- Home
- »
- Market Trend Reports
- »
-
Dulaglutide (Trulicity) Market: Navigating The Patent Cliff And Strategic Implications
Report Overview
Dulaglutide (Trulicity) is a once-weekly GLP-1 receptor agonist used in type 2 diabetes management and for lowering cardiovascular risk in patients with underlying conditions. It supports glycemic control, weight management, and cardiovascular protection, while weekly dosing improves treatment adherence. Adoption is increasing with the growing diabetes population and wider use of GLP-1 therapies in clinical practice. Key challenges include gastrointestinal side effects, cost barriers, and rising competition from newer GLP-1 agonists offering broader benefits. Its role is expected to expand as treatment guidelines continue to position GLP-1 therapies as standard options in diabetes and cardiovascular risk management.
Key Report Deliverables
-
Analyze the Dulaglutide (Trulicity) market landscape, detailing its current size, growth drivers, and key industry trends particularly in light of the upcoming patent expiration.
-
Forecast market growth by projecting future trends, highlighting emerging opportunities, and assessing potential risks to growth.
-
Identify regulatory and market barriers, providing insights into challenges that could impact future market expansion and product development.
-
Concurrent Competitive Landscape, identifying key players, examining both direct and indirect competitors within the market, their strategic moves, and the distribution of market share to understand competitive positioning.
-
Regulatory Barriers, identify key regulatory challenges related to biosimilar entry and their potential impact on market expansion.
-
Strategic Implications, evaluating strategic moves for Eli Lilly & Company and competitors to maintain market leadership, including innovation, pricing, and geographic expansion.
Patent Cliff and Price Erosion Analysis
-
A detailed assessment of expected sales erosion following the expiry of key patents.
-
Evaluation of price declines triggered by the first generic launch and subsequent competitor entries.
-
Quantification of brand revenue loss due to rapid payer-driven substitution toward lower-cost generics.
-
Analysis of market share displacement, highlighting how quickly branded volume shifts to multi-entrant generic competitors.
-
A two-phase erosion model showing initial impact from early entry and steeper decline as more generics launch.
-
Insights into purchasing behavior, formulary changes, and contracting pressure post-generic entry.
-
Forecast of the brand’s transition into a value-driven, mature market environment.
Trulicity’s core composition patents and regulatory data exclusivity are projected to lapse in the mid-to-late 2020s, with exact expiry dates differing by country due to regional patent filings and local regulatory frameworks. Eli Lilly has layered in secondary patents covering delivery devices, formulations, and manufacturing processes, which could slow biosimilar entry in certain jurisdictions. The impact of this patent cliff will not be uniform worldwide. In the U.S. and EU, where biosimilar regulatory pathways are well established and payer systems actively encourage adoption, Trulicity is likely to face earlier and more aggressive biosimilar competition, leading to rapid erosion of market exclusivity. In contrast, markets in Asia-Pacific and Latin America may experience a delayed effect due to slower regulatory approval timelines, weaker biosimilar manufacturing capacity, and varied intellectual property enforcement. Nonetheless, once biosimilars are introduced in these regions, pricing pressures could be sharper given the higher cost sensitivity and prevalence of tender-based procurement models.
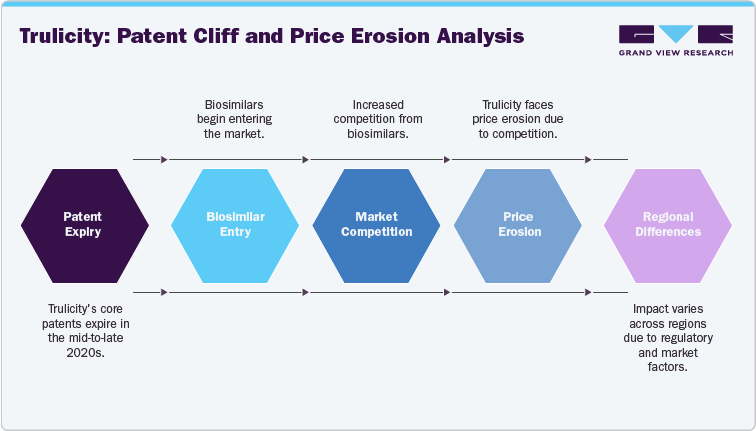
Following the patent cliff, Trulicity will face price erosion as biosimilars enter and payers renegotiate contracts. Unlike small-molecule generics, biosimilars typically cause gradual list-price reductions but more pronounced net revenue declines due to higher rebates, tendering, and competitive formulary management. In Europe, price drops are likely to be steep because of tender-based procurement, while in the U.S., pharmacy benefit managers (PBMs) will exert pressure through rebate escalation and formulary exclusion. In emerging markets such as India and Latin America, once biosimilars are approved, erosion may be sharper given high price sensitivity and government-driven procurement policies.
