- Home
- »
- Market Trend Reports
- »
-
Givosiran (Givlaari) Market: Revenue Analysis, Clinical Adoption Trends, Reimbursement Landscape, And Competitive Benchmarking, 2025 - 2033
Report Overview
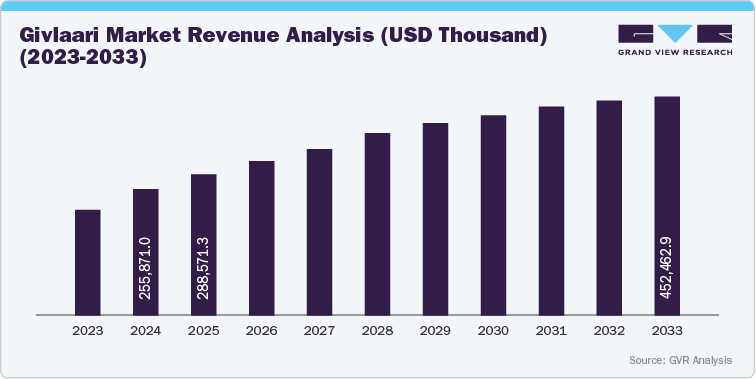
Givosiran (Givlaari) is a first-in-class RNA interference (RNAi) therapy developed by Alnylam Pharmaceuticals for the treatment of acute hepatic porphyria (AHP). The drug offers disease-modifying benefits by reducing aminolevulinic acid (ALA) and porphobilinogen (PBG) levels, resulting in a marked reduction in acute attack frequency and hospitalization rates. Market adoption is concentrated in specialty treatment centers and tertiary referral hospitals, reflecting its positioning within a rare disease market segment supported by precision medicine advancements. The commercial landscape is shaped by premium pricing, orphan drug exclusivity, and targeted reimbursement frameworks. Givlaari commands a high annual treatment cost, requiring payer engagement, value-based access agreements, and patient support programs to sustain uptake. Market penetration is strongest in North America and Western Europe, supported by robust diagnostic networks and early payer alignment on clinical value. I
Key Report Deliverables
-
Analyze the Givosiran (Givlaari) Market landscape, detailing the current market size, growth drivers, and key industry trends, particularly in light of the upcoming patent expiration and the impact of biosimilars entering the market.
-
Forecast Market Growth, projecting future trends for the Givosiran (Givlaari) Market, highlighting emerging opportunities within the biosimilar space, and assessing potential risks to growth as competition increases following patent expiry.
-
Identify Regulatory and Market Barriers, providing insights into regulatory and market barriers that could impact future market expansion and product development, with a specific focus on the challenges biosimilars may face in gaining approval and market access.
-
Concurrent Competitive Landscape, identifying key players in the Givosiran (Givlaari) Market, including both originator and biosimilar manufacturers. Examine their strategic moves, partnerships, and distribution of market share to understand competitive positioning and potential shifts as biosimilars are introduced.
-
Regulatory Barriers, identifying key regulatory challenges related to the entry of Givosiran (Givlaari) biosimilars, including approval processes and market access restrictions, and assessing their potential impact on the speed and scope of market expansion.
-
Strategic Implications, evaluating strategic moves for manufacturer and its competitors to maintain leadership in the market. This includes exploring innovation, differentiation, potential patient support programs, and geographic expansion strategies.
