- Home
- »
- Market Trend Reports
- »
-
Enhertu (Trastuzumab Deruxtecan) Market Outlook: Patent Cliff, Strategic Implications
Report Overview
Enhertu, marketed as Enhertu by Daiichi Sankyo and AstraZeneca, has become a key therapeutic for HER2-positive and HER2-low cancers, including advanced breast cancer, metastatic non-small cell lung cancer (NSCLC), and gastric cancer, with strong market performance driven by its targeted mechanism of action. However, as the expiration of its patents approaches, the market for Enhertu is expected to undergo significant changes. The impending patent cliff presents both challenges and opportunities for the companies, as biosimilars and competing ADCs prepare to enter. This article examines the strategic implications of the patent expiration for Daiichi Sankyo, AstraZeneca, and the broader oncology therapeutics landscape.
Key Report Deliverables
-
Analyze the ENHERTU market landscape, detailing its current size, growth drivers, and key industry trends particularly in light of the upcoming patent expiration.
-
Forecast market growth by projecting future trends, highlighting emerging opportunities, and assessing potential risks to growth.
-
Identify regulatory and market barriers, providing insights into challenges that could impact future market expansion and product development.
-
Concurrent Competitive Landscape, identifying key players, examining both direct and indirect competitors within the market, their strategic moves, and the distribution of market share to understand competitive positioning.
-
Regulatory Barriers, identify key regulatory challenges related to biosimilar entry and their potential impact on market expansion.
-
Strategic Implications, evaluating strategic moves for Novo Nordisk and competitors to maintain market leadership, including innovation, pricing, and geographic expansion.
Patent Cliff Analysis
-
ENHERTU’s key patent(s) are projected to expire in the U.S. in 2033, and in Europe between 2033 and 2035.
-
With patent expiry, biologic competition (biosimilars / competing ADCs) becomes possible, but entry will depend on regulatory pathways, data exclusivity, formulation / manufacturing complexity, and legal challenges.
-
Before 2033, ENHERTU will likely enjoy exclusivity in most major markets; post-2033, increased competitive pressure is expected, particularly in more mature healthcare systems and in regions with strong biosimilar/ADC development capacity.

Current Market Scenarios
Enhertu (trastuzumab deruxtecan) is under global clinical development in both monotherapy and combination regimens across HER2-targetable cancers, including trials with immunotherapies. The drug is approved in more than 75 countries for breast cancer, over 50 countries for non-small cell lung cancer (NSCLC), and around 65 countries for gastric cancer. Development strategies are focused on expanding indications into earlier lines of treatment and combination therapies, reinforcing its role in AstraZeneca and Daiichi Sankyo’s oncology portfolios.
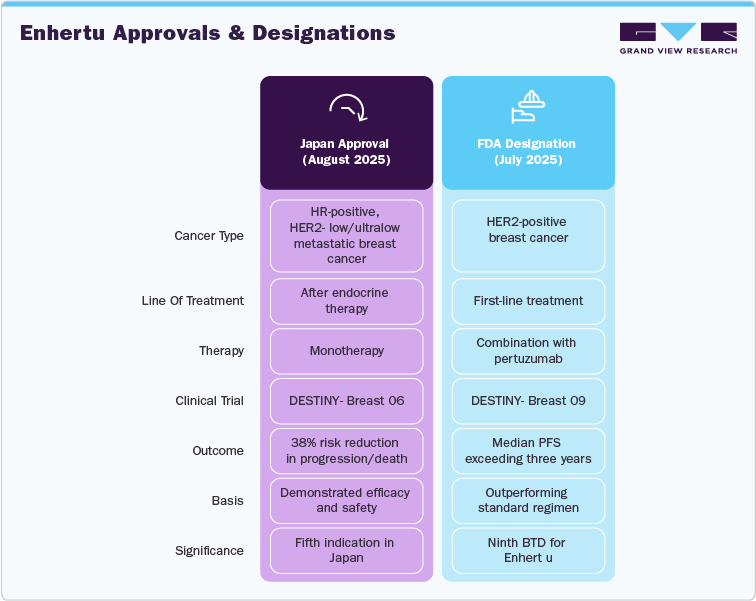
In August 2025, Japan’s Ministry of Health, Labour and Welfare approved Enhertu for adults with hormone receptor (HR)-positive, HER2-low or HER2-ultralow metastatic breast cancer after endocrine therapy. The DESTINY-Breast06 phase 3 trial demonstrated a 38% risk reduction in progression or death versus chemotherapy, with median progression-free survival (PFS) of 13.2 months compared to 8.1 months. The safety profile was consistent with prior studies, though interstitial lung disease (ILD) occurred in 20% of Japanese patients. This marks the fifth indication for Enhertu in Japan, extending treatment to broader HER2 expression categories.
