- Home
- »
- Market Trend Reports
- »
-
Insulin Glargine Market Analysis: Impact Of Biosimilars And Patent Expiry On Global Market Dynamics
Report Overview
Insulin glargine, marketed under the brand names Lantus and Toujeo by Sanofi, has been a key player in the management of diabetes for many years. Lantus, a long-acting insulin, has been widely used for the treatment of type 1 and type 2 diabetes, helping patients maintain stable blood glucose levels over an extended period. Toujeo, a more concentrated formulation of insulin glargine, offers a longer duration of action, catering to patients with higher insulin requirements or those who need more consistent insulin coverage. With the expiration of Lantus' patent, the market is experiencing significant shifts, particularly with the emergence of biosimilars such as Semglee, which present cost-effective alternatives to the original product. This change introduces competitive pressures that are reshaping the market dynamics, particularly in terms of pricing, market share, and treatment protocols. The increasing availability of biosimilars is driving adjustments in both clinical practices and pricing strategies, influencing the future landscape of insulin glargine therapy.
Key Report Deliverables
-
Market Landscape Analysis, assessing the insulin glargine market, focusing on size, growth drivers, and trends in light of Lantus' patent expiration and the rise of biosimilars. Evaluate the impact of biosimilars like Semglee on market dynamics.
-
Market Growth Forecast, highlighting growth opportunities within the biosimilar segment and potential risks, including pricing pressures and market share shifts post-patent expiry.
-
Regulatory and Market Barriers, identify regulatory challenges for biosimilars, such as approval processes, pricing regulations, and market access, and assess their impact on market expansion.
-
Competitive Landscape, analyzing key players, including Sanofi and biosimilar manufacturers, examining their strategies, partnerships, and market share distribution as biosimilars enter the market.
-
Regulatory Barriers, evaluating the regulatory hurdles for insulin glargine biosimilars, including approval and interchangeability, and their effect on market penetration.
-
Strategic Implications, assess strategic moves for Sanofi and competitors, including innovation, patient support, and geographic expansion, to maintain market leadership amidst growing biosimilar competition.
Patent Cliff Analysis
The patent expiration of Lantus has created a significant shift in the insulin glargine market, introducing both challenges and opportunities. Lantus, a widely used long-acting insulin, has been a cornerstone in diabetes management for years, but with the expiry of its patent, biosimilars such as Semglee have entered the market, offering cost-effective alternatives to the brand-name product. The introduction of biosimilars has the potential to erode Lantus' market share, as they typically offer lower prices, making them attractive to healthcare providers and patients, particularly in price-sensitive markets.
However, while Lantus faces pressure from biosimilars, Sanofi's Toujeo, a more concentrated formulation of insulin glargine, remains largely unaffected by this competition as no biosimilars for Toujeo have entered the market yet. This positioning gives Sanofi a strategic advantage in maintaining its market share for Toujeo, as it continues to cater to patients with higher insulin needs or those requiring more stable insulin coverage.
The impact of the patent cliff for Lantus has also prompted the development of new pricing strategies and patient support programs aimed at maintaining market presence. Additionally, regulatory approvals for interchangeable biosimilars, such as Semglee, will likely drive further competition and market dynamics, especially as reimbursement policies and market access conditions evolve globally.

Current Market Scenarios
The insulin glargine market is experiencing notable changes, primarily driven by the expiration of the patent for Lantus, Sanofi's long-acting insulin. Lantus has been a dominant treatment for managing type 1 and type 2 diabetes, widely adopted for its ability to provide stable, 24-hour blood glucose control. The product has contributed significantly to Sanofi’s revenue. However, as its patent expired in 2020 in some markets and is expiring in others, the market landscape is shifting with the entry of biosimilars, such as Semglee by Biocon and Viatris. These biosimilars represent a significant challenge to Lantus’s market share due to their lower cost, typically 15-30% less than the originator, making them attractive alternatives, especially in price-sensitive regions.
The entry of biosimilars marks a crucial moment for the insulin glargine market. Semglee was the first biosimilar insulin glargine to receive FDA approval in 2020 and has since been introduced to the U.S. market. The competition is expected to intensify with additional biosimilars such as Basaglar (by Novo Nordisk) and other future products entering the U.S. and European markets. These biosimilars provide cost-effective alternatives and are positioned to capture market share, particularly in regions where healthcare systems emphasize cost containment, such as the U.S. and Europe. The price reduction from biosimilars will likely increase patient access to insulin, especially in emerging markets where affordability remains a major barrier.
