- Home
- »
- Market Trend Reports
- »
-
Insulin Lispro (Humalog) Market And Forecast, 2025 - 2033
Report Overview
Insulin Lispro, marketed as Humalog by Eli Lilly, is one of the leading rapid-acting insulins used in the management of Type 1 and Type 2 diabetes. Its formulation provides a rapid onset of action, making it a preferred choice for patients needing quick postprandial glucose control. Humalog’s market dominance has been driven by its clinical efficacy, extensive regulatory approvals, and widespread availability. However, the market dynamics are shifting as Humalog approaches the expiration of its patent. The expiration is expected to introduce increased competition from biosimilars, which offer lower-cost alternatives while maintaining comparable therapeutic benefits. The emergence of these biosimilars, along with increasing pricing pressures, especially in cost-sensitive regions, will likely challenge Humalog’s market share.
Key Report Deliverables
-
Insulin Lispro (Humalog) Market Landscape: Humalog, by Eli Lilly, is a leading rapid-acting insulin for diabetes management. As its patent nears expiration, biosimilars will increase market competition, especially in price-sensitive regions. Additionally, the rise of insulin pumps and continuous glucose monitoring is shifting the treatment landscape.
-
Market Growth Forecast: Growth will slow as biosimilars emerge, but insulin pumps and digital health solutions will drive opportunities, particularly in emerging markets with growing diabetes prevalence.
-
Regulatory and Market Barriers: Biosimilars face rigorous approval processes, delaying market entry. Reimbursement models may also create barriers to market adoption, influencing the biosimilar landscape.
-
Competitive Landscape: Eli Lilly faces competition from biosimilar makers like Mylan and Sandoz, with market share determined by pricing, clinical outcomes, and device partnerships.
-
Regulatory Barriers: FDA and EMA approval processes for biosimilars are complex, with clinical similarity and market access posing challenges for rapid entry.
- Strategic Implications: Eli Lilly will focus on innovation and integrated therapy solutions. Biosimilars will prioritize regulatory approval and competitive pricing for market penetration.

The expiration of Humalog’s patents marks a critical juncture for Eli Lilly in the rapidly evolving diabetes care market. Humalog, originally approved in 1996, had a strong patent portfolio protecting its compound and formulation, with patents expiring in 2013 and 2014. This expiration allowed for the development and entry of biosimilars into the market, most notably Mylan’s and Sandoz’s insulin lispro formulations, which offer lower-cost alternatives to the original product. The biosimilars, while therapeutically similar to Humalog, pose a significant competitive threat due to their reduced price points. The introduction of these biosimilars is expected to drive increased market competition, particularly in price-sensitive regions where access to insulin therapies remains a key concern.
The impact of Humalog’s patent cliff is further amplified by the expiration of patents covering Humalog’s delivery devices, including the KwikPen, scheduled for 2024. The delivery devices have been integral to the product’s success, offering a user-friendly insulin delivery method. With these patents expiring, generic versions of the devices will become available, intensifying market competition. Biosimilars will not only target Humalog’s core insulin formulations but will also have the opportunity to enter the device market, potentially capturing market share from Eli Lilly’s established user base. This dual threat from biosimilars targeting both the insulin and device components of Humalog necessitates strategic adaptations from Eli Lilly to maintain its market position.
In response to the patent cliff, Eli Lilly is likely to adjust its revenue and market strategies to retain its leadership position in the insulin market. The company may explore the expansion of its biosimilar portfolio, as well as the development of next-generation insulin products to differentiate itself from biosimilar competitors. Additionally, Eli Lilly could increase its focus on patient support programs and partnerships with healthcare providers to ensure continued brand loyalty despite the availability of biosimilars. In conclusion, while the expiration of Humalog’s patents presents challenges, it also offers opportunities for Eli Lilly to diversify its offerings and leverage innovation to sustain revenue streams in a more competitive market environment.
