- Home
- »
- Market Trend Reports
- »
-
Bimekizumab (Bimzelx) Market Outlook: Dual IL-17 Inhibition, Patent Positioning, & Competitive Landscape Analysis
Report Overview
Bimekizumab (Bimzelx), developed by UCB, is emerging as a highly differentiated biologic in the global immunology market through its unique dual inhibition of IL-17A and IL-17F, delivering rapid, deep, and sustained clinical responses that surpass many existing IL-17 therapies. Since securing FDA approval for plaque psoriasis in 2023 and expanding into psoriatic arthritis, non-radiographic axial spondyloarthritis, ankylosing spondylitis, and moderate-to-severe hidradenitis suppurativa, the therapy has steadily strengthened its competitive position across dermatology and rheumatology segments. Supported by robust Phase III data showing high PASI 90 and PASI 100 clearance rates, Bimekizumab is gaining accelerated adoption in the U.S. and EU, where clinicians increasingly view it as a superior option to single-pathway IL-17 inhibitors and select TNF blockers. As UCB navigates competition from secukinumab, ixekizumab, and emerging IL-23 agents, the company is leveraging clinical differentiation, broad label expansion, and strategic global market rollouts to solidify its position. Real-world evidence continues to validate its durable efficacy, reinforcing Bimekizumab as a next-generation therapy in autoimmune and inflammatory disease management.
Key Report Deliverables
-
A comprehensive analysis of the Bimekizumab (Bimzelx) market landscape, including global revenue performance, uptake trends across dermatology and rheumatology indications (plaque psoriasis, psoriatic arthritis, non-radiographic axial spondyloarthritis, ankylosing spondylitis, and hidradenitis suppurativa), and the evolving treatment paradigms driven by dual IL-17A/IL-17F inhibition, which differentiates the therapy from existing IL-17A and TNF-alpha inhibitors.
-
Forecasts evaluating post-launch market dynamics, covering expected geographic expansion across major regions, anticipated penetration in newly approved rheumatology and dermatology indications, and projected impacts on long-term revenue growth as Bimekizumab competes with established agents such as secukinumab and ixekizumab, as well as emerging IL-23-based biologics that are reshaping prescribing behavior.
-
Identification of regulatory and market access factors influencing Bimekizumab adoption, including pricing assessments, payer-driven step therapy requirements, formulary positioning, reimbursement variations across the U.S., Europe, Japan, and emerging markets, and the role of real-world evidence in securing favorable health technology assessments.
-
An in-depth competitive landscape overview, profiling leading IL-17 and IL-23 inhibitor developers, late-stage pipeline entrants, evolving therapeutic strategies across chronic inflammatory diseases, strategic partnerships, and innovation trends such as dual-pathway cytokine blockade and long-acting formulations that could shift future treatment preferences.
-
Strategic implications for UCB, including lifecycle management initiatives, continued indication expansion, optimization of dosing and delivery platforms, pricing and contracting strategies, and global commercialization approaches aimed at sustaining market leadership and maximizing Bimekizumab’s long-term value across the autoimmune and inflammatory disease therapeutic ecosystem.