The regulatory landscape surrounding biosimilars is accelerating, with authorities like the U.S. FDA and the European Medicines Agency (EMA) streamlining approval processes to facilitate faster market entry. The FDA’s approval of interchangeable biosimilars such as Semglee is setting a precedent, allowing for substitution at the pharmacy level without requiring a new prescription. This could lead to more widespread adoption of biosimilars, further enhancing competition for Lantus. In addition, regulatory support for biosimilars is expected to reduce barriers to entry in global markets, especially in emerging economies, where the demand for affordable insulin is increasing.
For Sanofi, the challenge lies in defending its market leadership in the face of growing competition from biosimilars. The company will need to adapt by focusing on reinforcing the value of its branded products like Toujeo, which remains the more concentrated version of insulin glargine (U-300) with a longer duration of action (up to 36 hours). As of now, there are no biosimilars for Toujeo, which provides Sanofi with a temporary advantage in maintaining its position in the market for more specialized insulin needs. Moreover, Sanofi may explore new indications for Toujeo or other innovative therapies to stay competitive.
The market dynamics are expected to evolve as biosimilars continue to grow in popularity and as more regulatory approvals facilitate access. With an increasing shift toward biosimilars, the insulin glargine market is becoming more competitive, and patient access to affordable treatments is improving. However, companies like Sanofi must adopt strategies, such as reinforcing patient loyalty, focusing on innovation, and exploring new markets, to sustain their market positions in the face of mounting competition.
Market Dynamics
“Expanding Healthcare Access and Technological Advancements”
The insulin glargine market is driven by the increasing accessibility of diabetes care, especially in emerging markets. As healthcare infrastructure improves in regions like Asia Pacific and Latin America, the demand for insulin therapies grows. Additionally, advancements in diabetes management technologies, including continuous glucose monitors (CGMs) and insulin pumps, have streamlined the treatment process, enhancing patient outcomes. The rising adoption of digital health technologies and the integration of smart insulin pens are expected to accelerate the use of insulin products like Lantus and Toujeo, as they allow for more personalized care and better management of blood glucose levels.
“Cost Containment Measures and Competitive Pricing Pressures”
While the demand for insulin glargine products is increasing, cost containment measures and growing competition from biosimilars pose significant challenges. Governments and insurers in both developed and emerging markets are increasingly focusing on reducing healthcare expenditures, which intensifies the pressure on insulin pricing. Biosimilars, priced 15-30% lower than Lantus, are gaining ground, particularly in cost-sensitive regions. These cost-effective alternatives threaten the market share of Lantus and Toujeo, forcing Sanofi to adjust its pricing strategies and potentially affecting profitability in these regions. Furthermore, healthcare reforms targeting drug pricing in major markets like the U.S. and Europe may limit reimbursement for branded products, reducing overall market potential.
“Increased Focus on Preventive Healthcare and Patient Support Programs”
There is a growing opportunity for insulin glargine products in markets where preventive healthcare initiatives are gaining traction. As more healthcare systems focus on the prevention of diabetes through screening programs, earlier diagnosis and treatment will increase the demand for long-acting insulins like Lantus and Toujeo. Additionally, patient support programs that offer affordability solutions, such as co-pay assistance and patient education, present an opportunity for Sanofi to strengthen its market presence. As governments and non-governmental organizations emphasize chronic disease management, this creates an opportunity for Sanofi to expand its reach in underserved regions, particularly in emerging economies where diabetes incidence is rising rapidly.
“Increasing Shift Towards Personalized Diabetes Management”
A notable trend in the insulin glargine market is the increasing shift towards personalized diabetes management. With a growing focus on tailoring treatments based on patient needs, the demand for insulin formulations that offer more precise and flexible options is rising. Toujeo, with its longer duration of action compared to Lantus, is positioned to cater to patients requiring more stable insulin control. This trend is reinforced by the increasing use of wearable devices, such as insulin pumps and smart insulin pens, which help patients manage their insulin therapy more effectively. As the market moves towards more individualized treatment plans, Sanofi has the opportunity to capitalize on the benefits of Toujeo’s extended action and innovative delivery systems to differentiate its products in an increasingly competitive market.
Overview of Alternative Therapeutics
As the insulin glargine market faces increased competition, several alternative therapeutics are emerging. Rapid-acting insulins like Insulin Lispro (Humalog) and Insulin Aspart (NovoLog) offer quicker onset and shorter duration of action, commonly used alongside long-acting insulins for comprehensive diabetes management. Competing directly with Lantus are biosimilars such as Semglee and Basaglar, which provide cost-effective alternatives while maintaining similar clinical outcomes.