Patent Landscape and Exclusivity Outlook
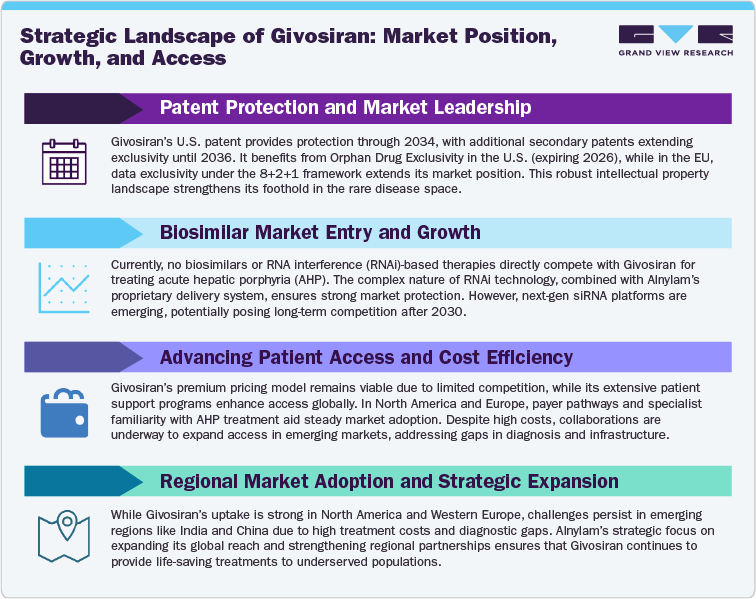
The patent landscape and exclusivity outlook for Givosiran (Givlaari) reflects a strong, multi-layered intellectual property strategy designed to protect long-term market leadership within the RNA interference (RNAi) therapeutics space. Developed by Alnylam Pharmaceuticals, Givosiran holds robust global patent coverage across its composition of matter, manufacturing processes, and therapeutic use, providing substantial barriers to generic or biosimilar entry. The core U.S. composition patent and related filings are projected to expire around 2030, forming the earliest potential point for market exclusivity loss under current patent protection. Additional secondary patents, including those covering formulation, dosage optimization, and delivery chemistry, extend protection in several jurisdictions, with some patent families reportedly active until 2036. Together, these patents create a layered exclusivity framework, effectively safeguarding Givosiran’s position in the rare disease therapeutics market well beyond the expiration of regulatory exclusivity.
From a regulatory standpoint, Givosiran benefits from Orphan Drug Exclusivity and New Chemical Entity (NCE) data exclusivity in the United States and European Union. The U.S. Orphan Drug Exclusivity protects the product until November 2026, while NCE exclusivity provides an additional safeguard against generic submissions for the same period. The European data exclusivity window aligns closely, granting protection under the standard 8+2+1 framework (data exclusivity, market protection, and potential extension). Currently, there are no biosimilar or follow-on RNAi competitors in development or approved for Givosiran, given the technical complexity of RNA-based drug manufacturing and regulatory uncertainty surrounding RNAi therapeutic replication. These factors, combined with Alnylam’s extensive platform patent portfolio and proprietary delivery technology (GalNAc-conjugate system), create a high entry barrier for potential challengers
Current Market Scenarios
The current market scenario for Givosiran (Givlaari) reflects a stable and strategically positioned growth phase within the rare disease therapeutics and RNA interference (RNAi) landscape. The therapy continues to demonstrate strong performance in treating acute hepatic porphyria (AHP), supported by robust real-world evidence, positive long-term safety data, and an expanding global diagnostic and treatment ecosystem. The drug’s orphan designation, premium pricing model, and limited competition have allowed Alnylam Pharmaceuticals to maintain consistent revenue growth and strong brand recognition in the genetic and metabolic disorder market.
In North America and Western Europe, adoption remains steady due to well-established specialty care networks, payer reimbursement pathways, and growing clinical familiarity among hepatologists and metabolic specialists. The availability of comprehensive patient support programs and value-based reimbursement agreements has further supported sustained market uptake. Conversely, in emerging regions, market access remains constrained by high treatment costs, limited specialist infrastructure, and diagnostic underpenetration, though ongoing collaborations with regional partners aim to improve accessibility.
Competitive activity in the segment remains limited. No direct biosimilars or RNAi-based competitors have entered clinical or commercial stages for acute hepatic porphyria till 2030, reinforcing Givosiran’s market exclusivity. However, the broader RNAi space is gaining momentum with several next-generation siRNA platforms under development, indicating potential long-term competition beyond 2030.
Strategically, Alnylam is focused on expanding geographic reach, enhancing diagnostic capabilities to identify undiagnosed AHP patients, and strengthening real-world data collection to support payer negotiations and regulatory renewals. The company’s continued investment in RNAi innovation, pipeline diversification, and global access programs positions Givosiran as a durable, high-value asset within Alnylam’s rare disease portfolio.