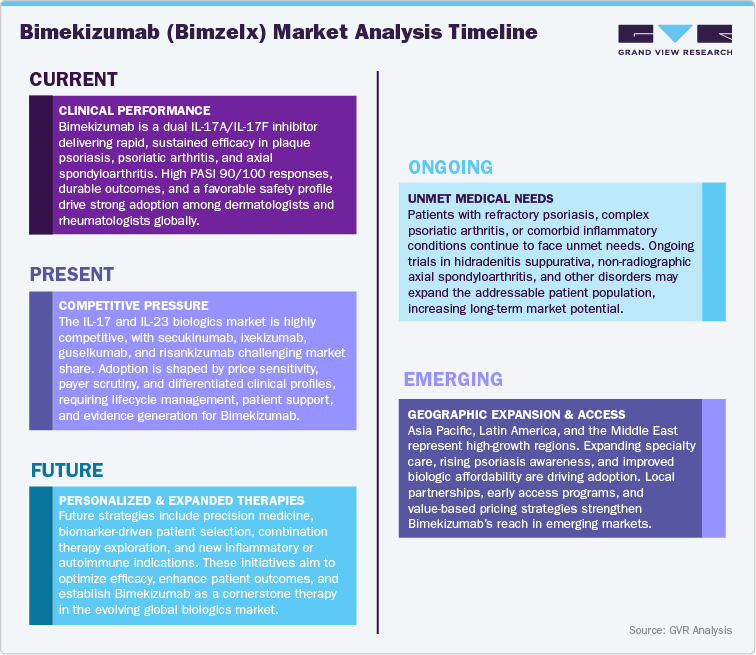
Current Market Scenarios
Bimekizumab (Bimzelx), developed by UCB, has rapidly established itself as a high-impact biologic within the global immunology market, leveraging its differentiated dual inhibition of IL-17A and IL-17F to deliver superior clinical outcomes across multiple chronic inflammatory diseases. Following its approvals in moderate-to-severe plaque psoriasis and subsequent expansion into psoriatic arthritis, non-radiographic axial spondyloarthritis, ankylosing spondylitis, and hidradenitis suppurativa, Bimekizumab is solidifying its commercial footprint across key therapeutic segments. Its robust efficacy-characterized by high PASI 90 and PASI 100 clearance rates-combined with durable long-term outcomes and a consistent safety profile, has positioned it as a preferred next-generation biologic among dermatologists and rheumatologists.
In the U.S. and Europe, market adoption continues to accelerate, driven by strong clinician confidence, favorable reimbursement decisions, and a growing shift toward dual-pathway IL-17 targeting as a new benchmark in skin and joint disease management. Strategic pricing, optimized dosing options, and expanding real-world evidence are reinforcing its competitive advantage over established IL-17A inhibitors such as secukinumab and ixekizumab, as well as IL-23 agents that previously dominated the psoriasis treatment landscape. Meanwhile, in the Asia Pacific region including Japan, Australia, and emerging Southeast Asian markets-rising awareness of advanced biologics, strengthening regulatory pathways, and expanding payer support are creating significant opportunities for early and sustained uptake.
Across Latin America and the Middle East & Africa, improvements in biologics accessibility, greater investment in specialty care infrastructure, and partnerships with regional distributors are broadening patient reach, though variability in reimbursement frameworks and budget constraints remain ongoing hurdles to rapid penetration. Despite intensifying competition in the immunology ecosystem, Bimekizumab’s clinical differentiation, expanding label coverage, and UCB’s strategic commitment to global commercialization and patient access initiatives position the therapy for continued growth. Looking ahead, the market trajectory will be influenced by further indication expansions, competitive pressure from next-wave cytokine inhibitors, and ongoing innovation in biologic delivery platforms factors that collectively reinforce Bimekizumab’s role as a key driver within the evolving autoimmune and inflammatory disease treatment landscape.
Market Dynamics
Growing Demand for Advanced Immunology Therapies
The rising global burden of chronic autoimmune and inflammatory disorders-such as plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, non-radiographic axial spondyloarthritis, and hidradenitis suppurativa-is fueling strong demand for next-generation biologics like Bimekizumab (Bimzelx). As the first approved therapy targeting both IL-17A and IL-17F, Bimekizumab delivers deeper inflammatory suppression and industry-leading skin and joint clearance rates, distinguishing it from single-pathway IL-17 inhibitors. Its versatile applicability across dermatology and rheumatology, combined with growing clinician confidence and robust long-term efficacy data, positions it as a cornerstone therapy for patients requiring high-intensity disease control. Supported by favorable early adoption trends, broad label expansion, and persistent unmet need in immune-mediated diseases, Bimekizumab is poised for sustained global growth across major and emerging markets.