Alternative long-acting insulins, such as insulin degludec (Tresiba) and insulin detemir (Levemir), offer extended durations of action, with Tresiba lasting up to 42 hours, providing more flexible dosing schedules compared to Lantus. Additionally, the development of oral insulins, such as Oramed Pharmaceuticals' Oral Insulin, may transform treatment by offering a non-injection option, though they are still in the development phase.
Insulin pumps and continuous glucose monitors (CGMs) are also becoming important tools in diabetes management, enabling more precise insulin delivery and blood glucose control. These devices work alongside insulin therapies to offer more convenience and improved patient outcomes. These emerging alternatives, along with biosimilars to insulin glargine, are intensifying competition in the insulin market. As a result, Sanofi's Lantus and Toujeo will need to focus on differentiation, patient support, and innovation to maintain their market position. The insulin market is evolving, with cost-effectiveness, convenience, and innovation becoming key drivers of growth.
Competitive Landscape
The insulin glargine market is increasingly competitive following the patent expiration of Lantus and the rise of biosimilars. Key competitors include Semglee (Biocon and Viatris), the first FDA-approved interchangeable biosimilar to Lantus, which is priced 15-30% lower, providing a cost-effective alternative. Basaglar, developed by Novo Nordisk, is another prominent insulin glargine biosimilar that has captured market share due to its lower price and similar clinical outcomes.
Novo Nordisk also competes with its long-acting insulin formulations like Tresiba (insulin degludec), offering a longer duration of action (up to 42 hours) compared to Lantus, providing patients with more flexible dosing options. Levemir (insulin detemir), another offering from Novo Nordisk, competes directly with Lantus in the long-acting insulin segment, though it has a slightly shorter duration of action.
Technological advancements, such as continuous glucose monitors (CGMs) and insulin pumps, are also influencing the competitive landscape, improving diabetes management but not directly competing with insulin products. Additionally, oral insulin formulations, such as those being developed by Oramed Pharmaceuticals, could provide an alternative to injectable insulins in the future.
As competition intensifies, Sanofi must leverage strategies such as expanding the use of Toujeo (a more concentrated form of insulin glargine) and focusing on patient support programs to maintain market share. Biosimilars and alternative insulin products are reshaping the insulin market, driving the need for Sanofi and other companies to innovate and differentiate their offerings to remain competitive.
Regional Analysis
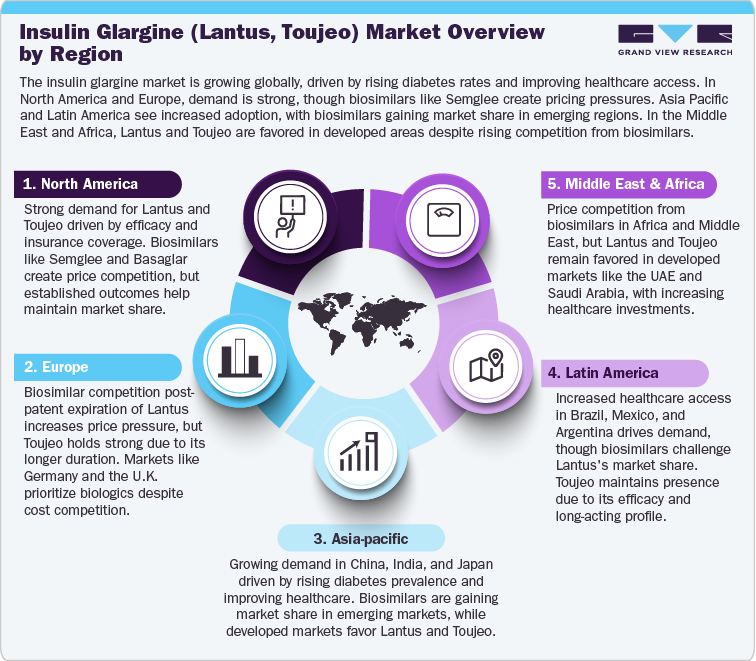
North America Insulin Glargine (Lantus, Toujeo) Market
The insulin glargine market in North America, particularly in the U.S., remains a key segment in diabetes management. Lantus and Toujeo are widely used for type 1 and type 2 diabetes, with Medicare and private insurance providing substantial coverage for these products. As of 2020, Lantus continues to hold a strong position, though the market is increasingly facing competition from biosimilars like Semglee (approved as the first interchangeable biosimilar insulin glargine by the FDA in 2020) and Basaglar (approved by the FDA in 2015). These biosimilars are priced 15-30% lower than Lantus, and with ongoing healthcare reforms focusing on cost containment, biosimilars are likely to capture a growing share, especially in cost-sensitive Medicare markets. Despite biosimilar competition, the clinical reliability and strong market positioning of Toujeo, with its longer duration of action (up to 36 hours), provide Sanofi with a competitive edge in the long-acting insulin market.