Current Market Scenarios

Trulicity (dulaglutide) continues to play an important role in type 2 diabetes management and cardiovascular risk reduction, supported by its once-weekly dosing, reliable glycemic control, and demonstrated cardiovascular protection. These attributes have established it as a widely adopted therapy among both primary care physicians and specialists, particularly in patients with comorbid cardiometabolic conditions. However, the competitive environment is shifting rapidly with the introduction of newer GLP-1 receptor agonists and dual agonists that show superior outcomes in weight loss and glycemic improvement. As patients and physician demand increasingly favors treatments with broader benefits, Trulicity risks losing share to alternatives were reimbursement policies and payer coverage support uptake. Its continued adoption will depend on maintaining differentiation in real-world outcomes and patient adherence advantages.
The market environment for Trulicity is shaped heavily by payer strategies and pricing pressures. In developed markets such as the U.S., payers are tightening formulary access by introducing stricter prior-authorization protocols, implementing step therapy, and negotiating deeper rebates, all of which affect Trulicity’s net pricing and competitiveness. While international markets offer more stable demand, competitive intensity remains high, limiting growth potential. To address these challenges, Eli Lilly’s strategic focus is on lifecycle management such as new delivery formats, device innovation, and potential combination therapies to sustain patient adherence and clinical relevance. At the same time, targeted expansion in emerging markets, where diabetes prevalence is rising and infrastructure is improving, presents an opportunity to offset slower growth in saturated markets. Trulicity’s long-term relevance will be tied to Lilly’s ability to adapt its strategy as clinical guidelines and payer frameworks increasingly shift toward broader GLP-1 adoption.
Market Dynamics

“Proven Efficacy and Simplified Dosing”
Trulicity’s sustained market presence is largely driven by its proven efficacy in type 2 diabetes management and its additional benefit of cardiovascular risk reduction. Clinical trial data have shown that it not only lowers HbA1c levels but also reduces the risk of major adverse cardiovascular events (MACE) in high-risk patients. This dual role aligns closely with treatment guidelines, which prioritize therapies that address both glycemic control and cardiovascular outcomes. As diabetes and cardiovascular disease are often comorbid, Trulicity’s profile supports broad prescribing in primary care and specialty settings, reinforcing its clinical and commercial relevance. This strong evidence base secures Trulicity’s ongoing role in managing patients with complex metabolic needs. Trulicity’s adoption is its once-weekly dosing format, which directly improves treatment adherence and patient persistence in real-world use. Compared with daily injections, the weekly administration reduces treatment burden and integrates more easily into patients’ routines. This advantage is particularly relevant in populations managing complex medication regimens, where adherence is critical to long-term outcomes. Physicians recognize the importance of simplified dosing, especially in chronic conditions like diabetes where non-adherence is a major barrier. The simplified regimen enhances both patient satisfaction and physician willingness to recommend Trulicity as a reliable therapy.
“Pricing pressure and competitive intensity”
Trulicity’s growth is increasingly constrained by pricing pressures and evolving payer dynamics. In the U.S., the drug faces reduced realized net prices due to tougher rebate negotiations, formulary tier restrictions, and broader use of prior-authorization protocols by insurers. These mechanisms are designed to manage healthcare spending but limit Trulicity’s revenue potential, even when prescription volumes remain steady. In cost-sensitive markets across Asia-Pacific and Latin America, high out-of-pocket costs continue to restrict patient access, particularly in public health systems that prioritize lower-cost alternatives. As a result, affordability remains a critical barrier that impacts adoption across both developed and emerging regions.