Pricing Dynamics and Competitive Landscape
As the IL-17 inhibitor segment continues to evolve, competition from established biologics such as secukinumab, ixekizumab, and newer IL-23 agents is expected to influence pricing, payer decision-making, and access strategies. In cost-sensitive markets including Europe, China, and India, payers are increasingly focused on comparative effectiveness and real-world value, prompting manufacturers to adopt competitive pricing and value-based contracting approaches. In contrast, in markets like the U.S. and Japan-where physician preference for high-efficacy therapies and early reimbursement pathways are strong-Bimekizumab is well-positioned to maintain premium pricing backed by differentiated dual-pathway efficacy. Although pricing pressure may intensify as more IL-17 and IL-23 competitors secure broader indications, Bimekizumab’s superior PASI 90/100 performance, durable outcomes, and expanding clinical footprint will continue to anchor its competitive advantage.
Opportunities in Lifecycle Management and Regional Expansion
UCB is proactively strengthening Bimekizumab’s market resilience through a comprehensive lifecycle strategy, including continued indication expansion, enhancement of dosing and delivery options, and clinical evaluation in broader inflammatory conditions. The company’s emphasis on generating real-world evidence, engaging dermatology and rheumatology key opinion leaders, and advancing global market access initiatives is expected to support long-term therapy differentiation. Regional performance will vary mature markets will sustain steady biologic adoption driven by clinician familiarity and reimbursement stability, while emerging regions particularly Asia Pacific, Latin America, and the Middle East are likely to experience accelerated growth due to improving specialty care infrastructure, increasing biologics access programs, and strategic regional partnerships. Moving forward, innovation-driven differentiation, expanding therapeutic relevance, and targeted commercialization will underpin Bimekizumab’s continued leadership in the dynamic autoimmune and inflammatory disease treatment landscape.
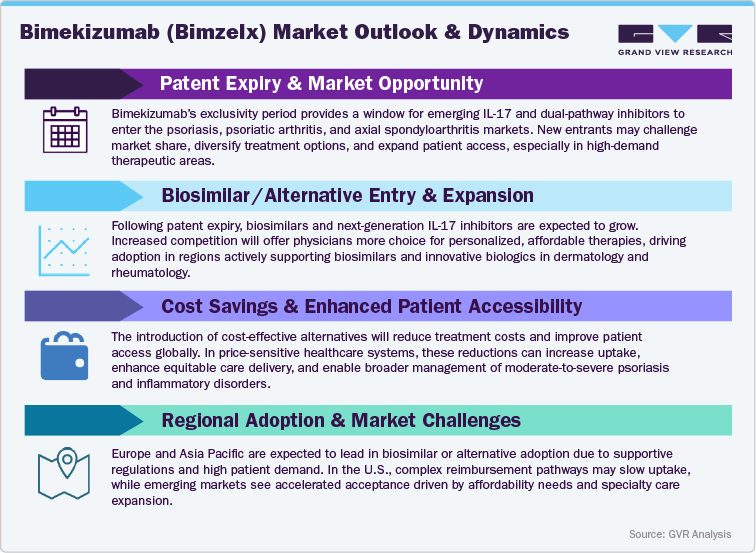
The Pressure of Pricing and Market Erosion Amid Competitive Biologics
As Bimekizumab (Bimzelx) scales its presence across dermatology and rheumatology indications, the therapeutic biologics market is confronting growing pricing pressures and competitive headwinds. With established IL-17A agents (such as secukinumab and ixekizumab) and emerging IL-23 inhibitors (for example guselkumab and risankizumab) expanding their label coverage and discounting strategies, payers are intensifying scrutiny of value, durability, and real-world effectiveness. In cost-sensitive regions-especially China, India, Latin America and parts of the Middle East-governments are deploying cost-containment frameworks, encouraging biosimilar entry, and prioritizing formulary inclusion of competitively priced biologics, which could erode pricing flexibility. In contrast, in mature markets such as the U.S., Europe and Japan, prescriber loyalty toward high-efficacy therapies, established clinical trust, and differentiated positioning allow Bimekizumab to maintain premium pricing in the short term. Nevertheless, as additional dual-cytokine inhibitors, long-acting formulations, and biosimilar IL-17/IL-23 products emerge, the pricing choreography and reimbursement landscape will evolve requiring proactive contracting, outcome-based models, and lifecycle strategy. Despite these dynamics, Bimekizumab’s strong efficacy data, long-term durability, and UCB’s disciplined commercial execution provide resilience. Strategic investment in patient access programs, real-world outcomes generation, and flexible global pricing approaches remain vital for sustaining market leadership.