Europe Insulin Glargine (Lantus, Toujeo) Market
The insulin glargine market in Europe remains robust, particularly in Germany, France, and the U.K., where Lantus and Toujeo are commonly used in type 1 and type 2 diabetes management. In 2023, Lantus faced the expiration of its patent, opening the door for biosimilars such as Semglee and Abasaglar (both already approved in multiple European countries). These biosimilars, priced at 15-30% lower than Lantus, will likely increase market pressure, particularly in price-sensitive markets within Eastern Europe and the U.K., where cost containment measures are a primary focus. In Germany and France, where reimbursement policies favor cost-effective biologics, biosimilars are expected to gain significant market share in the coming years, despite Lantus’ established efficacy. Toujeo's relatively higher cost and longer duration (up to 36 hours) may help it maintain a strong position in markets requiring more stable insulin control.
Asia Pacific Insulin Glargine (Lantus, Toujeo) Market
The insulin glargine market in Asia Pacific is growing, driven by increasing diabetes prevalence in China, India, and Japan. In Japan, Lantus and Toujeo continue to be primary treatments for type 1 and type 2 diabetes, with Toujeo benefiting from its longer duration of action and a growing base of diabetes patients. However, competition is intensifying due to the rise of biosimilars such as Semglee and Basaglar, which have been approved in several Asian markets, including India and China, where price sensitivity is a critical factor. In China, the biosimilar market is rapidly expanding, and the Chinese government’s focus on lowering healthcare costs is likely to favor biosimilars, particularly in cost-sensitive regions. Despite these challenges, the clinical efficacy of insulin glargine in diabetes management ensures that Lantus and Toujeo maintain relevance in Japan, where there is strong demand for reliable long-acting insulin therapies.
Latin America Insulin Glargine (Lantus, Toujeo) Market
The insulin glargine market in Latin America is seeing growth due to improved healthcare access and rising diabetes awareness in countries such as Brazil, Mexico, and Argentina. Lantus and Toujeo are well-established in these regions, with Brazil being a key market where Lantus remains the dominant long-acting insulin. However, the entry of biosimilars, such as Semglee and Basaglar, is expected to significantly impact the market, especially as biosimilars are priced 15-40% lower than the original products. Biosimilars are particularly attractive in Argentina and Mexico, where cost sensitivity is high, and healthcare systems are increasingly focused on reducing drug expenditures. While Lantus and Toujeo continue to have a strong presence in these regions, particularly in more established healthcare systems, the growth of biosimilars will likely shift market dynamics in favor of more affordable alternatives in emerging markets. Despite this, Sanofi's established reputation and the clinical efficacy of its insulin glargine products will continue to maintain a foothold in these countries.
Middle East and Africa Insulin Glargine (Lantus, Toujeo) Market
The insulin glargine market in the Middle East and Africa is growing, driven by increasing diabetes prevalence and improving healthcare infrastructure. In developed markets like the UAE and Saudi Arabia, demand for long-acting insulins such as Lantus and Toujeo remains strong, with both products being widely used due to their proven efficacy in managing type 1 and type 2 diabetes. In these markets, the healthcare system supports access to biologic therapies, and the use of insulin glargine remains a cornerstone of diabetes management. However, in emerging markets across Africa and certain parts of the Middle East, cost-sensitive healthcare systems limit the affordability and access to Lantus and Toujeo. Biosimilars, including Semglee and Basaglar, are expected to play a crucial role in addressing these affordability challenges. These biosimilars are generally priced 15-30% lower than the branded insulin glargine, making them attractive alternatives in regions where healthcare budgets are constrained.
Analyst Perspective
The insulin glargine market in the Middle East and Africa is growing, with Lantus and Toujeo holding strong positions in developed markets like the UAE and Saudi Arabia, driven by rising diabetes prevalence and improving healthcare infrastructure. In emerging markets across Africa and parts of the Middle East, biosimilars such as Semglee and Basaglar are expected to gain traction due to their lower pricing (Less than branded products), offering a more affordable alternative. While Sanofi benefits from established clinical outcomes, the rise of biosimilars in cost-sensitive regions poses a challenge. To maintain its market share, Sanofi will need to focus on product differentiation and patient support programs.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028-2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU -15-30% Yr-1, deepening to -45-60% by Yr-3; U.S. -10-25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25-40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30-40% global revenue decline by Year-3 post-LOE.
-
Downside: 45-55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~-20-25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


