- Home
- »
- Market Trend Reports
- »
-
Leuprolide (Lupron): Navigating Patent Expiry With Strategic Precision
Report Overview
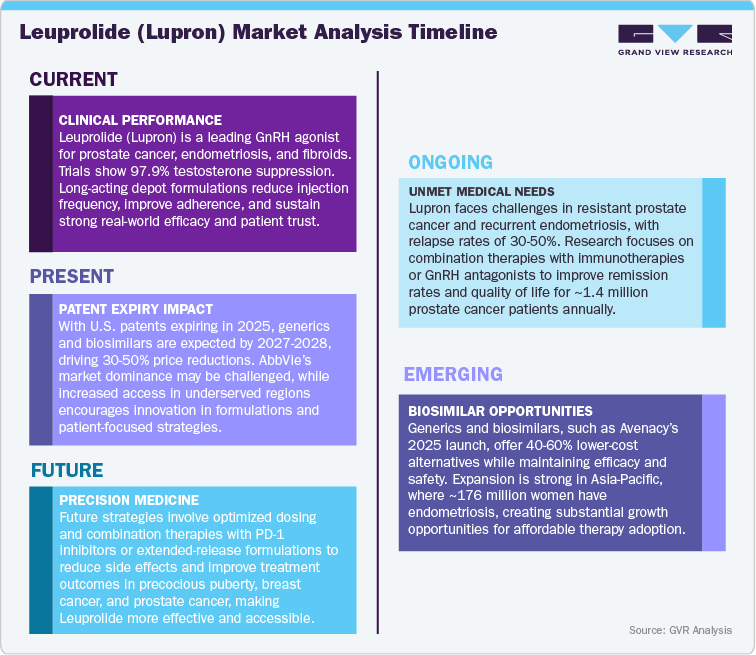
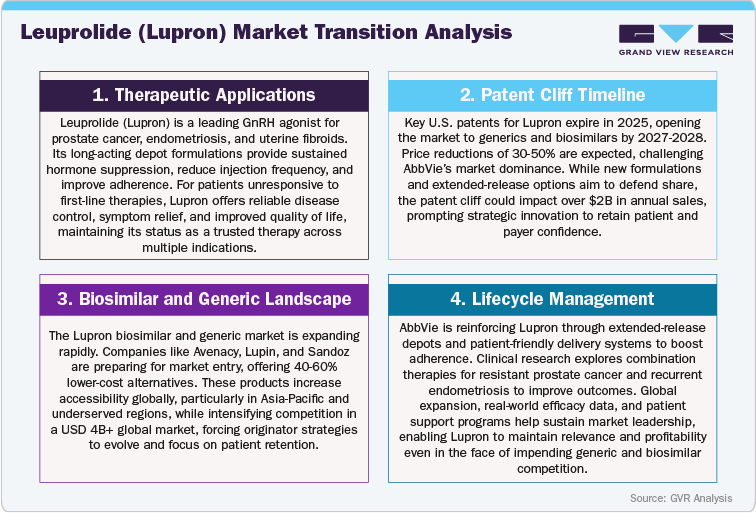
Leuprolide (Lupron), developed by Takeda Pharmaceutical Company and co-marketed by AbbVie in several regions, is a long-acting gonadotropin-releasing hormone (GnRH) agonist widely used in the treatment of advanced prostate cancer, endometriosis, uterine fibroids, and central precocious puberty (CPP). Approved across more than 80 countries, Lupron has established itself as a cornerstone therapy in hormone regulation, offering multiple long-acting depot formulations that enable sustained efficacy and patient compliance. With key patents having expired in major markets and additional formulation protections nearing their end, the Lupron market is entering a competitive phase marked by the rise of generic leuprolide depots and next-generation oral GnRH antagonists such as relugolix (Orgovyx) and elagolix (Orilissa). Despite increasing pricing pressure, Lupron continues to hold a strong market position due to its extensive clinical history, diverse indications, and brand trust. Going forward, strategic life-cycle management, delivery innovation, and expansion into emerging markets will be critical for sustaining market relevance as the brand navigates a maturing yet evolving hormonal therapy landscape.
Key Report Deliverables
-
A comprehensive analysis of the Leuprolide (Lupron) market landscape, covering global revenue performance, key growth drivers in oncology and women’s health (including prostate cancer, endometriosis, uterine fibroids, and central precocious puberty), evolving treatment paradigms across depot and oral GnRH therapies, and the shifting competitive context within the hormonal therapy segment.
-
Forecasts evaluating post-patent market dynamics, including expected generic leuprolide depot entry timelines across major regions, and the projected impact on revenue growth, pricing pressures, and market access following patent expirations and formulation exclusivity lapses in key markets.
