- Home
- »
- Market Trend Reports
- »
-
Ibalizumab (Trogarzo) Market: Patent Landscape, Competitive Dynamics, And Growth Outlook
Report Overview
The Ibalizumab (Trogarzo) market represents a niche but critical segment of the global HIV therapeutics industry, targeting multidrug-resistant (MDR) HIV-1 infections in heavily treatment-experienced patients. As a CD4-directed post-attachment HIV-1 inhibitor, Trogarzo delivers a novel mechanism of action distinct from conventional antiretroviral therapies, making it essential for patients with limited treatment options. Developed by TaiMed Biologics and commercialized by Theratechnologies, the drug has held FDA approval since 2018 and maintains strong presence in North America and Europe. Market growth is supported by unmet medical needs, premium biologic pricing, and the expansion of specialty infusion-based HIV treatment centers. However, uptake remains constrained by the small target population, intravenous administration, and competition from newer entrants like Fostemsavir (Rukobia) and Lenacapavir (Sunlenca). Despite these challenges, Trogarzo continues to sustain steady revenue within the high-value biologics market, with potential biosimilar entry expected post 2030.
Key Report Deliverables
-
Analyze the Ibalizumab (Trogarzo) market landscape, detailing the current market size, growth drivers, and key industry trends, particularly in light of the upcoming patent expiration and the impact of biosimilars entering the market.
-
Forecast Market Growth, projecting future trends for the Ibalizumab (Trogarzo) market, highlighting emerging opportunities within the biosimilar space, and assessing potential risks to growth as competition increases following patent expiry.
-
Identify Regulatory and Market Barriers, providing insights into regulatory and market barriers that could impact future market expansion and product development, with a specific focus on the challenges biosimilars may face in gaining approval and market access.
-
Concurrent Competitive Landscape, identifying key players in the Ibalizumab (Trogarzo) market, including both originator and biosimilar manufacturers. Examine their strategic moves, partnerships, and distribution of market share to understand competitive positioning and potential shifts as biosimilars are introduced.
-
Regulatory Barriers, identifying key regulatory challenges related to the entry of Ibalizumab (Trogarzo) biosimilars, including approval processes and market access restrictions, and assessing their potential impact on the speed and scope of market expansion.
-
Strategic Implications, evaluating strategic moves for Janssen Biotech and its competitors to maintain leadership in the Aflibercept market. This includes exploring innovation, differentiation, potential patient support programs, and geographic expansion strategies.
Patent Landscape and Exclusivity Outlook
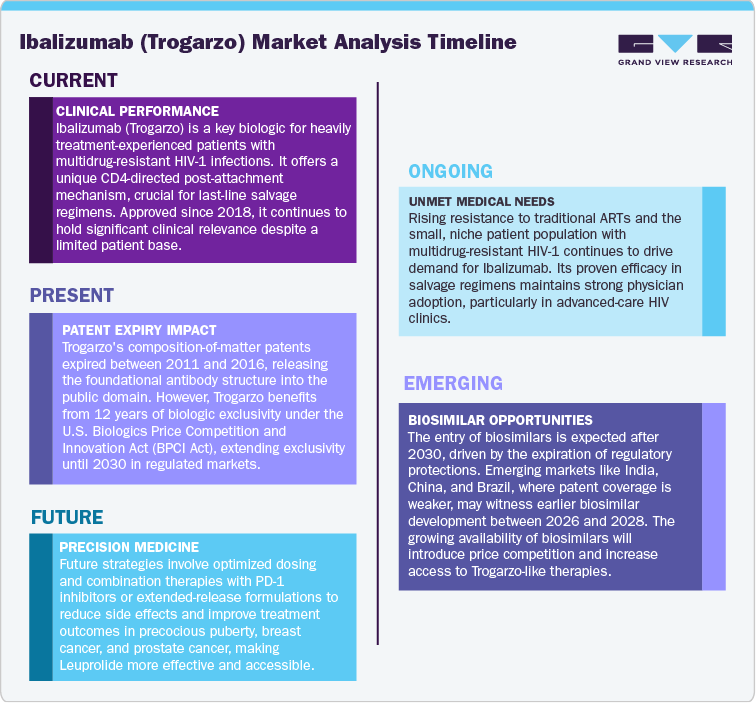
The patent landscape for Ibalizumab (Trogarzo) reflects the typical progression of a biologic product that transitioned from early research to niche commercialization. The core composition-of-matter patents, covering the antibody structure and its complementary determining regions (CDRs), were initially filed in the early 1990s and expired between 2011 and 2016 across major jurisdictions including the United States, Europe, Canada, and Australia. This expiry effectively released the foundational antibody structure into the public domain, eliminating the primary intellectual property barriers around the molecule itself.