Innovating Beyond the Core Patent - Unlocking Future Growth Paths
In response to intensifying competition and tighter payer regimes, UCB is actively advancing innovation-driven strategies to extend Bimekizumab’s lifecycle and fortify its competitive moat. These efforts include exploring new delivery formats (such as less frequent dosing, autoinjector options), expanding into additional autoimmune and inflammatory disease indications (for example dermatologic niches beyond psoriasis, rare rheumatologic or inflammatory-skin conditions), and generating real-world evidence to support value-based care models. Regional growth prospects are particularly strong in Asia Pacific, Latin America, and the Middle East, where rising biologic adoption rates, expanding specialty care infrastructure, and evolving reimbursement policies for advanced therapies are unlocking broader access. As the global immunology market matures, UCB’s strategy centered on clinical differentiation, global access expansion, and outcome-driven contracting will be instrumental in keeping Bimekizumab a central player in the future of biologic therapy for inflammatory and autoimmune disease.
Shaping the Future - Innovation, Accessibility, and Regional Dynamics
The global dermatology and immunology landscape is undergoing a major shift, with Bimekizumab (Bimzelx) emerging as a transformative therapy driving next-generation treatment standards. By uniquely inhibiting both IL-17A and IL-17F, Bimekizumab delivers rapid, deep, and sustained skin clearance setting a new clinical benchmark in moderate-to-severe plaque psoriasis and expanding its relevance across psoriatic arthritis and axial spondyloarthritis. As patient and physician expectations evolve toward therapies offering faster onset, higher PASI responses, and long-term durability, Bimekizumab’s dual-pathway mechanism positions it as a differentiated alternative to established IL-17 and IL-23 inhibitors.
The growing adoption of value-based care models and accelerated reimbursement pathways is expected to support Bimekizumab’s penetration in mature markets such as the U.S., Europe, and Japan, where specialist dermatology networks and biologic-friendly formularies provide strong commercial footing. In emerging regions-including Asia Pacific, Latin America, and the Middle East-increasing biologic accessibility, improving diagnostic ecosystems, and the expansion of public insurance coverage will be central to unlocking broader uptake. Nonetheless, variations in regulatory sequencing, physician familiarity, and affordability may create region-specific adoption trajectories.
To maintain long-term leadership, UCB’s strategy is anchored in lifecycle innovation-spanning additional autoimmune indications, enhanced delivery platforms, and potential real-world differentiation versus IL-17A and IL-23 competitors. Complemented by patient support initiatives, clinician education programs, and regionally tailored market access approaches, Bimekizumab is well positioned to remain a cornerstone therapy in the evolving immunology arena-balancing scientific innovation, broader accessibility, and meaningful patient outcomes on a global scale.

Overview of Alternative Therapeutics
The competitive environment surrounding Bimekizumab (Bimzelx) is rapidly intensifying as multiple IL-17 and IL-23 inhibitors continue to expand their clinical and commercial footprints. Leading alternatives such as secukinumab (Cosentyx), ixekizumab (Taltz), risankizumab (Skyrizi), and guselkumab (Tremfya) are strengthening their presence across plaque psoriasis, psoriatic arthritis, and axial spondyloarthritis-creating a dynamic marketplace defined by expanding biologic options and shifting treatment preferences. These agents, each offering differentiated mechanisms, dosing intervals, and long-term efficacy profiles, are enhancing physician choice and supporting tailored treatment strategies across diverse autoimmune patient populations.