Current Market Scenarios
The Insulin Lispro (Humalog) market is undergoing a significant shift following the expiration of its key patents. Humalog, developed by Eli Lilly, has been a leading treatment for Type 1 and Type 2 diabetes, driven by its effectiveness in managing postprandial blood glucose levels. The expiration of its compound and formulation patents in 2013 and 2014 opened the market to biosimilars, with companies like Mylan, Sandoz, and Wockhardt developing biosimilars to Humalog. These biosimilars are expected to be priced 15-40% lower than the originator product, increasing competition, particularly in price-sensitive markets. The market for insulin biosimilars is growing, and the FDA approved the first biosimilar to Humalog in 2019, with others anticipated to enter the market soon.
The regulatory environment has adapted to accommodate biosimilars, with agencies like the FDA and EMA fast-tracking approvals to facilitate quicker market entry. The expiration of Humalog’s delivery device patents, including for the KwikPen, in 2024, will further increase competitive pressures, allowing generic versions of the delivery devices to enter the market. For Eli Lilly, the challenge is to maintain its market share as biosimilars provide lower-cost alternatives. The company is likely to focus on expanding its biosimilar portfolio, advancing next-generation insulin formulations, and reinforcing patient loyalty programs to address the competitive dynamics and sustain its market leadership.
Market Dynamics
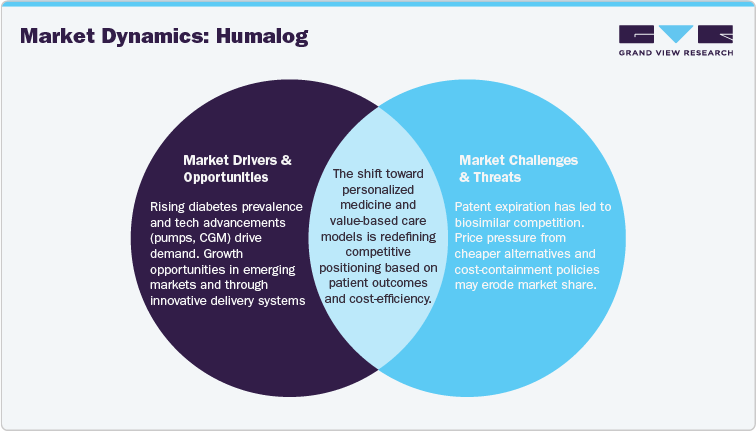
"Rising Diabetes Prevalence and Technological Advancements"
The increasing global prevalence of Type 1 and Type 2 diabetes continues to drive the demand for effective insulin therapies like Humalog. As more individuals are diagnosed with diabetes, the need for rapid-acting insulin to manage postprandial glucose levels grows. Additionally, advancements in insulin delivery technologies, such as the rise of insulin pumps and integration with continuous glucose monitoring (CGM) systems, are enhancing the precision and efficiency of diabetes management. These innovations not only improve patient adherence but also support better long-term health outcomes, reinforcing the ongoing demand for Humalog in the market.
"Biosimilar Competition and Price Pressure"
The expiration of Humalog’s patents has led to the entry of biosimilars into the insulin market, creating significant price competition. These biosimilars, typically priced 15-40% lower than the originator product, are expected to capture market share, particularly in cost-sensitive regions. The increasing pressure on pricing flexibility, compounded by regulatory changes focusing on cost containment in developed markets like the U.S. and Europe, is pushing healthcare providers to adopt more affordable alternatives. As a result, Humalog's market share could experience a decline, especially in regions where price is a crucial factor in treatment choices.
"Expansion in Emerging Markets and Innovations"
Emerging markets present a significant growth opportunity for Humalog, driven by improvements in healthcare access and rising demand for biologic treatments. With healthcare infrastructure improving in regions like Asia, Latin America, and Africa, the adoption of insulin therapies is increasing. By adjusting pricing strategies to meet the demands of these price-sensitive markets and forming partnerships with local healthcare providers, Humalog can expand its footprint in these regions. Additionally, innovations such as next-generation insulin delivery systems integrated with digital health solutions could further enhance the market position and patient adherence to Humalog.