Overall, the Givosiran market is characterized by high clinical value, limited competition, and strong pricing power, supported by long-term patent protection and sustained global expansion initiatives. The product’s stability and specialized nature make it one of the most defensible assets in the orphan drug and RNAi therapeutics landscape
Market Dynamics

“Expanding Adoption of RNA Interference (RNAi) Therapeutics and Growing Diagnosis of Acute Hepatic Porphyria (AHP)”
The Givosiran (Givlaari) market is being propelled by the accelerating adoption of RNA interference (RNAi) therapeutics as a validated and high-impact treatment platform in rare genetic and metabolic disorders. Givosiran represents one of the most successful clinical applications of RNAi technology, providing a disease-modifying mechanism that directly targets the underlying genetic cause of acute hepatic porphyria (AHP). Its ability to significantly reduce the accumulation of neurotoxic intermediates such as aminolevulinic acid (ALA) and porphobilinogen (PBG) has established a new therapeutic benchmark in precision medicine.
Global recognition of AHP as a serious, underdiagnosed condition has also improved through increased clinical awareness, specialist training, and genetic screening initiatives. Healthcare systems in North America and Europe have integrated targeted diagnostic algorithms to identify AHP patients earlier, enabling timely treatment initiation and improved long-term outcomes. Supportive orphan drug policies, premium pricing frameworks, and favorable reimbursement pathways continue to sustain strong uptake in major markets. Furthermore, the growing body of real-world evidence confirming Givosiran’s efficacy and safety has enhanced physician confidence and payer acceptance, reinforcing its position as a cornerstone therapy in RNAi-based rare disease management.
“High Treatment Cost and Limited Accessibility in Emerging Markets”
Despite its clinical success, the commercial trajectory of Givosiran is constrained by high treatment costs and limited market access, particularly in developing economies. The annual cost of therapy, reflective of its orphan drug pricing structure, poses a significant challenge for healthcare systems with constrained budgets and limited reimbursement capacity. Even in mature markets, payer scrutiny around cost-effectiveness and long-term value outcomes is intensifying as rare disease spending grows as a proportion of overall pharmaceutical expenditure.
In emerging regions such as Asia-Pacific, Latin America, and parts of the Middle East, the adoption of Givosiran remains limited due to insufficient diagnostic infrastructure, scarcity of metabolic specialists, and low disease awareness. Many eligible patients remain undiagnosed or untreated because of gaps in genetic testing availability and specialist referral networks. To overcome these challenges, Alnylam and its partners are focusing on innovative pricing strategies, tiered market access models, and collaborations with local health authorities to expand reach. Establishing sustainable access programs, strengthening public-private partnerships, and leveraging real-world outcome data will be essential for improving affordability and accelerating patient access in high-potential but underserved markets.
“Geographic Expansion and Market Access Optimization”
The most significant opportunity for the Givosiran (Givlaari) market lies in geographic expansion and market access optimization, particularly across Asia-Pacific, Latin America, and the Middle East. These regions are witnessing rapid improvements in rare disease awareness, diagnostic capabilities, and specialty care infrastructure, creating a favorable environment for the introduction of advanced RNA interference (RNAi) therapies. As healthcare systems evolve and national rare disease frameworks mature, expanding Givosiran’s presence beyond mature markets such as North America and Europe represents a major growth avenue.
Alnylam can capitalize on this opportunity by adopting tiered pricing strategies, forming local partnerships, and engaging in early dialogue with regional regulators and payers to streamline reimbursement pathways. Establishing diagnostic support programs, training initiatives for metabolic specialists, and public-private collaborations will further enable patient identification and therapy access. This strategic expansion will not only drive incremental revenue but also strengthen Alnylam’s global leadership in RNAi therapeutics, positioning Givosiran as the benchmark therapy for acute hepatic porphyria (AHP) across both developed and emerging healthcare systems.
Trends: Increasing Integration of RNA Interference (RNAi) Therapeutics into Mainstream Treatment Paradigms, Expansion of Rare Disease Diagnosis and Patient Identification Programs, Growing Focus on Market Access, Affordability, and Value-Based Care Models are contributing to the market
-
Increasing Integration of RNA Interference (RNAi) Therapeutics into Mainstream Treatment Paradigms
The global acceptance of RNA interference (RNAi) technology as a validated therapeutic modality is reshaping the rare disease landscape. Givosiran (Givlaari), as one of the first commercialized RNAi therapies, has demonstrated the clinical viability and long-term safety of gene-silencing approaches. Healthcare systems and regulatory authorities are increasingly recognizing RNAi-based drugs as disease-modifying interventions, encouraging greater investment and adoption. The success of Givosiran has catalyzed further development of next-generation siRNA molecules with improved stability and delivery efficiency, reinforcing confidence in the platform’s scalability. As precision medicine gains momentum, RNAi therapeutics are being integrated into multidisciplinary treatment frameworks, enhancing patient outcomes and setting a new benchmark for targeted genetic therapies.