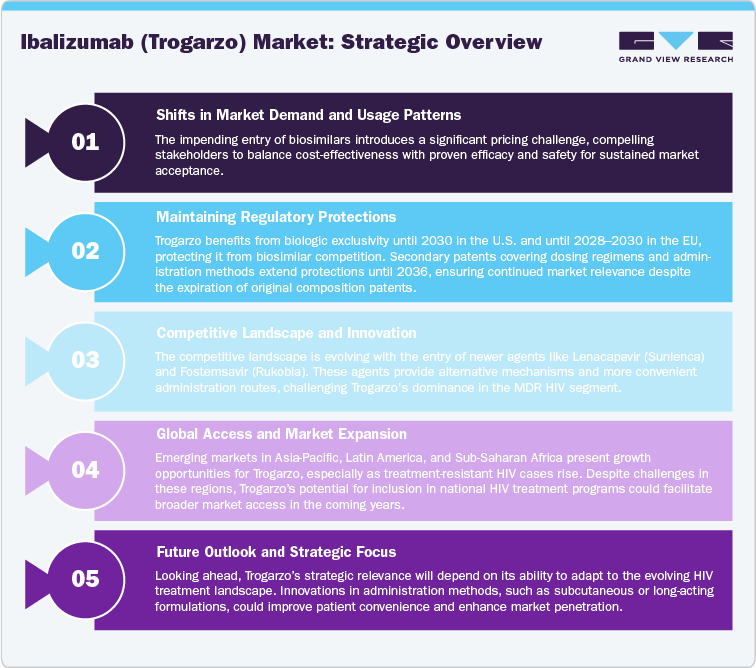
Despite the expiration of the original patents, regulatory exclusivity now serves as the primary protection mechanism. Under the U.S. Biologics Price Competition and Innovation Act (BPCI Act), Trogarzo benefits from 12 years of biologic exclusivity, protecting it from biosimilar competition until March 2030. Similarly, in the European Union, data and market exclusivity are expected to extend until 2028–2030, depending on national extensions.
Beyond these regulatory protections, secondary patents covering formulations, dosing regimens, and administration methods have been filed, with at least one U.S. patent extending until November 2036. Additional filings listed in the Medicines Patent Pool (MPP) indicate certain method-of-use or process patents potentially valid until 2040, though these are narrower in scope and jurisdictionally dependent.
In practical terms, biosimilar entry is not anticipated before 2030 in regulated markets such as the U.S. and EU. Emerging markets like India, China, and Brazil, where patent coverage has lapsed or was never filed, may witness earlier biosimilar development between 2026 and 2028. Overall, the patent expiry pattern positions Trogarzo as a protected biologic asset through 2030, with extended defensive IP layers offering incremental exclusivity until the mid-2030s.
Current Market Scenarios
The Ibalizumab (Trogarzo) market in 2025 represents a highly specialized segment of the HIV therapeutics landscape, addressing patients with multidrug-resistant (MDR) HIV-1 infection who have failed multiple prior treatments. As the only CD4-directed post-attachment inhibitor currently approved, Trogarzo continues to hold strong clinical importance in salvage therapy regimens. The drug’s unique mechanism of action differentiates it from conventional antiretrovirals, positioning it as a last-line therapeutic option in combination treatment settings.
The current market environment is defined by stable clinical adoption and limited competition within its specific niche. However, growth is naturally constrained by the small patient population and the complex infusion-based administration process that limits broader accessibility. The introduction of the IV push formulation has improved treatment convenience, reduced infusion time and expanding potential use across outpatient and specialty HIV clinics.
From a competitive perspective, Trogarzo faces emerging challenges from newer agents such as Lenacapavir (Sunlenca) and Fostemsavir (Rukobia), which offer alternative mechanisms and more convenient administration routes. Despite this, Trogarzo retains strategic value as a biologic therapy for multidrug-resistant cases, supported by established physician awareness and strong clinical data. The current market scenario reflects a mature yet sustainable position, with the drug expected to maintain steady relevance in advanced HIV treatment protocols through the end of its exclusivity period.