As competitors secure additional indications, real-world evidence, and broader international approvals, innovation within the IL-17 and IL-23 therapeutic segments is accelerating. Uptake of alternative biologics is expected to be particularly pronounced in value-conscious regions such as Asia Pacific, Latin America, and parts of Europe, where pricing structures, reimbursement criteria, and biologic accessibility significantly shape prescribing behaviors. Meanwhile, in mature markets including the U.S. and Japan, factors such as dermatologist familiarity, rapidity of clinical response, durability of remission, and patient convenience will be increasingly important in guiding therapy selection and influencing market share.
To sustain and expand its leadership, UCB is expected to leverage Bimekizumab’s unique dual inhibition of IL-17A and IL-17F an advantage linked to superior skin clearance rates and durable responses. Strategic emphasis on lifecycle management, expanded autoimmune indications, subcutaneous delivery optimization, and enhanced patient support infrastructures will be pivotal in strengthening competitive positioning. As IL-17 and IL-23-based therapies continue to redefine standards of care in dermatology and rheumatology, Bimekizumab’s differentiated mechanism, strong efficacy profile, and global commercial expansion efforts are set to shape its long-term role in this increasingly competitive immunology landscape.
Competitive Landscape
The competitive landscape for Bimekizumab (Bimzelx) is intensifying as the IL-17 and IL-23 inhibitor segments continue to expand across dermatology and rheumatology. UCB is positioning Bimekizumab as a next-generation therapy with its unique dual inhibition of IL-17A and IL-17F, delivering superior skin clearance, rapid onset of efficacy, and durable responses across plaque psoriasis, psoriatic arthritis, and axial spondyloarthritis. Competing therapies-including secukinumab (Cosentyx), ixekizumab (Taltz), guselkumab (Tremfya), and risankizumab (Skyrizi) are broadening their market reach through additional indications, optimized dosing schedules, and expanded geographic presence, intensifying the competitive race in the biologic immunology market.
Emerging competitors are advancing novel IL-17, IL-23, and dual-pathway biologics through late-stage clinical trials, aiming to address gaps in efficacy, dosing convenience, and long-term durability. Market adoption is expected to vary regionally: North America and Europe are projected to remain key revenue drivers due to mature reimbursement frameworks, specialist familiarity, and strong clinician confidence, while Asia Pacific, Latin America, and the Middle East are likely to experience faster uptake, driven by expanding access to specialty care, improving biologic affordability, and growing physician awareness.
To sustain and expand leadership, UCB is emphasizing strategic differentiation, including additional autoimmune and inflammatory indications, subcutaneous delivery optimization, and targeted patient support programs. Competitors are pursuing aggressive pipeline diversification, real-world evidence generation, and competitive pricing strategies to capture market share. In this evolving immunology landscape, long-term success will hinge on clinical superiority, consistent real-world performance, and patient-centric outcomes, with Bimekizumab’s dual-pathway efficacy and broad global rollout positioning it as a pivotal player in shaping the future of autoimmune and inflammatory disease management.
North America Bimekizumab (Bimzelx) Market
North America leads the Bimekizumab market, with the U.S. serving as the primary launch hub for UCB’s dual IL-17A/IL-17F inhibitor. Rapid adoption by dermatologists and rheumatologists, strong clinical awareness, and favorable reimbursement policies underpin the region’s dominance. The U.S. benefits from early FDA approvals across plaque psoriasis, psoriatic arthritis, and axial spondyloarthritis, integration into specialty care networks, and widespread familiarity with biologic treatment regimens. Canada mirrors these trends, though formulary restrictions and cost-containment measures moderate uptake. Overall, North America’s advanced immunology infrastructure and innovation-friendly ecosystem make it a critical revenue driver for Bimekizumab.
Europe Bimekizumab (Bimzelx) Market
Europe represents a key growth territory for Bimekizumab, with Germany, France, and the U.K. leading adoption. Established autoimmune treatment networks, strong specialist engagement, and growing experience with biologics are driving uptake in psoriasis, psoriatic arthritis, and axial spondyloarthritis. Health technology assessments (HTAs) and national payer negotiations may constrain premium pricing, but increasing recognition of dual-pathway IL-17 inhibition as a best-in-class approach is expected to accelerate adoption. Partnerships with academic centers, patient support initiatives, and early access programs are critical to securing long-term presence across European markets.