Intensifying competition within the GLP-1 class. Newer agents, especially tirzepatide, have demonstrated superior outcomes in weight management and glycemic control, gaining traction among physicians and quickly becoming preferred options for payers seeking better long-term health outcomes. This shift accelerates patient switching away from Trulicity, undermining its established position as a leading once-weekly GLP-1 therapy. Competitors are also expanding rapidly into new indications such as obesity, which further narrows Trulicity’s relevance in the broader metabolic care market. Without lifecycle innovations, such as combination therapies or delivery enhancements, Trulicity risks continued erosion of its competitive standing.
Emerging Market and Clinical Opportunities
The growing prevalence of type 2 diabetes in Asia-Pacific and Latin America represents a significant opportunity for Trulicity. These regions are experiencing rising obesity rates, lifestyle-related risk factors, and increasing awareness of advanced diabetes therapies. Governments are investing in prevention and management programs for diabetes, while healthcare infrastructure continues to improve. Although affordability remains a barrier, expanding reimbursement frameworks and partnerships with local stakeholders could enhance accessibility. By tailoring pricing strategies and leveraging its brand recognition, Eli Lilly can position Trulicity to capture demand in these growth markets, balancing slower adoption in saturated developed economies.
The evolution of clinical practice guidelines provides another opportunity for Trulicity. International and regional bodies increasingly recommend GLP-1 receptor agonists not only for glucose management but also for cardiovascular risk reduction. This trend aligns directly with Trulicity’s clinical profile and outcomes data, reinforcing its role in both primary care and specialist-led management of cardiometabolic diseases. As payers and providers shift toward guideline-driven decision-making, drugs with proven efficacy across multiple endpoints stand to benefit. Trulicity’s established presence offers an advantage, enabling continued adoption even as the GLP-1 competitive landscape becomes more crowded.
Trulicity’s demonstrated ability to lower the risk of major adverse cardiovascular events (MACE) presents an opportunity to extend its relevance beyond conventional diabetes care. Cardiovascular disease is the leading cause of morbidity and mortality in people with diabetes, and therapies that address both conditions are increasingly valued by clinicians and payers.
Eli Lilly can sustain Trulicity’s competitiveness through lifecycle management initiatives. Potential strategies include introducing improved delivery devices, expanding indications through label extensions, and developing combination therapies that address both glycemic control and weight management. These efforts would help maintain relevance against newer GLP-1 and dual agonists, which currently dominate due to superior clinical outcomes. Device innovation, such as user-friendly injection pens or digital health integration, could further enhance adherence and patient satisfaction. By focusing on innovation beyond the molecule itself, Lilly can extend Trulicity’s commercial life and protect market share in a highly competitive therapeutic class.
Key Therapeutic Competitors to Trulicity
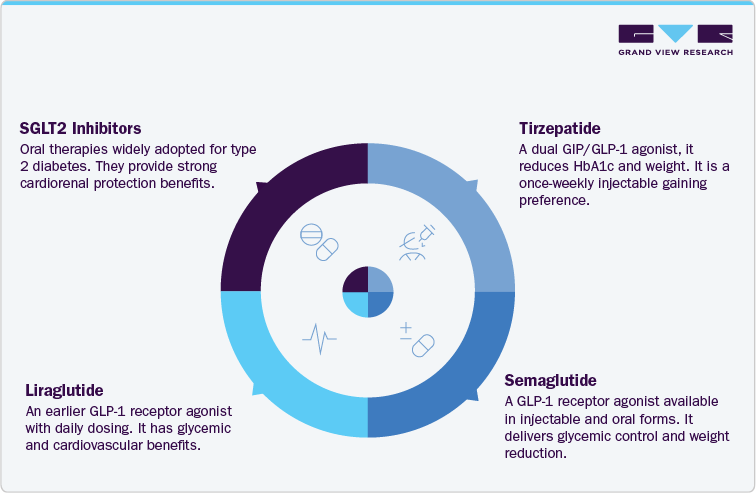
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has demonstrated superior outcomes in both glycemic control and weight loss compared to dulaglutide in head-to-head trials. Its once-weekly injectable format makes it convenient, while its efficacy in achieving double-digit weight loss has positioned it as a leading therapy in both type 2 diabetes and obesity management. Physicians are increasingly prescribing tirzepatide due to its broad cardiometabolic benefits, and payers are recognizing its value in reducing long-term disease complications. This rapid uptake makes tirzepatide the most direct competitor to Trulicity, particularly in markets where reimbursement supports access.