-
Identification of regulatory and market access barriers influencing generic and next-generation GnRH therapy adoption, including approval complexities, interchangeability standards, pricing and reimbursement frameworks, and payer-driven formulary decisions in key geographies such as the U.S., Europe, Asia Pacific, and Latin America.
-
An in-depth competitive landscape overview, profiling direct competitors in the GnRH agonist market, emerging generic leuprolide developers, next-generation oral GnRH antagonists (e.g., relugolix, elagolix), ongoing pipeline innovations, and evolving treatment strategies shaping the future of hormone-sensitive disease management.
-
Strategic implications for AbbVie and Takeda, including lifecycle management initiatives, defense against generic erosion, new formulation development (e.g., extended depot or patient-friendly delivery devices), expansion into pediatric and emerging indications, pricing optimization, and regional market expansion to maintain leadership in the global hormonal therapy market amid intensifying generic and oral competitor competition.
Patent Cliff Analysis:
The revenue trajectory for Leuprolide (Lupron), a GnRH agonist widely used in treating prostate cancer, endometriosis, and precocious puberty, is expected to undergo gradual erosion over the next decade as patents expire, with substantial market share losses anticipated in the early 2030s. The key U.S. and European patents for Lupron formulations are set to expire between 2025 and 2027, which will mark the beginning of intensified competition from generic alternatives. In the short term, up until 2025, Lupron will maintain a dominant market position, facing minimal competitive pressure and experiencing only modest revenue decline of around 2-4%, thanks to its established clinical preference and long-standing safety profile.
As patents begin to lapse post-2025, Lupron will confront increasing competition from generic leuprolide acetate, as well as new long-acting formulations developed by competitors. Between 2026 and 2029, the market will see a gradual shift, with generics capturing 10-20% of Lupron’s share, resulting in a revenue reduction. This period will also witness the uptake of alternative GnRH agonists and antagonists, which offer improved dosing schedules or reduced side effects, further intensifying competitive pressures. Pricing negotiations with payers in major markets such as the U.S. and Europe will begin to erode Lupron’s premium pricing advantage.
The most dramatic phase of the patent cliff is expected between 2030 and 2033, when Lupron faces widespread generic adoption. During this period, Lupron’s market share could decline by 50-65%, with generics and biosimilars providing highly cost-effective alternatives for healthcare providers and patients. The entry of alternative hormone therapies and next-generation treatments for prostate cancer and endometriosis will further fragment the market, accelerating the decline in Lupron’s dominance.
By 2034, Lupron is expected to hold only 25-30% of its current global market share, with generics and biosimilars firmly established as the mainstay in hormonal therapy. While the drug will remain available, particularly in certain specialized or low-income markets, its role will be considerably diminished. This transition will mark the final phase of Lupron’s patent cliff, with pricing pressures stabilizing and the competitive landscape fully mature. Ultimately, Leuprolide will retain a presence in the market, but its former leadership in hormone-dependent therapies will be largely supplanted by more affordable alternatives.
Current Market Scenarios
Leuprolide (Lupron), developed by Takeda Pharmaceuticals and marketed in key regions by AbbVie, continues to hold a significant position in global hormonal therapy markets, including oncology and women’s health. Although many of the original depot formulation patents have expired, certain formulation- and indication-specific protections have historically extended market exclusivity in select regions, allowing Lupron to maintain a strong revenue base and clinical adoption. However, with generic leuprolide depots gaining traction and next-generation oral GnRH antagonists (e.g., relugolix and elagolix) entering the market, the competitive landscape is shifting, creating pressure on pricing and brand share. Regional differences in patent expiry timelines, regulatory approval pathways, and payer policies will heavily influence the speed of generic and oral therapy adoption.
In Europe, cost-containment initiatives and tender-driven procurement are likely to accelerate uptake of generics and oral alternatives, applying downward pressure on Lupron’s pricing. In Asia Pacific-particularly in China, India, and South Korea-local manufacturing and regulatory incentives for generics are expected to drive early market penetration, enhancing patient access while intensifying competition for branded depots. Conversely, in the U.S. and Japan, clinical conservatism, prescriber familiarity with depot formulations, and complex interchangeability rules may allow Lupron to retain a premium market position for the near term.
Despite growing competition, demand for Lupron remains robust due to its proven efficacy across multiple indications, well-established depot options, and strong physician and patient trust. Ongoing efforts in lifecycle management, including development of enhanced depot formulations, patient-friendly delivery systems, and pediatric labeling, are designed to reinforce market relevance. Moving forward, the hormonal therapy landscape will be defined by generic penetration, adoption of oral GnRH therapies, and innovation-led differentiation, with Takeda and AbbVie expected to focus on strategic formulation improvements, geographic expansion, and targeted market access initiatives to sustain leadership in this evolving therapeutic segment.