"Shift Toward Personalized Medicine and Value-Based Care"
The trend toward personalized medicine is reshaping diabetes treatment, with an emphasis on genomic profiling and biomarker identification to tailor therapies. This shift allows Humalog to integrate into more targeted treatment regimens, potentially increasing its role in individualized care. Furthermore, the adoption of value-based care models, which focus on patient outcomes and cost-efficiency, is influencing the way Humalog is priced and marketed. This transition toward outcome-based pricing models and patient-centric approaches could redefine its competitive positioning, especially as biosimilars continue to make price-sensitive inroads into the market.
Competitive Landscape
The competitive landscape for Insulin Lispro (Humalog) has evolved significantly following the expiration of its key patents, leading to the introduction of biosimilars into the market. Companies such as Biocon Biologics, Mylan, and Wockhardt have developed biosimilars to insulin lispro, offering cost-effective alternatives to Humalog. These biosimilars are expected to capture market share, particularly in price-sensitive regions, due to their lower pricing compared to the originator product. The entry of these biosimilars introduces significant price competition, which may impact Humalog’s market share, especially in emerging markets where affordability is a major factor in treatment decisions.
In addition to biosimilars, Humalog faces competition from other rapid-acting insulins such as Novolog (insulin aspart) and Apidra (insulin glulisine). Novolog, developed by Novo Nordisk, is a strong competitor, benefiting from a broad global presence and clinical adoption. Apidra, produced by Sanofi, offers an alternative in the rapid-acting insulin category, although it holds a smaller market share compared to Humalog and Novolog. Despite this competition, Humalog continues to maintain its position due to its extensive clinical history and well-established use in insulin pumps and smart insulin delivery systems, which enhance patient adherence and blood glucose control.
The adoption of insulin pumps and continuous glucose monitoring (CGM) systems continues to grow, further increasing competition within the rapid-acting insulin segment. These systems offer enhanced insulin delivery and better glucose management, making it increasingly important for insulin providers like Humalog to remain compatible with the latest smart technologies. As more biosimilars and alternative insulin products are introduced, Humalog will need to focus on product innovation, pricing strategies, and patient engagement to retain its market position and address growing competitive pressures.
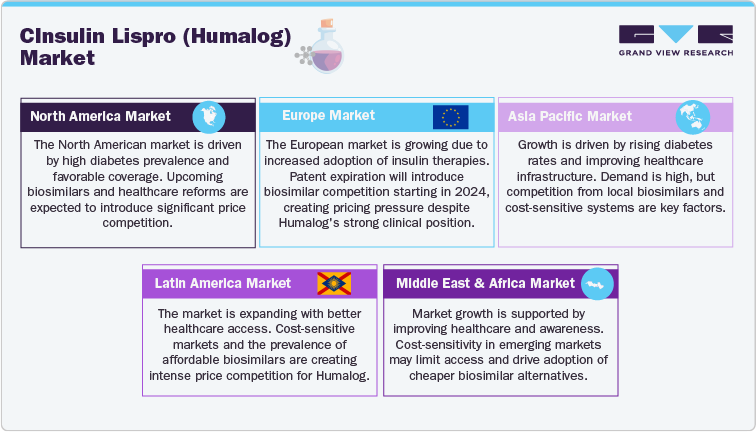
North America Insulin Lispro (Humalog) Market
The Insulin Lispro (Humalog) market in North America, particularly the U.S., remains a key region, driven by the growing prevalence of Type 1 and Type 2 diabetes. The U.S. healthcare system, which supports the use of biologic insulin therapies, provides favorable coverage for Humalog through Medicare and private insurers, ensuring its continued presence in treatment regimens. Despite this, the entry of biosimilars to insulin lispro, expected to launch in the coming years, will introduce substantial price competition, particularly as healthcare reforms continue to emphasize cost containment in biologic therapies. The market dynamics in the U.S. are likely to shift as insurance providers increasingly seek more affordable insulin alternatives, though Humalog’s proven clinical efficacy and established position in the market should help retain its relevance.
