with a share of 34.66% in 2022. With a rising demand for preventative healthcare, improving healthcare infrastructure, and increasing funding for vaccine research and development, the Asia-Pacific region is expected to continue its growth in the coming years. Key factors contributing to the dominance of this region include the large population base, increasing focus on immunization programs, and rising awareness of the benefits of vaccination. The distribution of vaccines is dominated by the South-East Asia Region (WHO South-East Asia Region) and Western Pacific Region (WHO Western Pacific Region), which account for over 50% of total vaccine doses, with China (22%) and India (17%) being the largest contributors.
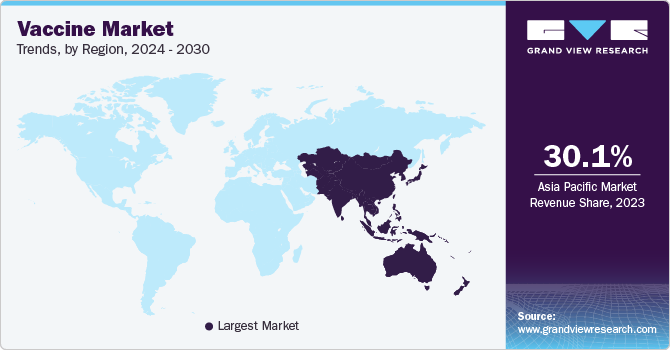
The North America market is expected to grow at the fastest CAGR during the forecast period, owing to the commercialization of COVID-19 vaccines in the U.S., national immunization programmes for other diseases, and high healthcare expenditure. The privatization of COVID-19 vaccines will lead to price increases and the entry of private insurers into the vaccines ecosystem.
Key Companies & Market Share Insights
The vaccine market is highly competitive, with a few large players. The vaccine supply landscape underwent a significant change in 2021 due to the pandemic. Many manufacturers entered the market to capture the global share, however, the top 10 manufacturers accounted for almost 90% of the global value, reflecting greater market concentration than all other vaccines.Companies are engaged in the development of vaccines using new technology for diseases such as RSV, flu, and influenza. For instance, in April 2023, Pfizer announced data from phase 3 trials of its RSV shots. The trial conducted in adult patients demonstrated that the shot was 67% more effective in preventing infections with two related symptoms and 86% effective in case of severe disease. The successful approval of this vaccine will make it the first product in the RSV market. Some prominent players in the global vaccine market include:
-
Serum Institute of India Pvt. Ltd.
-
Seqirus
-
Sanofi
-
GSK Plc.
-
Merck & Co., Inc.
-
Pfizer Inc.
-
Moderna Inc.
-
Sinovac
-
BioNTech SE
-
AstraZeneca
Vaccine Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 102.94 billion
Revenue forecast in 2030
USD 114.66 billion
Growth rate
CAGR of 1.4% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Report updated
July 2023
Quantitative units
Revenue in USD billion and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, route of administration, disease indication, age group, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Germany; UK; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Mexico, Argentina; South Africa; Saudi Arabia, UAE; Kuwait
Key companies profiled
Serum Institute of India Pvt. Ltd.; Seqirus, Sanofi; GSK Plc.; Merck & Co. Inc.; Pfizer Inc.; Moderna Inc.; Sinovac; BioNTech SE; AstraZeneca
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Global Vaccine Market Report Segmentation
This report forecasts revenue growth and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this report, Grand View Research has segmented the vaccine market based on type, route of administration, disease indication, age group, distribution channel, and region:
-
Type Scope Outlook (Revenue, USD Billion, 2018 - 2030)
-
Subunit Vaccines
-
Recombinant vaccines
-
Conjugate Vaccines
-
Toxoid vaccines
-
-
Inactivated
-
Live Attenuated
-
mRNA vaccines
-
Viral vector vaccines
-
-
Route of Administration Outlook (Revenue, USD Billion, 2018 - 2030)
-
Oral
-
Parenteral
-
Nasal
-
-
Disease Indication Outlook (Revenue, USD Billion, 2018 - 2030)
-
Viral Diseases
-
Hepatitis
-
Influenza
-
HPV
-
MMR
-
Rotavirus
-
Herpes Zoster
-
Covid-19
-
Others
-
-
Bacterial Vaccines
-
Meningococcal Diseases
-
Pneumococcal diseases
-
DPT
-
Others
-
-
Cancer Vaccines
-
Allergy Vaccines
-
-
Age Group Outlook (Revenue, USD Billion, 2018 - 2030)
-
Pediatric
-
Adult
-
-
Distribution Channel Scope Outlook (Revenue, USD Billion, 2018 - 2030)
-
Hospital & Retail Pharmacies
-
Government Suppliers
-
Others
-
-
Regional Outlook (Revenue, USD Billion, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global vaccine market size was estimated at USD 124.2 billion in 2022 and is expected to reach USD 102.94 billion in 2023.
b. The global vaccine market is expected to grow at a compound annual growth rate of 1.4% from 2023 to 2030 to reach USD 114.66 billion by 2030.
b. The viral diseases segment dominated the vaccine market with a share of 85.13% in 2022. This is attributable to the increasing incidence of viral diseases, growing awareness about the benefits of vaccination, and government initiatives to promote immunization programs.
b. Some key players operating in the vaccine market include Merck &Co., Inc., Emergent BioSolutions, Inc., Johnson and Johnson Services, Inc., Sanofi, Pfizer, Inc., Novartis AG, CSL Ltd., and GlaxoSmithKline plc.
b. Key factors that are driving the market growth include rising demand for better healthcare infrastructure and high awareness levels of the benefits of immunization.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