Market Dynamics
“Growing Clinical Need for Advanced Multidrug-Resistant HIV Therapies”
The Ibalizumab (Trogarzo) market is propelled by the rising prevalence of multidrug-resistant (MDR) HIV-1 infections and the growing demand for innovative biologics that can address treatment failure in heavily pretreated patients. Unlike conventional antiretrovirals, Ibalizumab acts as a CD4-directed post-attachment inhibitor, blocking viral entry while preserving immune function - a mechanism that remains unmatched in current HIV therapy portfolios. Its inclusion in global HIV treatment guidelines reinforces its clinical indispensability and supports continued physician confidence in its use for complex, late-line cases.
The introduction of the IV push formulation has been a major market catalyst, reducing infusion time and improving patient and clinician convenience. This innovation enhances the drug’s usability across specialty HIV centers and outpatient infusion clinics, expanding its reach within advanced-care networks. Additionally, the global increase in treatment-resistant HIV cases, combined with greater awareness among infectious disease specialists, sustains consistent prescription demand.
From a strategic standpoint, Trogarzo benefits from premium biologic pricing, strong payer recognition, and a favorable safety profile compared to conventional combination therapies. Continued investments in HIV resistance surveillance programs and real-world evidence studies are expected to reinforce its value proposition. These dynamics, along with supportive reimbursement mechanisms in developed markets, position Ibalizumab as a cornerstone therapy for salvage treatment, driving steady demand and long-term clinical relevance in the evolving global HIV care landscape.
“Limited Patient Base and Competitive Therapeutic Pressures”
Despite its clinical importance, the Ibalizumab (Trogarzo) market faces significant constraints that limit its scalability and global adoption. The most pronounced restraint is its narrow target population - patients with multidrug-resistant HIV-1 represent a very small fraction of the total HIV population, naturally restricting commercial expansion potential. Moreover, the intravenous mode of administration presents logistical barriers, requiring trained professionals, infusion setups, and cold-chain storage, which limit accessibility in resource-constrained healthcare systems.
Even with the availability of the IV push dosing format, the therapy remains less convenient than oral or long-acting injectable alternatives, leading to lower patient preference in markets with advanced treatment options. High cost of treatment, coupled with payer authorization challenges, further affects uptake, especially in regions where healthcare budgets are tightly managed.
The competitive landscape is also tightening as newer next-generation entry inhibitors, notably Lenacapavir (Sunlenca) and Fostemsavir (Rukobia), offer simpler dosing schedules and improved patient compliance. Additionally, Theratechnologies’ strategic withdrawal from the European market reduces international exposure, confining revenue dependence largely to North America. Over the longer term, patent expirations and potential biosimilar development post-2030 are expected to introduce pricing pressures and diminish exclusivity advantages.
“Pipeline Expansion and Emerging Market Penetration Potential”
The Ibalizumab (Trogarzo) market holds notable opportunities driven by therapeutic innovation, formulation enhancement, and geographic expansion. As global ART resistance rates continue to rise, Trogarzo’s distinct CD4 post-attachment inhibition mechanism offers long-term potential to serve as part of combination salvage regimens in resistant HIV management. Collaboration with research institutions and pharmaceutical partners could expand its role in dual-mechanism or multi-drug salvage combinations, enhancing its clinical utility beyond its current scope.
A significant opportunity lies in administration optimization and patient accessibility. The FDA-approved IV push format already reduces infusion burden; however, future development of subcutaneous or long-acting formulations could transform Trogarzo into a community-level therapy suitable for decentralized HIV care models. These innovations would align with global health system trends emphasizing home-based or outpatient biologic delivery, enhancing market penetration.
Emerging markets present another untapped frontier. Countries in Asia-Pacific, Latin America, and Sub-Saharan Africa, where MDR HIV incidence is rising, offer potential for biosimilar partnerships, technology transfer, or tiered pricing strategies post-2030. Additionally, inclusion in national HIV treatment programs, World Health Organization (WHO) initiatives, and public-private collaborations could drive access expansion while reinforcing brand equity.