Market Dynamics
Growing Demand for Hormonal Therapies
The rising global prevalence of hormone-sensitive conditions, including advanced prostate cancer, endometriosis, uterine fibroids, and central precocious puberty (CPP), has driven sustained demand for Leuprolide (Lupron). As a long-acting GnRH agonist, Lupron offers consistent and reliable hormone suppression, with established clinical efficacy and well-understood safety profiles, making it a preferred choice for patients and physicians across oncology and women’s health. The drug’s multiple depot formulations and dosing options, along with ongoing research into enhanced delivery systems and pediatric indications, continue to reinforce its market position. Supported by strong real-world evidence, broad geographic adoption, and high prescriber confidence, Lupron remains a cornerstone therapy in hormonal management. Increasing awareness of hormonal modulation benefits and expanded treatment paradigms will further sustain demand across both developed and emerging markets.
Pricing and Market Erosion Post-Patent
With generic leuprolide depots entering key markets and emerging oral GnRH antagonists such as relugolix (Orgovyx) and elagolix (Orilissa) gaining adoption, the Lupron market is facing competitive pressures on pricing and market share. In cost-sensitive regions such as Europe, India, and Asia Pacific, payers are likely to favor generics and oral alternatives, accelerating uptake and applying downward pricing pressure on branded Lupron. In contrast, in markets like the U.S., Japan, and Western Europe, prescriber familiarity with depot formulations, clinical conservatism, and regulatory complexities may slow generic adoption, allowing Lupron to retain premium positioning in the near term. Overall, revenue and market share could gradually erode as competitive products gain traction, though brand trust and clinical evidence will remain key differentiators.
Opportunities in Lifecycle Management and Regional Divergence
AbbVie and Takeda are addressing market pressures through lifecycle management strategies, including development of longer-acting depots, patient-friendly delivery systems, and expanded pediatric labeling. Clinical research exploring new dosing regimens, combination therapies, and emerging indications could provide additional growth pathways in the post-patent era. Competitive impact will vary by region: while stringent regulations and physician loyalty may delay uptake of generics and oral therapies in the U.S., Japan, and Western Europe, emerging markets such as China, India, and Latin America are likely to experience faster adoption due to affordability initiatives and local manufacturing incentives. To sustain leadership, strategic emphasis on formulation innovation, regional pricing optimization, and targeted market access initiatives will be critical, ensuring Lupron remains a dominant force in the evolving global hormonal therapy landscape.
The Pressure of Pricing and Market Erosion Post-Patent
As Leuprolide (Lupron) faces increasing competition from generic depot formulations and next-generation oral GnRH antagonists such as relugolix (Orgovyx) and elagolix (Orilissa), pricing pressures and potential market share erosion are becoming significant considerations for AbbVie and Takeda. Generics are expected to gain rapid adoption in cost-sensitive regions such as China, India, and Latin America, driven by government incentives, local manufacturing, and affordability initiatives. Conversely, adoption in mature markets like the U.S., Europe, and Japan may progress more gradually due to prescriber familiarity with Lupron depots, regulatory complexities, and loyalty to the established brand. Over time, payer-driven pricing policies, value-based contracts, and tendering systems are likely to accelerate uptake of generics and oral alternatives, reshaping market access and competitive dynamics in oncology and women’s health. Despite these challenges, Lupron’s proven long-term efficacy, multiple depot options, and trusted safety profile will help AbbVie and Takeda mitigate competitive pressures. Strategic focus on lifecycle management, formulation enhancements, and patient support programs will be essential to preserve market leadership in the hormonal therapy segment.
Innovating Beyond the Patent - Unlocking Future Growth Paths
Amid rising generic and oral GnRH competition, significant opportunities remain for Lupron to sustain growth and clinical relevance. Innovations such as longer-acting depot formulations, patient-friendly delivery systems, and optimized dosing schedules can improve convenience, adherence, and overall therapy experience. Ongoing research exploring expanded pediatric applications, combination therapies, and novel indications further differentiates Lupron in the hormonal therapy landscape. AbbVie and Takeda’s strategy, centered on formulation innovation, clinical evidence generation, and geographic expansion, will be critical in maintaining brand relevance. Additionally, the growing availability of affordable generic and oral alternatives presents opportunities to enhance patient access in emerging markets, where healthcare systems prioritize cost-effective therapies. As infrastructure and awareness improve across regions such as Asia Pacific, Latin America, and the Middle East, demand for both branded Lupron and lower-cost options is expected to rise, enabling broader patient access and reinforcing strategic leadership in the global hormonal therapy market.