-
Expansion of Rare Disease Diagnosis and Patient Identification Programs
Advances in genetic screening, biomarker testing, and digital diagnostic tools are significantly improving the identification of acute hepatic porphyria (AHP) patients. Historically, AHP has been underdiagnosed due to its non-specific clinical presentation, but broader implementation of next-generation sequencing (NGS) and metabolic screening initiatives has increased diagnostic accuracy. Governments and healthcare organizations are establishing national rare disease registries and funding programs to support early diagnosis and specialist referrals. For Alnylam, this trend represents a major opportunity to expand the treatable patient population, improve treatment timelines, and enhance real-world evidence generation. Greater diagnostic clarity directly translates into improved therapy uptake and stronger payer justification for high-value orphan drugs like Givosiran.
-
Growing Focus on Market Access, Affordability, and Value-Based Care Models
As the orphan drug market matures, payers are demanding outcomes-driven pricing models to justify the high cost of therapies such as Givosiran. The shift toward value-based reimbursement emphasizes real-world performance metrics including reduced hospitalizations, improved quality of life, and long-term clinical stability. Pharmaceutical companies are increasingly engaging in risk-sharing agreements, managed access programs, and tiered pricing structures to balance affordability with commercial sustainability. Alnylam is adapting to this trend by strengthening its health economics and outcomes research (HEOR) framework and expanding patient access programs in underpenetrated regions. This strategic alignment with payer expectations is essential to maintain reimbursement continuity and ensure sustainable global adoption of Givosiran.
Overview of Alternative Therapeutics
The alternative therapeutics landscape for Givosiran (Givlaari) is characterized by a small but evolving field of rare disease treatments targeting acute hepatic porphyria (AHP) and related metabolic disorders. As of now, no direct RNA interference (RNAi) competitors are available, giving Givosiran a strong first-mover advantage. However, alternative approaches using enzyme replacement, substrate reduction, and gene therapy are gaining research traction and may reshape the competitive environment over the next decade.
The most relevant alternative treatment is hemin-based therapy, such as Panhematin (hemin for injection) and Normosang (human hemin), which have been standard of care for acute porphyria attacks. These therapies help replenish deficient heme pools, reducing neurotoxic precursor accumulation. However, their use is limited by intravenous administration, short duration of effect, and hospital-based management requirements, which reduce patient convenience and long-term efficacy. Givosiran’s subcutaneous dosing and sustained preventive benefits provide a clear clinical advantage over these legacy options.
Emerging research in gene therapy and mRNA-based therapeutics is introducing next-generation competition. Companies are exploring adeno-associated viral (AAV) gene delivery platforms to achieve permanent suppression of the ALAS1 enzyme pathway. If proven safe and effective, such candidates could offer curative potential, though they remain several years from commercialization.
Overall, the alternative therapeutic landscape remains fragmented and clinically limited, with Givosiran maintaining clear superiority in efficacy, dosing convenience, and preventive disease control. Over the medium term, the market is expected to remain monopolized by Givosiran, with disruptive competition likely emerging only from gene therapy entrants or next-generation RNAi platforms in the early to mid-2030s.