Semaglutide is a GLP-1 receptor agonist available in multiple formats, including a once-weekly injection (Ozempic), a higher-dose injectable for obesity (Wegovy), and an oral tablet (Rybelsus). It provides greater HbA1c reduction and weight loss compared to dulaglutide and has strong cardiovascular outcomes data from large-scale clinical trials. The availability of both injectable and oral options enhances patient choice and adherence, particularly among those hesitant to use injectables. Semaglutide’s expanding role across diabetes and obesity markets, combined with guideline endorsements, positions it as a preferred therapy and a strong alternative to Trulicity.
Liraglutide is an earlier-generation GLP-1 receptor agonist, administered as a once-daily injection. It is approved for type 2 diabetes management (Victoza) and for obesity treatment at higher doses (Saxenda). Liraglutide offers proven glycemic control and cardiovascular benefits, with long-standing real-world use supporting its credibility among prescribers. Although it delivers less weight reduction than semaglutide or tirzepatide, its established clinical profile, widespread availability, and dual approval for diabetes and obesity ensure it remains an important therapeutic option. Its role is particularly relevant in patients where daily dosing is acceptable, and cost or access considerations favor established products.
SGLT2 inhibitors represent a distinct therapeutic class and are widely prescribed in type 2 diabetes due to their cardiorenal protection benefits. These oral agents lower blood glucose by promoting urinary glucose excretion and have demonstrated strong evidence in reducing heart failure hospitalizations and slowing chronic kidney disease progression. While their glycemic effects are modest compared to GLP-1 receptor agonists, they are often used alongside or as alternatives when cardiovascular or renal outcomes are prioritized. Their oral formulation, broad adoption in guidelines, and complementary role in combination therapy make SGLT2 inhibitors a major competitor and partner class to Trulicity.
Competitive Landscape
Trulicity (dulaglutide), with its once-weekly dosing, proven glycemic control, and demonstrated cardiovascular outcomes data, has established itself as a widely prescribed therapy in type 2 diabetes management. Its consistent safety profile and strong physician familiarity support ongoing use, particularly in patients with overlapping cardiometabolic risks. This entrenched role in treatment pathways has built strong brand recognition and a recurring patient base. Access to Trulicity is shaped by payer decisions, formulary placement, and rebate negotiations. In the U.S. diabetes market, insurers are enforcing stricter utilization controls and demanding deeper discounts, reducing net revenue despite steady prescriptions. In Europe, tender-based procurement exerts similar downward pressure. These pricing challenges signal ongoing margin compression that will likely intensify under rising competitive pressure.
Eli Lilly is focusing on lifecycle management strategies, including device enhancements, label extensions, and adherence support programs. These initiatives are designed to preserve differentiation as payers and providers increasingly value outcomes-based therapies in diabetes and cardiovascular care. Improving patient experience through simplified delivery devices and generating further clinical evidence are central to sustaining market relevance. Trulicity’s patents are expected to expire in the mid-to-late 2020s, raising the risk of biosimilar entry and significant price erosion. While secondary patents may delay competition in some markets, the looming patent cliff poses a major threat to global revenues. Anticipated biosimilar launches, combined with aggressive payer negotiations, will intensify pricing pressure, making pre-expiry strategies essential to protect market share.
Regional Analysis
In the U.S. diabetes market, Trulicity (dulaglutide) faces strong commercial pressure as payers tighten formulary access, increase rebate demands, and apply stricter prior-authorization requirements. Eli Lilly’s financial disclosures confirm declining sales due to reduced prescription volumes and net pricing compression. While Trulicity remains widely prescribed, ongoing pricing pressure and competitive dynamics are eroding its market share, making outcomes-based contracting and patient-support programs critical for sustained relevance. In Europe, high GLP-1 adoption is coupled with aggressive tender-based procurement that accelerates price erosion and market-share decline once patent exclusivity ends. Varied reimbursement frameworks across EU member states increase complexity, with health technology assessments (HTAs) shaping access decisions. Maintaining presence will depend on country-specific strategies, stronger cardiovascular outcomes evidence, and lifecycle initiatives such as device improvements to secure favorable tender outcomes.