“Shift Toward Long-Acting and Simplified HIV Therapies, Rising Focus on Real-World Evidence and Outcomes-Based Adoption, Repurposing and Emergency Use in Emerging Viral Threats contributing to the market
- Shift Toward Long-Acting and Simplified HIV Therapies
A major trend shaping the Ibalizumab (Trogarzo) market is the industry-wide transition toward long-acting and simplified antiretroviral therapies. Patients and providers increasingly prefer less frequent dosing regimens that enhance adherence and reduce clinic visits. This trend has intensified with the introduction of Lenacapavir (Sunlenca) and Cabotegravir/Rilpivirine injectables, which compete for attention in the advanced HIV segment. While Trogarzo remains unique as a CD4 post-attachment inhibitor, it must adapt to this market evolution by pursuing alternative administration routes such as subcutaneous or depot formulations. The move toward long-acting biologics underscores a clear market demand for convenience, cost-efficiency, and patient-centric care, influencing future innovation and positioning strategies for Trogarzo.
- Rising Focus on Real-World Evidence and Outcomes-Based Adoption
The growing emphasis on real-world data (RWD) to validate treatment effectiveness and justify reimbursement. Healthcare payers and providers increasingly rely on real-world evidence (RWE) to assess long-term patient outcomes, resistance management, and treatment durability in MDR HIV. For Trogarzo, post-marketing surveillance and registry studies are becoming pivotal tools to demonstrate clinical value beyond trial settings. Companies that effectively utilize RWE to highlight improved quality of life, viral suppression stability, and cost-effectiveness can secure stronger payer positioning. This trend reinforces the importance of data-driven storytelling and evidence-based market access strategies in sustaining Trogarzo’s relevance.
- Expansion into Emerging and Untapped HIV Markets
The globalization of advanced HIV care presents a growing opportunity for Trogarzo in emerging markets, particularly in Asia-Pacific, Latin America, and Sub-Saharan Africa. As ART-resistant HIV strains become more prevalent, demand for non-traditional biologic therapies is rising. Expanding access through biosimilar collaborations, technology transfer, and public health partnerships can enable Trogarzo to penetrate new territories. The focus is gradually shifting from high-income regions to middle-income countries with expanding healthcare infrastructure and improved biologics regulation. This geographic diversification aligns with global HIV response programs and ensures that Trogarzo maintains long-term strategic relevance beyond its current North American dominance.
Overview of Alternative Therapeutics
The alternative therapeutics landscape for multidrug-resistant (MDR) HIV-1 infection has expanded significantly over the past five years, introducing novel mechanisms and more convenient dosing options that complement or compete with Ibalizumab (Trogarzo). Among these, Lenacapavir (Sunlenca, Gilead Sciences) and Fostemsavir (Rukobia, ViiV Healthcare) represent the most clinically relevant alternatives, each addressing distinct aspects of advanced HIV management.
Lenacapavir, a first-in-class capsid inhibitor, offers a breakthrough long-acting subcutaneous injection administered every six months. It provides potent antiviral activity with minimal cross-resistance, making it particularly attractive for treatment-experienced patients. Its biannual dosing schedule and favorable resistance profile position as a direct competitor to Trogarzo’s biweekly intravenous regimen, especially in outpatient and decentralized care settings.
Fostemsavir, on the other hand, is an oral gp120 attachment inhibitor designed for patients with limited therapeutic options. By preventing viral attachment to CD4 receptors, it serves as a mechanistically complementary option to Ibalizumab, often used in combination salvage regimens. Its oral formulation enhances accessibility and adherence, creating an advantage in patient convenience.
Additional pipeline agents, including Islatravir (Merck) and Broadly Neutralizing Antibodies (bNAbs) like VRC07-523LS, indicate a growing shift toward multi-mechanism and long-acting biologics. Collectively, these alternatives are reshaping the competitive landscape by prioritizing durability, simplified administration, and improved quality of life for patients with resistant HIV. As the therapeutic paradigm evolves, Ibalizumab’s differentiation will increasingly depend on its clinical positioning, combination use, and next-generation formulation development.
Competitive Landscape
The competitive landscape for Ibalizumab (Trogarzo) is characterized by a mix of specialty biologics, small-molecule antiretrovirals, and emerging long-acting therapies that target multidrug-resistant (MDR) HIV-1 and treatment-experienced patient populations. While Trogarzo remains the only CD4-directed post-attachment inhibitor on the market, its competition stems from drugs offering novel mechanisms of action, simplified dosing, and enhanced patient adherence, all of which influence prescriber preferences and market positioning.