Asia Pacific Bimekizumab (Bimzelx) Market
The Asia Pacific region offers high-growth potential for Bimekizumab, led by Japan, Australia, and South Korea. Japan’s strong biologic innovation ecosystem and clinician familiarity with targeted immunotherapies support early adoption. In China, India, and Southeast Asia, rising psoriasis awareness, expanding specialty care infrastructure, and government initiatives to improve biologic access are driving patient reach, although pricing and regulatory requirements may influence adoption speed. Collaborations with regional distributors, payer engagement, and patient access programs are expected to enhance UCB’s footprint, positioning Asia Pacific as a strategic growth frontier.
Latin America Bimekizumab (Bimzelx) Market
Latin America is an emerging yet cost-sensitive market for Bimekizumab, with Brazil, Mexico, and Argentina leading adoption. Increasing psoriasis diagnosis rates and the expansion of dermatology and rheumatology specialty centers are creating demand for advanced biologics. Limited reimbursement coverage and affordability challenges remain significant barriers. Early-entry strategies including tiered pricing, compassionate use programs, and partnerships with local healthcare providers will be pivotal for improving accessibility. As regulatory frameworks mature, Latin America is expected to witness steady uptake of dual IL-17 inhibitor therapies, with Bimekizumab positioned as a premium, high-efficacy option.
Middle East and Africa Bimekizumab (Bimzelx) Market
The Middle East and Africa (MEA) region represents a developing opportunity for Bimekizumab, with Saudi Arabia, UAE, and South Africa driving initial adoption. Growing awareness of chronic inflammatory diseases and expanding specialty care infrastructure are improving access, though high biologic costs limit broad penetration. Accelerated regulatory pathways in GCC countries will facilitate early entry, while wider African markets may experience gradual adoption. Investments in clinician education, hospital partnerships, and regional patient access programs are expected to expand uptake, making MEA an increasingly important frontier for dual-pathway IL-17 inhibitor therapies.

Analyst Perspective
The Bimekizumab (Bimzelx) market is entering a transformative growth phase as UCB solidifies its position in the IL-17 and dual-pathway biologics segment, addressing a broad spectrum of dermatologic and rheumatologic disorders. Following approvals for moderate-to-severe plaque psoriasis, psoriatic arthritis, and axial spondyloarthritis, Bimekizumab is rapidly gaining traction across additional inflammatory indications, supported by its unique dual inhibition of IL-17A and IL-17F, high PASI 90/100 response rates, and durable long-term efficacy. Competition from established IL-17 inhibitors such as secukinumab (Cosentyx) and ixekizumab (Taltz), as well as emerging IL-23 agents including guselkumab and risankizumab, is intensifying market dynamics and pricing pressures, particularly in mature regions like the U.S., Europe, and Japan.
Bimekizumab’s early adoption advantage, differentiated dual-pathway mechanism, and scalable biologic production position UCB favorably for sustained commercial growth. Its rapid onset of clinical response, durable skin and joint improvements, and strong safety profile are enabling expanded patient access and driving confidence among dermatologists and rheumatologists alike.
To maintain momentum, UCB is focusing on lifecycle management, including additional autoimmune and inflammatory indications, optimized delivery formats, and real-world evidence generation to reinforce clinical value across diverse geographies. Patient-centric initiatives such as support programs, education for specialty centers, and improved dosing convenience-are expected to further enhance therapy adoption and brand loyalty. Strategic attention to global market access, emerging region expansion, and value-based reimbursement approaches will be critical in sustaining Bimekizumab’s leadership in the evolving biologics landscape and securing its role as a cornerstone therapy in next-generation autoimmune and inflammatory disease management.

Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028-2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU -15-30% Yr-1, deepening to -45-60% by Yr-3; U.S. -10-25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25-40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30-40% global revenue decline by Year-3 post-LOE.
-
Downside: 45-55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~-20-25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