Market Dynamics
Enhertu Market Dynamics
The market for Enhertu (trastuzumab deruxtecan) is defined by a combination of regulatory momentum, expanding clinical indications, and competitive positioning in the HER2-targeted therapy landscape. Approvals across more than 75 countries for breast cancer, over 50 for non-small cell lung cancer (NSCLC), and approximately 65 for gastric cancer highlight its rapid global penetration. Recent regulatory advances, such as Japan’s approval for HR-positive, HER2-low or HER2-ultralow metastatic breast cancer and the U.S. FDA’s Breakthrough Therapy Designation (BTD) for Enhertu in combination with pertuzumab in first-line HER2-positive breast cancer, underscore its growing role across disease settings.
Key market drivers include the high unmet need in HER2-low and ultralow populations, where Enhertu has demonstrated superior efficacy over chemotherapy, and the shift toward earlier-line therapy enabled by strong phase 3 outcomes such as DESTINY-Breast06 and DESTINY-Breast09. Expanding use beyond breast cancer into NSCLC, gastric, colorectal, and other solid tumors further diversifies the addressable market. Strategic collaboration between Daiichi Sankyo and AstraZeneca continues to support commercialization and pipeline development, ensuring global scale and presence.
Restraints are centered on safety considerations, particularly interstitial lung disease (ILD), which remains a key clinical management challenge, along with neutropenia and cardiac risks. In addition, competition from established HER2 therapies and emerging antibody-drug conjugates (ADCs) may affect pricing and market share.
Enhertu’s market dynamics reflect a balance of strong clinical efficacy, expanding indications, and strategic partnerships, tempered by safety concerns and competitive pressures. Its trajectory positions it as a cornerstone asset in the evolving HER2 oncology segment.
Innovating Beyond the Patent - Unlocking Future Growth Paths
Enhertu is transitioning from a second-line treatment to a cornerstone therapy in HER2-targeted oncology. Recent clinical advancements underscore its potential to redefine treatment paradigms across multiple cancer types.
In the first-line setting for HER2-positive metastatic breast cancer, Enhertu combined with pertuzumab demonstrated a median progression-free survival exceeding three years, surpassing the standard taxane, trastuzumab, and pertuzumab regimen. This marks the first significant improvement in over a decade for this patient population. Additionally, Enhertu has been approved in the U.S. as the first HER2-directed therapy for hormone receptor-positive, HER2-low or HER2-ultralow metastatic breast cancer patients who have progressed on endocrine therapy. This approval expands treatment options for a broader patient demographic
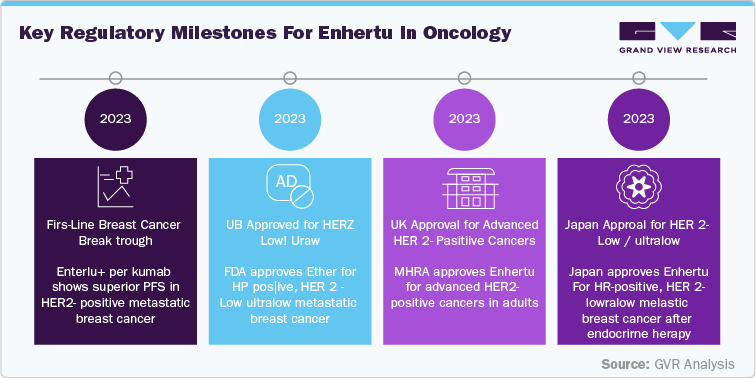
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has also approved Enhertu for adults with HER2-positive cancers that have spread or cannot be removed by surgery, further broadening its clinical application. In Japan, Enhertu has been approved for HR-positive, HER2-low or HER2-ultralow metastatic breast cancer after endocrine therapy, marking it as the first HER2-directed treatment for this group in the country.