Lenacapavir (Sunlenca®, Gilead Sciences) currently stands as Trogarzo’s strongest competitor. As a first-in-class capsid inhibitor, Lenacapavir is administered subcutaneously every six months, representing a major advancement in treatment convenience and durability. Its approval for heavily treatment-experienced patients provides a direct alternative to Ibalizumab, offering comparable viral suppression rates with a more patient-friendly administration route. Gilead’s extensive distribution network and investment in long-acting HIV solutions further strengthen its market reach and competitive leverage.
Fostemsavir (Rukobia®, ViiV Healthcare) serves as another key rival in the MDR segment. This oral gp120 attachment inhibitor targets viral entry through a distinct mechanism, allowing for daily oral administration. Fostemsavir’s inclusion in treatment guidelines and its compatibility with multiple combination regimens enhance its physician adoption, especially for patients unwilling or unable to receive intravenous therapy.
Beyond these approved products, the pipeline includes Islatravir (Merck), an investigational nucleoside reverse transcriptase translocation inhibitor (NRTTI) being developed for both combination and long-acting formulations, and Broadly Neutralizing Antibodies (bNAbs) such as VRC07-523LS, which are under investigation for both treatment and prophylaxis.
Overall, the Ibalizumab market environment is evolving toward long-acting, patient-centric therapies, with competition driven by efficacy, dosing convenience, and combination flexibility. To sustain its relevance, Trogarzo’s strategic differentiation will depend on formulation innovation, real-world evidence generation, and potential partnerships to explore combination biologic regimens that reinforce its positioning within the advanced HIV treatment continuum.
Regional Analysis
North America Ibalizumab (Trogarzo) Market
North America remains the largest and most mature market for Ibalizumab (Trogarzo), driven by high treatment awareness, strong reimbursement systems, and the concentration of specialized HIV treatment centers. The United States accounts for the majority of global prescriptions, supported by favorable coverage through Medicare, Medicaid, and private insurers. The FDA’s 2018 approval established a solid regulatory foundation, with the IV push administration update further improving clinical adoption among infectious disease specialists. Growing awareness of multidrug-resistant (MDR) HIV-1 among clinicians, coupled with well-established HIV surveillance programs, ensures sustained demand within this segment. Canada also contributes to steady growth, although market access is limited by smaller patient numbers and cost-containment measures. The region benefits from the presence of Theratechnologies’ core commercial operations, active patient support programs, and an extensive distribution network. However, the entry of competitors such as Lenacapavir (Sunlenca) and Fostemsavir (Rukobia) is intensifying market competition, particularly among advanced-line therapies. Despite these dynamics, North America will remain the anchor market for Trogarzo through 2030, driven by high biologic adoption rates, strong payer infrastructure, and continued emphasis on innovation in advanced HIV care.
Europe Ibalizumab (Trogarzo) Market
The European market for Ibalizumab (Trogarzo) is relatively limited, largely due to restricted commercial activity and a narrow patient pool. While Trogarzo received European Commission approval in 2019, Theratechnologies has since scaled back operations in the region, citing high distribution costs and limited reimbursement coverage. The European HIV landscape is dominated by well-established ART regimens, and resistance rates remain comparatively lower than in other regions, which reduces immediate demand for advanced biologics. Key markets such as Germany, France, and the UK have shown interest in Trogarzo for specialty use within tertiary hospitals, but adoption is hampered by stringent health technology assessments (HTAs) and budgetary controls under national healthcare systems. Furthermore, increasing access to oral alternatives such as Fostemsavir and the expanding use of long-acting injectables like Cabotegravir/Rilpivirine present competitive challenges. Nonetheless, Europe remains strategically important for regulatory benchmarking and potential biosimilar partnerships post-2030. The future outlook depends on policy reforms that enhance biologic accessibility and the potential re-entry of Trogarzo through regional licensing or collaboration models.