Competitive Landscape
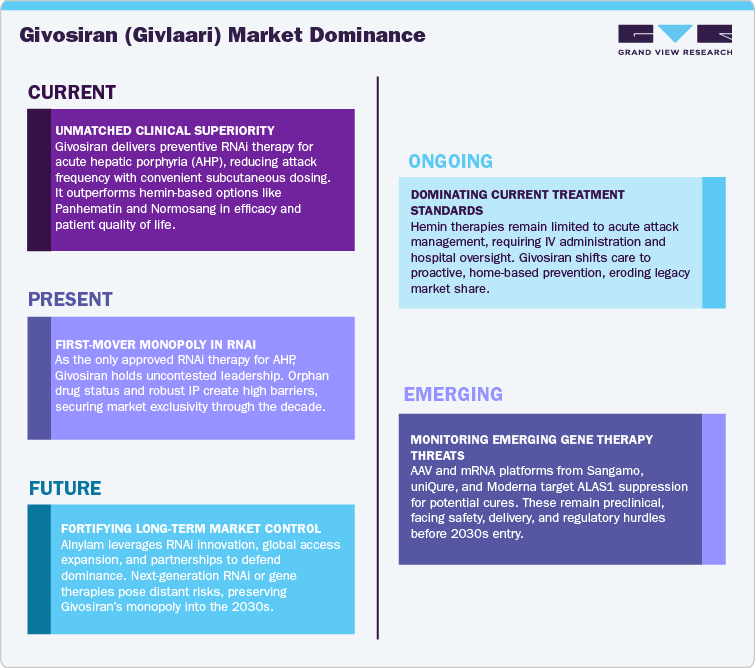
The competitive landscape for Givosiran (Givlaari) remains highly consolidated and innovation-driven, with Alnylam Pharmaceuticals holding an uncontested leadership position in the acute hepatic porphyria (AHP) treatment space. Givosiran is the only approved RNA interference (RNAi) therapy for AHP, providing it with a distinct first-mover advantage and long-term commercial defensibility. Its orphan drug designation, patent exclusivity, and data protection across major regions have created strong entry barriers, limiting direct competition.
Current market competition is largely indirect, coming from hemin-based therapies such as Panhematin (Recordati Rare Diseases) and Normosang (Orphan Europe), which are used for acute management of porphyria attacks rather than chronic prevention. These older agents maintain presence in hospital settings but are gradually losing market share due to Givosiran’s superior clinical outcomes, easier subcutaneous administration, and reduced attack frequency. Givosiran’s ability to deliver disease-modifying benefits and improve quality of life has shifted treatment preference toward preventive, RNAi-based care models.
Emerging competition is expected to arise from gene therapy developers such as Sangamo Therapeutics, uniQure, and Moderna, which are exploring AAV-based and mRNA-based platforms for long-term or potentially curative interventions. However, these programs remain at early research or preclinical stages, and their eventual commercialization faces substantial challenges in delivery optimization, safety validation, and regulatory acceptance. In the broader rare disease and RNAi space, Alnylam’s leadership is being complemented by the emergence of Ionis Pharmaceuticals, Silence Therapeutics, and Arrowhead Pharmaceuticals, all developing targeted RNA-based therapies in adjacent Treatment Types. Nonetheless, none currently compete directly within the AHP segment.
Overall, Givosiran operates in a low-competition, high-barrier market, supported by strong intellectual property protection, specialty market control, and a differentiated therapeutic profile. Over the next decade, Alnylam’s sustained investment in RNAi innovation, global access expansion, and strategic partnerships will be critical to defending market exclusivity ahead of potential gene therapy entrants or next-generation RNAi challengers anticipated post-2030.
Regional Analysis
North America Givosiran (Givlaari) Market
North America serves as the primary commercial hub for Givosiran (Givlaari), accounting for the majority of global revenue. The United States is the dominant market, driven by advanced diagnostic infrastructure, specialized treatment centers, and high payer acceptance of orphan drug pricing models. The strong presence of metabolic disease specialists and patient advocacy organizations has supported early diagnosis and treatment adoption. Reimbursement coverage under both public and private health plans remains favorable, supported by value-based access agreements and demonstrated reductions in hospitalizations and attack frequency. Canada has shown moderate but consistent uptake, supported by regional funding programs and emerging rare disease frameworks. Continued real-world evidence generation and health economics studies are essential to reinforce payer confidence and sustain market access amid growing scrutiny on specialty drug budgets. The strategic focus in North America will remain on expanding diagnostic networks, strengthening physician education, and maintaining long-term payer alignment to defend market share against potential RNAi or gene therapy entrants post-2030.
Europe Givosiran (Givlaari) Market
The European market presents a diverse and highly regulated environment, with market access governed by national health technology assessment (HTA) processes and budget impact evaluations. Western European countries such as Germany, the U.K., France, and Italy demonstrate solid adoption due to well-established reimbursement systems and rare disease programs. However, Central and Eastern Europe remain underpenetrated due to cost containment policies, infrastructure gaps, and delayed diagnostic adoption. Pricing negotiations are typically lengthy, requiring strong pharmacoeconomic data and real-world evidence to justify reimbursement. The region’s emphasis on cost-effectiveness necessitates ongoing collaboration with national payers, academic hospitals, and patient advocacy groups to highlight Givosiran’s long-term benefits. Strategic opportunities exist in expanding specialist referral networks, creating pan-European patient registries, and supporting local education initiatives to enhance early detection of acute hepatic porphyria (AHP).