Shaping the Future - Generics, Patient-Centric Models, and Regional Shifts
The shift toward patient-centric hormonal therapies is gaining momentum, emphasizing treatment convenience, adherence, and overall quality of life for patients with prostate cancer, endometriosis, uterine fibroids, and central precocious puberty. Innovations such as longer-acting depot formulations, optimized dosing schedules, and patient-friendly delivery devices are enabling more personalized therapy approaches, reinforcing Lupron’s clinical relevance even as generic depots and oral GnRH antagonists enter the market. The adoption of value-based healthcare models is expected to accelerate uptake of cost-effective alternatives in price-sensitive regions, reshaping the competitive dynamics between branded Lupron and lower-cost generics or oral therapies. Regional variations will remain critical: in mature markets like the U.S., Europe, and Japan, regulatory complexity, clinical conservatism, and physician loyalty may slow generic or oral therapy penetration, whereas emerging markets such as China, India, and Latin America are likely to experience rapid adoption driven by affordability initiatives, local manufacturing incentives, and streamlined regulatory pathways. These regional differences will create distinct competitive landscapes, requiring AbbVie and Takeda to implement tailored strategies that sustain brand leadership, optimize market access, and expand patient reach across the global hormonal therapy market.

Overview of Alternative Therapeutics - Leuprolide (Lupron) Market
Leuprolide (Lupron) is facing increasing competition from generic leuprolide depot formulations and emerging oral GnRH antagonists such as relugolix (Orgovyx) and elagolix (Orilissa). These alternatives are expanding their indications, treatment schedules, and geographic presence to capture larger shares of the global hormonal therapy market across oncology and women’s health. In parallel, innovations including longer-acting depots, patient-friendly delivery systems, and combination therapy approaches are reshaping the therapeutic landscape, intensifying competitive pressures for prostate cancer, endometriosis, uterine fibroids, and central precocious puberty management.
A growing wave of generic and oral competitors is advancing through regulatory approvals and market entry pathways, with adoption expected to accelerate in cost-sensitive regions such as China, India, and Latin America. Success for these alternatives will depend on demonstrating comparable efficacy and safety, securing regulatory clearance, offering cost advantages, and implementing effective pricing and market access strategies in regions where payers and healthcare systems heavily influence treatment decisions.
Overall, the evolving competitive landscape underscores the need for AbbVie and Takeda to leverage formulation innovation, lifecycle management, and strategic partnerships to maintain leadership in the GnRH therapy segment. As generics and oral GnRH antagonists reshape the hormonal therapy market, the companies must focus on differentiating Lupron through enhanced depot options, expanded pediatric applications, and patient-centric strategies to sustain market relevance and reinforce their position in a rapidly evolving therapeutic environment.
Competitive Landscape
The competitive landscape for Leuprolide (Lupron) is undergoing significant transformation as generic leuprolide depots and next-generation oral GnRH antagonists intensify competition across prostate cancer, endometriosis, uterine fibroids, and central precocious puberty (CPP) treatment. AbbVie and Takeda, as the primary rights-holders, continue to leverage Lupron’s established market position through a broad formulation portfolio, multiple depot options, and pediatric labeling, complemented by lifecycle management initiatives such as longer-acting depots and patient-friendly delivery systems. Competitors, including generic leuprolide manufacturers and oral GnRH agents like relugolix (Orgovyx) and elagolix (Orilissa) are expanding their indications, clinical data, and geographic reach, creating a highly competitive environment in the hormonal therapy market.
Generic leuprolide products are advancing through regulatory approvals, with adoption expected to accelerate in emerging markets such as China, India, and Latin America due to government incentives, local manufacturing, and cost-effectiveness initiatives. Meanwhile, oral GnRH antagonists are positioned as convenient alternatives with differentiated mechanisms of action, representing longer-term competition in both oncology and women’s health segments.
To maintain market leadership, AbbVie and Takeda are focusing on formulation innovation, lifecycle extensions, strategic collaborations, and patient-centric programs, while competitors emphasize pipeline diversification, oral therapy adoption, and expanding access in cost-sensitive regions. The rise of generics and oral alternatives is expected to drive pricing pressures, broaden therapy accessibility, and reshape prescribing patterns. Ultimately, the ability to innovate, differentiate, and deliver patient-focused solutions will be critical for Lupron and its competitors to sustain resilience and leadership in the evolving global hormonal therapy landscape.