These developments position Enhertu as a pivotal asset in the oncology landscape, with expanding indications and a robust clinical profile paving the way for sustained growth beyond its initial patent exclusivity.
“Driver Shaping the Future of Enhertu: Expansion of Clinical Utility Across HER2-Expressing Cancers”
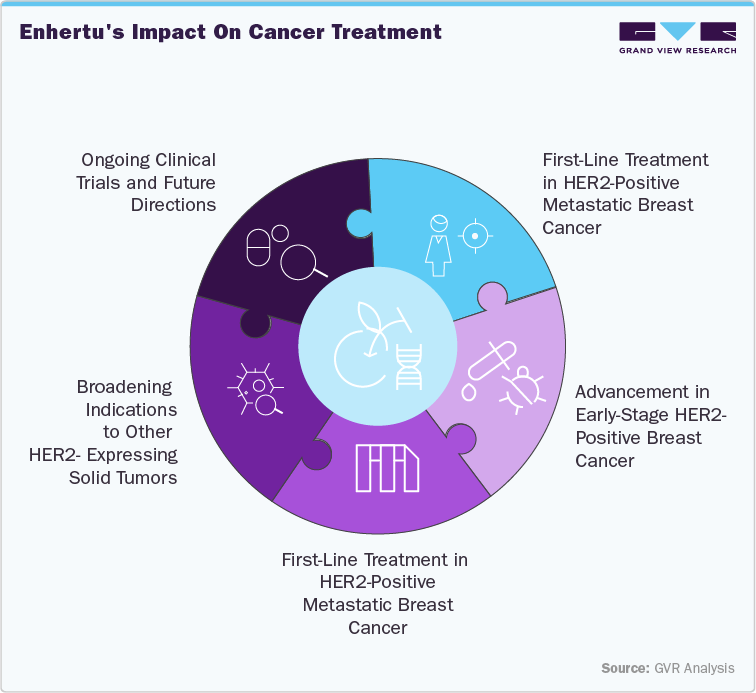
1. Expansion into HER2-Low and HER2-Ultralow Breast Cancer
Enhertu has demonstrated significant efficacy in patients with HER2-low (IHC 1+ or IHC 2+/ISH-) and HER2-ultralow (IHC 0) metastatic breast cancer. The DESTINY-Breast04 trial revealed a 36% reduction in the risk of disease progression or death with Enhertu compared to standard chemotherapy, with a median progression-free survival of 13.2 months versus 8.1 months, respectively. This led to FDA approval for patients who have received prior systemic therapy or developed disease recurrence during or within six months of completing adjuvant chemotherapy.
2. Advancement in Early-Stage HER2-Positive Breast Cancer
The DESTINY-Breast11 Phase III trial assessed Enhertu followed by taxane-based therapy (THP) in high-risk HER2-positive early-stage breast cancer. Preliminary results indicated a statistically significant and clinically meaningful improvement in pathologic complete response rates, suggesting a potential shift in treatment paradigms for this patient population
3. First-Line Treatment in HER2-Positive Metastatic Breast Cancer
The DESTINY-Breast09 trial evaluated Enhertu in combination with pertuzumab as a first-line treatment for HER2-positive metastatic breast cancer. The combination reduced the risk of disease progression or death by 44% compared to the standard THP regimen, highlighting its potential as a new first-line standard of care.
