- Home
- »
- Market Trend Reports
- »
-
Retatrutide (LY-3437943) Market Analysis, 2033: Triple-Agonist Therapy Landscape In Obesity And Metabolic Disorders
Report Overview
Retatrutide (LY-3437943), developed by Eli Lilly and Company, is an innovative triple agonist targeting the GLP-1, GIP, and glucagon receptors, engineered to deliver superior weight reduction and comprehensive metabolic benefits. By simultaneously enhancing insulin sensitivity, increasing energy expenditure, and improving lipid metabolism, Retatrutide addresses multiple pathways underlying obesity and metabolic disorders. Clinical studies have shown unprecedented efficacy in body-weight reduction and significant improvements in cardiometabolic health, positioning it as a best-in-class therapy. With convenient once-weekly administration and robust outcomes across type 2 diabetes and metabolic-associated steatohepatitis (MASH), Retatrutide represents a transformative advancement in metabolic disease management poised to redefine the global obesity market and reinforce Eli Lilly’s leadership in incretin-based innovation.
Key Report Deliverables:
-
Analyze the Retatrutide (LY-3437943) market landscape, outlining its clinical progress, phase III development, anticipated regulatory milestones, and evolving trends shaping obesity and cardiometabolic therapy adoption.
-
Evaluate the competitive environment, profiling key players such as Eli Lilly, Novo Nordisk (semaglutide), Zealand/AZ (cagrisema programs), noting differentiation by efficacy, dosing convenience, and safety outcomes.
-
Forecast market growth, projecting Retatrutide’s adoption trajectory, patient population expansion, pricing scenarios, and regional penetration across the U.S., Europe, and Asia-Pacific while analyzing payer dynamics, real-world data, and uptake drivers.
-
Identify regulatory and market barriers, offering insights into approval pathways, cardiovascular outcome expectations, pricing and reimbursement hurdles, biomarker-driven patient selection, and long-term safety surveillance that could influence commercialization.
-
Examine the concurrent competitive landscape, highlighting direct and indirect competitors, pipeline diversification, strategic collaborations, and manufacturing or supply-chain considerations shaping the emerging multi-agonist metabolic therapy market.
Introduction: Redefining the Future of Obesity and Metabolic Disease Management
Retatrutide (LY-3437943), developed by Eli Lilly and Company, is emerging as a groundbreaking therapy poised to transform the management of obesity and cardiometabolic disorders two interlinked global epidemics driving morbidity, mortality, and healthcare costs worldwide. These conditions are often rooted in insulin resistance, dysregulated lipid metabolism, and chronic low-grade inflammation, contributing to metabolic dysfunction and elevated cardiovascular risk. Despite major progress with GLP-1 and GIP receptor agonists, most current treatments offer limited weight reduction or metabolic durability, highlighting the need for next-generation agents that deliver comprehensive, multi-pathway efficacy. Retatrutide, a first-in-class triple agonist of the GLP-1, GIP, and glucagon receptors, introduces a new paradigm in metabolic medicine by simultaneously targeting glucose regulation, lipid oxidation, and energy expenditure.
Retatrutide’s mechanism of action integrates and amplifies endogenous incretin and glucagon signaling, promoting sustained weight loss, enhanced insulin sensitivity, and improved lipid and cardiovascular profiles. This triple-receptor synergy produces exceptional reductions in body weight-up to 24% in mid-stage trials-along with improvements in glycemic control, triglycerides, and non-HDL cholesterol. With its once-weekly dosing, Retatrutide offers both potent efficacy and patient convenience, setting a new benchmark for comprehensive metabolic correction. Its broad therapeutic reach extends beyond obesity to include type 2 diabetes and metabolic-associated steatohepatitis (MASH), reflecting its capacity to modify underlying disease biology rather than treat surface manifestations.
Currently in advanced Phase III development, Retatrutide has demonstrated strong safety and efficacy signals across multiple metabolic indications, positioning it for regulatory submissions in key global markets. As one of the most advanced multi-agonist incretin therapies, it epitomizes the next evolutionary step in metabolic innovation-one that merges weight-loss potency, cardiometabolic protection, and durable clinical outcomes. With escalating global obesity prevalence and expanding payer recognition of obesity as a chronic disease, Retatrutide is strategically positioned to reshape treatment paradigms and cement Eli Lilly’s leadership in the rapidly evolving obesity and metabolic therapeutics landscape.