Asia Pacific Ibalizumab (Trogarzo) Market
The Asia Pacific region offers emerging potential for the Ibalizumab (Trogarzo) market, driven by rising HIV prevalence, expanding healthcare infrastructure, and growing recognition of treatment-resistant infections. Countries such as China, Japan, South Korea, and Australia represent key focal points, with Japan having approved Trogarzo under its accelerated pathway for advanced HIV therapy. China and India are increasingly significant due to their large patient populations and rising incidence of MDR HIV cases, although accessibility remains limited by high biologic costs and regulatory constraints. Expanding investment in HIV diagnostic programs, international funding partnerships, and government-supported ART access initiatives create long-term opportunities for Trogarzo’s entry. Moreover, as the patent landscape opens post-2026, there is potential for biosimilar development or technology transfer collaborations, particularly with regional pharmaceutical firms. The market is expected to grow gradually as awareness of biologic-based HIV management increases and healthcare systems shift toward specialized infectious disease treatment frameworks. Over the next decade, Asia Pacific is projected to become a key growth frontier, with local partnerships and pricing strategies serving as critical enablers of market penetration.
Latin America Ibalizumab (Trogarzo) Market
In Latin America, the Ibalizumab (Trogarzo) market is in a nascent stage but shows growing promise due to increasing antiretroviral resistance rates and public health investments in advanced HIV management. Countries such as Brazil, Mexico, Chile, and Argentina are leading regional adoption efforts, supported by government-funded HIV treatment programs and collaborations with international donors. Brazil’s universal ART program presents a strong foundation for integrating novel biologics like Trogarzo, especially for treatment-experienced patients. However, widespread access remains limited by pricing challenges, import dependency, and procurement complexities across public healthcare systems. The Pan American Health Organization (PAHO) continues to facilitate regional cooperation for improved biologic availability, which could aid Trogarzo’s market expansion post-patent expiry. The region’s growing middle-income population, combined with improved clinical infrastructure and diagnostic capabilities, supports gradual uptake in tertiary hospitals. Long-term growth will depend on partnerships with local distributors, tiered pricing models, and regional regulatory harmonization. Overall, Latin America represents a strategic secondary market with high unmet need potential that can be unlocked through targeted access strategies and inclusion in national HIV resistance programs.
Middle East and Africa Ibalizumab (Trogarzo) Market
The Middle East and Africa region remains largely untapped for Ibalizumab (Trogarzo) but offers long-term potential driven by rising HIV incidence and gaps in multidrug-resistant treatment availability. Sub-Saharan Africa, which bears the highest global HIV burden, faces limited access to advanced biologics due to cost barriers, infrastructure deficits, and prioritization of first-line antiretroviral therapy (ART). However, as MDR HIV cases slowly increase and treatment failure rates become more prominent, there is a growing need for specialized rescue therapies like Trogarzo. Middle Eastern markets such as Saudi Arabia, United Arab Emirates, and South Africa present relatively better infrastructure and regulatory environments conducive to biologic introduction. International collaborations with the World Health Organization (WHO), PEPFAR, and Global Fund could facilitate subsidized access programs for eligible patients. The long-term regional strategy will depend on cost-effective pricing models, technology transfer, and biosimilar partnerships post-2030. Although the market remains at an early stage, rising government focus on infectious disease innovation and improving biologic manufacturing capabilities could make the Middle East and Africa a viable growth region for Trogarzo in the next decade.
Analyst Perspective
The Ibalizumab (Trogarzo) market occupies a strategically significant yet highly specialized position within the global HIV treatment ecosystem. Targeting multidrug-resistant (MDR) HIV-1 patients, the therapy’s differentiated CD4 post-attachment mechanism and proven clinical efficacy reinforce its role as a critical salvage-line option for treatment-experienced populations.
North America continues to serve as the core commercial hub, supported by robust reimbursement frameworks, established specialty networks, and clinical familiarity among infectious disease specialists. However, broader market scalability remains limited due to high treatment costs, intravenous administration requirements, and restricted access in cost-sensitive regions.
Competitive pressure from next-generation long-acting therapies such as Lenacapavir (Sunlenca) and Fostemsavir (Rukobia) is reshaping prescriber preferences toward more convenient regimens. To maintain its strategic relevance, Ibalizumab will need to leverage lifecycle management initiatives, including subcutaneous or long-acting formulations, and pursue regional partnerships to enhance global accessibility.
With regulatory exclusivity extending through 2030 and increasing clinical focus on resistant HIV cases, Trogarzo is expected to retain its clinical indispensability while operating within a commercially contained but stable niche of the advanced HIV biologics market.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028–2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU -15–30% Yr-1, deepening to -45–60% by Yr-3; U.S. -10–25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25–40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30–40% global revenue decline by Year-3 post-LOE.
-
Downside: 45–55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~-20–25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