4. Broadening Indications to Other HER2-Expressing Solid Tumors
Enhertu has received accelerated FDA approval for treating various HER2-expressing solid tumors, including biliary tract, bladder, cervical, endometrial, ovarian, and pancreatic cancers. This broadening of indications underscores its versatility and potential to address unmet needs across multiple cancer types.
5. Ongoing Clinical Trials and Future Directions
Ongoing trials, such as the DESTINY-Breast09, continue to explore Enhertu's efficacy in earlier lines of therapy and in combination with other agents. These studies are crucial in establishing Enhertu's role in broader treatment regimens and its long-term impact on patient outcomes.
Overview of Alternative Therapeutics
Enhertu (trastuzumab deruxtecan) is a targeted therapy for HER2-expressing cancers, but several alternatives exist across antibody-drug conjugates, monoclonal antibodies, tyrosine kinase inhibitors, and chemotherapy regimens. Trastuzumab emtansine (Kadcyla) is an established ADC used after progression on trastuzumab and taxane therapy, while datopotamab deruxtecan is emerging for HER2-low cancers. Trastuzumab biosimilars such as Herzuma, Ogivri, and Ontruzant offer cost-effective HER2-targeted treatment but lack the cytotoxic payload of ADCs. Margetuximab (Margenza) is another monoclonal antibody used with chemotherapy for treated HER2-positive metastatic breast cancer. Tyrosine kinase inhibitors like tucatinib (Tukysa) are oral alternatives with notable activity in brain metastases when combined with trastuzumab and capecitabine. Chemotherapy-based regimens remain an option where targeted therapy is unsuitable. Choice of alternative depends on cancer type, HER2 expression, prior treatments, and patient condition, with clinical guidance essential to optimize outcomes.
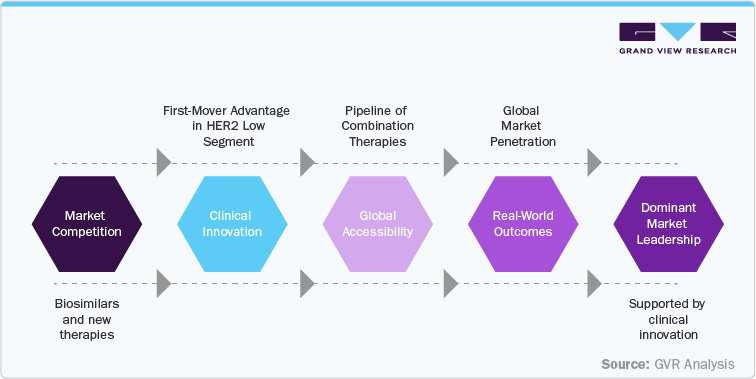
Competitive Landscape
The competitive landscape for Enhertu (trastuzumab deruxtecan) is defined by its position as a HER2-directed antibody-drug conjugate (ADC) targeting advanced breast cancer, HER2-mutant NSCLC, and HER2-positive gastric cancer. Jointly developed and commercialized by AstraZeneca and Daiichi Sankyo, Enhertu benefits from a broad clinical development program, including monotherapy and combination regimens, establishing it as a leading treatment in multiple HER2-positive and HER2-low settings.
In oncology, Enhertu faces competition from other HER2-targeted therapies such as trastuzumab (Herceptin), pertuzumab (Perjeta), T-DM1 (Kadcyla), and emerging ADCs and small molecules under development by competitors including Roche, Seagen, and Puma Biotechnology. While these competitors address similar patient populations, Enhertu’s differentiated mechanism-through DXd payload technology-provides potential for improved progression-free survival and activity in HER2-low expressing tumors.
Market dynamics are influenced by regional adoption patterns, reimbursement policies, and regulatory approvals. North America and Europe remain primary markets, driven by established oncology infrastructure, whereas Asia-Pacific represents a growing opportunity with increasing detection of HER2-positive cancers. Patent protection ensures exclusivity through the early-to-mid 2030s in major markets, but biosimilar ADCs and emerging HER2-targeted therapies are expected to intensify competition post-patent expiration.