Europe Insulin Lispro (Humalog) Market
In Europe, the Humalog market continues to grow, driven by increasing adoption of insulin therapies for diabetes management. Following the expiration of its patents, biosimilars are expected to enter the European market, starting in 2024, which will introduce competition, especially in price-sensitive regions. Markets like Germany, France, and the UK, with their robust healthcare systems, continue to prioritize insulin therapies, but the affordability of biosimilars will create pricing pressures for Humalog, particularly in countries where healthcare reforms are focusing on cost containment. Despite these pressures, Humalog’s established clinical outcomes and long-standing market presence help maintain its competitive position.
Asia Pacific Insulin Lispro (Humalog) Market
The Asia Pacific region is witnessing growth in the Humalog market, primarily driven by the increasing prevalence of diabetes and improving healthcare infrastructure, particularly in countries such as China, India, and Japan. As these countries expand their healthcare access, the demand for insulin therapies like Humalog is on the rise. In developed markets like Japan, Humalog holds a strong position due to its clinical efficacy and regulatory approval. However, the market dynamics in emerging markets are impacted by the rise of biosimilars and cost-sensitive healthcare systems, with the availability of affordable alternatives driving market shifts. Despite competition from local biosimilars and other insulin products, Humalog’s proven clinical performance continues to support its market presence in the region.
Latin America Insulin Lispro (Humalog) Market
The Humalog market in Latin America is expanding, driven by improvements in healthcare access and a growing understanding of diabetes management, especially in countries like Brazil, Mexico, and Argentina. As healthcare infrastructure in the region improves, there is greater demand for insulin therapies. However, cost-sensitive markets in Latin America pose challenges, as biosimilars to insulin lispro become more prevalent, offering affordable alternatives. These biosimilars are expected to intensify price competition, particularly as healthcare systems in the region prioritize cost-effective solutions. Nevertheless, Humalog’s established reputation and clinical efficacy continue to maintain its market presence, with significant growth opportunities as biologic access improves.
Middle East and Africa Insulin Lispro (Humalog) Market
The Humalog market in the Middle East and Africa is growing, supported by improving healthcare infrastructure and rising awareness of diabetes management in countries like UAE, Saudi Arabia, and other developed markets. As demand for biologic insulin therapies increases, Humalog continues to be a preferred treatment option. However, in many emerging markets across Africa and parts of the Middle East, cost-sensitive healthcare systems may limit access to biologics, driving the adoption of biosimilars. These biosimilars will likely increase price competition, particularly in regions where affordability is a major concern. Despite these challenges, Humalog’s clinical outcomes and increasing healthcare investments continue to support its market presence in the region.
Analyst Perspective
The Insulin Lispro (Humalog) market is at a critical juncture as it faces increasing pressure from biosimilars and the broader trend toward cost containment in healthcare systems globally. The expiration of Humalog’s patents has already enabled competitors like Mylan, Sandoz, and Wockhardt to launch lower-cost alternatives, particularly in price-sensitive regions such as emerging markets and parts of Europe and the U.S. While Humalog’s clinical efficacy and established use in insulin pumps and smart insulin systems help maintain its relevance, the growing adoption of biosimilars will significantly shift market dynamics. Eli Lilly’s strategy will need to adapt quickly, leveraging its existing brand recognition and exploring next-generation formulations and integrated digital solutions to maintain market leadership. As biosimilars continue to evolve, pricing strategies and patient loyalty programs will become critical to ensuring continued dominance in a rapidly changing landscape.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028–2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU −15–30% Yr-1, deepening to −45–60% by Yr-3; U.S. −10–25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25–40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30–40% global revenue decline by Year-3 post-LOE.
-
Downside: 45–55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~−20–25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