Asia Pacific Givosiran (Givlaari) Market
The Asia-Pacific (APAC) region represents a high-growth frontier for Givosiran, driven by the rapid expansion of genetic testing, specialist training, and healthcare modernization. Japan is the most advanced market, with strong regulatory support for RNAi therapeutics and inclusion in the country’s rare disease treatment framework. Australia and South Korea are following similar trajectories, with well-structured access pathways and early adoption in tertiary care centers. Conversely, large population markets such as China, India, and Indonesia remain at an early stage of market development due to limited diagnostic infrastructure and high out-of-pocket healthcare costs. Alnylam’s strategic approach in the region focuses on tiered pricing models, public-private partnerships, and training collaborations to expand clinical capacity. Increasing regional awareness through medical education and diagnostic screening programs will be key to unlocking patient potential. The long-term opportunity in Asia-Pacific lies in disease awareness expansion, policy advocacy, and integration into regional rare disease frameworks.
Latin America Givosiran (Givlaari) Market
In Latin America, Givosiran operates within a fragmented healthcare landscape, marked by varying reimbursement models and limited funding for high-cost orphan drugs. Brazil and Mexico represent the most promising markets, with growing networks of metabolic specialists and progressive rare disease policies that support market entry. Uptake in smaller markets such as Argentina, Chile, and Colombia is slower due to economic instability, restricted public budgets, and limited awareness among clinicians. Successful penetration requires a multi-channel strategy centered on stakeholder education, early diagnostic initiatives, and access partnerships with public health institutions. Alnylam’s focus in the region should emphasize cost-sharing mechanisms, patient assistance programs, and demonstration of clinical value through reduced hospitalization and improved quality of life metrics. Establishing regional centers of excellence and collaborating with international rare disease organizations can further enhance patient identification and therapy adoption.
Middle East and Africa Givosiran (Givlaari) Market
The Middle East and Africa region remains underdeveloped for Givosiran but offers long-term potential as healthcare infrastructure improves. Adoption is currently concentrated in Gulf Cooperation Council (GCC) countries such as Saudi Arabia, the United Arab Emirates, and Qatar, where advanced tertiary care centers, higher per capita healthcare spending, and established rare disease funding programs support access to innovative biologics. North African countries such as Egypt and Morocco are gradually emerging, aided by expanding healthcare reforms and interest in international pharmaceutical partnerships. However, sub-Saharan Africa continues to face significant barriers including diagnostic limitations, scarcity of metabolic specialists, and reimbursement constraints. To capture growth in this region, Alnylam should prioritize capacity building, training programs for clinicians, and collaborations with government health ministries to introduce pilot access schemes. Over time, continued investment in infrastructure development, disease awareness, and strategic partnerships will enable Givosiran to establish a presence in this nascent but evolving therapeutic landscape.
Analyst Perspective
The Givosiran (Givlaari) market remains strategically strong, supported by its first-in-class RNA interference (RNAi) technology, proven clinical efficacy, and long-term safety data. The therapy’s ability to reduce porphyria attack frequency and improve quality of life secures its position as the standard of care in acute hepatic porphyria (AHP).
Growth in North America and Europe will remain steady through sustained reimbursement support and real-world data generation, while emerging markets such as Asia-Pacific and Latin America present significant expansion opportunities through tiered pricing and access partnerships.
Competition remains limited, with no biosimilars or RNAi rivals currently in the pipeline. However, the rise of gene therapy and next-generation RNAi platforms could reshape the market post-2030.
Overall, Givosiran’s outlook is favorable and defensible, with success hinging on continued payer engagement, regional expansion, and innovation in RNAi therapeutics to sustain leadership and long-term value creation.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028-2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU -15-30% Yr-1, deepening to -45-60% by Yr-3; U.S. -10-25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25-40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30-40% global revenue decline by Year-3 post-LOE.
-
Downside: 45-55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~-20-25% with strong SC adoption.
-
- Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