Strategic differentiation for Enhertu relies on continued clinical validation, expansion into earlier treatment lines, and combination therapy approvals. The competitive environment underscores the need for market access strategies, physician engagement, and real-world evidence generation to maintain leadership. In summary, Enhertu holds a strong market position but faces evolving competitive pressures from established HER2 therapies and emerging next-generation ADCs.
North America Enhertu Market
North America remains a key market for Enhertu, primarily driven by the United States and Canada, where breast cancer prevalence is significant. The U.S. market is led by AstraZeneca and Daiichi Sankyo’s Enhertu, approved for HER2-positive, HER2-low, and HER2-ultralow metastatic breast cancer as well as HER2-mutant NSCLC and advanced gastric cancer. Strong adoption is supported by positive clinical trial outcomes, including DESTINY-Breast09. Patent protection will influence competition, and any biosimilars entering post-patent expiration could impact pricing and market share. Regulatory approval processes in the U.S. and Canada may delay competitor entry, providing short-term market protection. Continued investment in clinical trials and combination therapies is expected to further strengthen Enhertu’s position in this region.
Europe Enhertu Market
In Europe, Enhertu has gained traction across Germany, France, and the U.K., supported by approval for multiple HER2-targetable cancers. Market growth is driven by clinical adoption and demand for advanced oncology treatments. Patent expiration may lead to biosimilar competition, creating pricing pressure. Europe’s stringent regulatory environment could delay biosimilar entry, affecting the speed of market share shifts. Healthcare systems emphasizing cost-effectiveness may prioritize alternatives once available, influencing uptake and treatment accessibility. Growing emphasis on personalized oncology care is expected to support sustained demand.
Asia Pacific Enhertu Market
Asia Pacific presents growth opportunities, especially in China, Japan, and India. China may see earlier entry of competing therapies due to ongoing patent challenges, while India’s regulatory approvals will determine the pace of market access. Japan has high demand for oncology treatments, but strict regulatory barriers could slow biosimilar entry. The region offers significant growth potential but also faces challenges related to regulatory timelines and adoption patterns, with rising cancer awareness influencing uptake rates.
Latin America Enhertu Market
In Latin America, Brazil, Mexico, and Argentina represent key markets, with rising breast cancer prevalence driving demand. Cost considerations are central, and the eventual introduction of biosimilars post-patent expiration could increase affordability. Regulatory processes vary by country, impacting entry speed and market share shifts. Infrastructure and distribution challenges may influence adoption of new therapies, with government healthcare programs playing a key role.
Middle East and Africa Enhertu Market
The MEA region shows emerging uptake, with Saudi Arabia, UAE, and South Africa as leading markets. Rising cancer incidence is driving demand, though high costs limit access. Post-patent biosimilars will be critical for improving affordability. Regulatory efficiency varies, with some countries offering streamlined approval, while others may experience delays. Investment in healthcare infrastructure and access could expand market potential, supported by increasing oncology-focused initiatives.
Analyst Perspective
Enhertu’s market trajectory is shaped by robust clinical evidence, expanding indications, and strategic collaborations between AstraZeneca and Daiichi Sankyo. Strong uptake in North America and Europe reflects high unmet needs in HER2-driven cancers, while Asia Pacific and MEA offer significant growth potential amid rising cancer incidence. Patent protection currently limits competition, but biosimilar entry post-expiration will impact pricing and market share. Regional regulatory environments and healthcare infrastructure will influence adoption rates, with cost-effectiveness and patient access as key considerations. Continued innovation through combination therapies and earlier-line approvals will be critical to sustaining growth, positioning Enhertu as a cornerstone in the evolving oncology landscape.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price-Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy.
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028-2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU -15-30% Yr-1, deepening to -45-60% by Yr-3; U.S. -10-25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25-40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30-40% global revenue decline by Year-3 post-LOE.
-
Downside: 45-55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~-20-25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