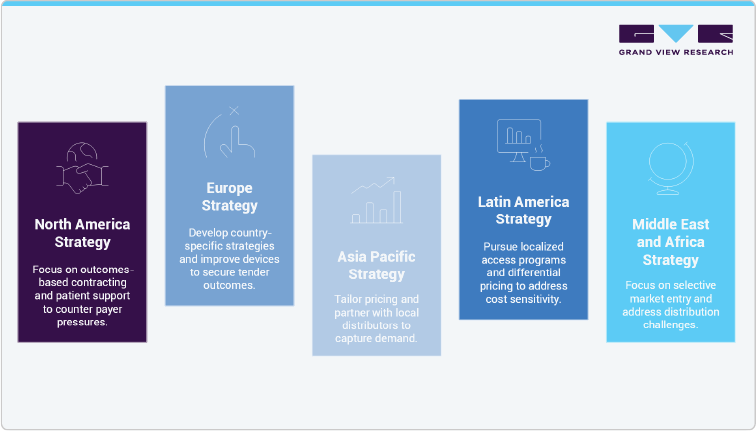
The Asia-Pacific (APAC) market presents strong growth potential for Trulicity due to rising diabetes prevalence and healthcare investment, but expansion is limited by affordability issues, regulatory delays, and fragmented reimbursement. Success will depend on differential pricing, local partnerships, and physician education. In Latin America, price sensitivity and government-led tendering constrain access, making localized access programs and collaboration with public health initiatives essential to address growing diabetes needs. The Middle East and Africa (MEA) regions are mixed: GCC countries offer growth through private insurance and higher affordability, while much of Africa faces barriers from weak infrastructure and limited budgets, requiring targeted partnerships and distribution support.
Analyst Perspective
Trulicity has been a cornerstone in the GLP-1 receptor agonist market, supported by once-weekly dosing convenience, proven glycemic control, and cardiovascular risk reduction data. These clinical advantages have translated into strong brand recognition, widespread physician familiarity, and a durable patient base. However, its competitive positioning has weakened as newer incretin therapies, especially tirzepatide and semaglutide, demonstrate superior outcomes in weight management and glycemic efficacy. Real-world trends confirm that payers and prescribers are gradually shifting preferences, resulting in volume and revenue pressure for Trulicity in key markets like the U.S.
From a commercial standpoint, Trulicity is entering a challenging phase. In North America and Europe, payer dynamics - including stricter formulary management, deeper rebate demands, and tender-driven procurement - are compressing net realizations even before the patent cliff. Core patents are expected to expire in the mid-to-late 2020s, creating risk of biosimilar entry and accelerating price erosion, particularly in Europe and price-sensitive markets such as Latin America and parts of Asia-Pacific. While secondary patents and device protections may delay competition in some jurisdictions, biosimilars and payer renegotiations remain significant threats to long-term revenue stability.
On the opportunity side, Trulicity can still leverage growth in emerging markets (APAC, LATAM, and GCC countries), where diabetes prevalence is climbing and healthcare access is expanding. However, success in these regions depends on differential pricing, localized access programs, and partnerships with governments and distributors. Additionally, lifecycle initiatives such as device enhancements, label extensions, and patient-support programs remain critical to sustaining differentiation and adherence advantages.
Overall, Trulicity’s near-term performance will rely on Lilly’s ability to manage gross-to-net pricing pressures, defend formulary position through outcomes-based contracting, and maximize returns in pre-expiry years. Longer term, its trajectory is constrained by the patent cliff, biosimilar competition, and therapeutic cannibalization from Lilly’s own tirzepatide portfolio. As such, while Trulicity retains relevance as part of guideline-based care, its strategic role is shifting from a growth driver to a cash-generating bridge product that supports Eli Lilly’s broader cardiometabolic portfolio until newer therapies dominate market share.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028-2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU -15-30% Yr-1, deepening to -45-60% by Yr-3; U.S. -10-25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25-40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30-40% global revenue decline by Year-3 post-LOE.
-
Downside: 45-55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~-20-25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