North America Leuprolide Market
North America remains the largest market for Leuprolide (Lupron), with the U.S. driving the majority of sales due to advanced healthcare infrastructure, high physician familiarity, and strong adoption of depot GnRH therapies for prostate cancer, endometriosis, uterine fibroids, and central precocious puberty (CPP). While many original depot patents have already expired, brand-level exclusivities and pediatric labeling have allowed Lupron to maintain market share. The growing presence of generic leuprolide depots and oral GnRH antagonists such as relugolix (Orgovyx) is expected to increase competition, creating pricing pressures and influencing payer-driven formulary decisions. In Canada, similar dynamics are emerging, with generics and oral alternatives gaining traction post-exclusivity, driving adoption and gradual price erosion.
Europe Leuprolide Market
Europe represents a key market for Lupron, with Germany, France, and the U.K. as primary contributors. Widespread generic availability and the entry of oral GnRH antagonists are expected to accelerate the transition toward cost-effective alternatives. Europe’s tender-based procurement systems and value-driven healthcare frameworks will drive rapid uptake of generics and oral therapies, reshaping competitive dynamics. While regulatory processes may initially slow market penetration, adoption of lower-cost alternatives is expected to rise swiftly, exerting downward pressure on branded Lupron pricing.
Asia Pacific Leuprolide Market
The Asia Pacific region offers substantial growth potential for Lupron, particularly in China, India, Japan, and South Korea, where awareness of hormone-sensitive conditions and adoption of depot therapies are increasing. In China, early generic launches and government support for local manufacturing are expected to drive competition, while India’s affordability-driven market will see rapid uptake of generics. Japan, despite strong demand for advanced hormonal therapies, may experience slower generic or oral competitor penetration due to stringent regulatory requirements. Overall, Asia Pacific presents opportunities for both branded Lupron and affordable alternatives, but careful navigation of regulatory timelines and local competition is essential.
Latin America Leuprolide Market
In Latin America, rising prevalence of hormone-sensitive conditions is driving demand for Lupron, with Brazil, Mexico, and Argentina as key contributors. Affordability remains a major concern, and post-patent generic competition is expected to reduce costs and expand access. Efficient regulatory pathways in Brazil and Mexico will facilitate faster adoption of generics and oral GnRH therapies, while logistical and infrastructure challenges in certain regions will require innovative pricing and distribution strategies to ensure broad patient access.
Middle East and Africa Leuprolide Market
The Middle East and Africa (MEA) market is emerging, with Saudi Arabia, UAE, and South Africa as primary contributors. Awareness of hormone-sensitive conditions and access to depot therapies is increasing, though high treatment costs remain a barrier. The introduction of generics and oral GnRH therapies is expected to improve accessibility. Regulatory efficiency varies, with faster approvals in the UAE and Saudi Arabia, while other nations may experience delays. As healthcare infrastructure strengthens, the MEA region is projected to witness long-term growth, with generics and patient-centric delivery innovations playing a pivotal role in expanding access to Lupron.

Analyst Perspective
The Leuprolide (Lupron) market is entering a critical phase as competition intensifies from generic leuprolide depots and next-generation oral GnRH antagonists such as relugolix (Orgovyx) and elagolix (Orilissa). Historically dominated by AbbVie and Takeda, the market is expected to experience pricing pressures and shifts in market share, particularly in cost-sensitive regions such as Europe, Asia Pacific, and Latin America. Emerging competitors are expanding indications, improving delivery systems, and exploring combination regimens, heightening competitive dynamics across prostate cancer, endometriosis, uterine fibroids, and central precocious puberty (CPP) therapy areas.
Despite rising competition, demand for Lupron remains strong due to its well-established efficacy, multiple depot formulations, and trusted safety profile, which have supported sustained adoption by physicians and patients globally. The product’s relevance is further reinforced by ongoing innovation, including longer-acting depot formulations, patient-friendly delivery systems, and expanded pediatric labeling, ensuring Lupron remains a central therapy in hormonal treatment guidelines.
To maintain competitiveness, AbbVie and Takeda are prioritizing lifecycle management, real-world evidence generation, and formulation innovation to sustain clinical differentiation. Patient-centric strategies, such as flexible dosing options, adherence support programs, and enhanced convenience formulations, will be critical for retaining market leadership. Strategic initiatives-including geographic expansion, targeted market access, and collaboration with healthcare providers will enable Lupron to navigate the evolving competitive landscape, maintaining its position as a leading therapy in the global hormonal therapy market.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028-2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU -15-30% Yr-1, deepening to -45-60% by Yr-3; U.S. -10-25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25-40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30-40% global revenue decline by Year-3 post-LOE.
-
Downside: 45-55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~-20-25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