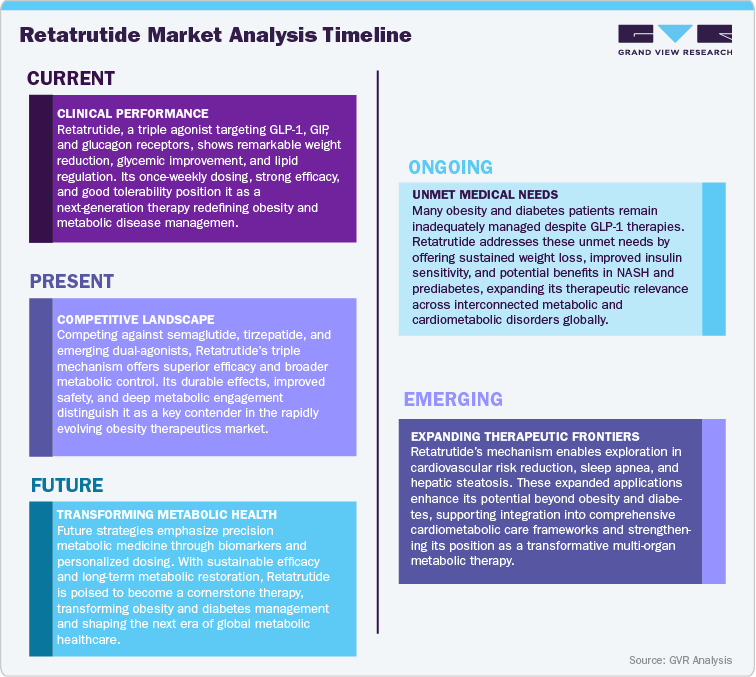
Current Market Scenarios
The global obesity and metabolic disorder therapeutics landscape is undergoing a profound transformation, fueled by increasing disease prevalence, growing awareness of obesity as a chronic condition, and expanding access to pharmacologic interventions. Despite the success of existing GLP-1 receptor agonists, many patients continue to face limited treatment durability and incomplete metabolic control, underscoring the need for next-generation therapies that target multiple hormonal pathways simultaneously. In this evolving market, Retatrutide (LY-3437943), developed by Eli Lilly and Company, stands out as a first-in-class triple agonist that engages the GLP-1, GIP, and glucagon receptors to deliver comprehensive metabolic restoration. By enhancing insulin sensitivity, promoting energy expenditure, and improving lipid metabolism, Retatrutide addresses the core drivers of obesity and related cardiometabolic diseases, offering the potential for true disease modification rather than symptomatic relief.
Clinical studies have demonstrated exceptional efficacy, showcasing meaningful and durable reductions in body weight, improved glycemic control, and favorable effects on lipid parameters and cardiometabolic health. The therapy’s once-weekly administration and strong safety profile enhance patient adherence and clinical appeal, overcoming limitations associated with earlier incretin-based approaches. Healthcare professionals increasingly view Retatrutide as a breakthrough metabolic therapy, capable of delivering sustained therapeutic outcomes across obesity, type 2 diabetes, and metabolic-associated steatohepatitis (MASH) populations.
Looking forward, Retatrutide is poised to redefine the global obesity and cardiometabolic therapeutics market, supported by Eli Lilly’s established leadership in incretin innovation and its expanding metabolic pipeline. As Phase III development progresses toward regulatory submissions, Lilly’s focus on real-world outcomes, payer engagement, and global market expansion will shape the therapy’s commercial trajectory. With rising patient demand, evolving treatment guidelines, and a strong shift toward multi-agonist biology, Retatrutide is strategically positioned to transform the metabolic disease landscape, improve long-term patient outcomes, and establish a new benchmark in precision metabolic medicine.
Mechanism of Action: Multi-Receptor Incretin Agonist for Comprehensive Metabolic Restoration
Retatrutide (LY-3437943) is a first-in-class triple agonist that simultaneously targets the GLP-1, GIP, and glucagon receptors, representing a next-generation approach to obesity and metabolic disease management. These interconnected pathways regulate glucose homeostasis, lipid metabolism, energy balance, and appetite control-key processes often disrupted in obesity, type 2 diabetes, and cardiometabolic disorders. By integrating signaling across these three hormonal axes, Retatrutide delivers a synergistic effect that enhances insulin sensitivity, accelerates energy expenditure, and promotes sustained weight loss, addressing the root causes of metabolic dysfunction rather than providing isolated symptomatic relief.
Activation of GLP-1 and GIP receptors enhances glucose-dependent insulin secretion, suppresses appetite, and improves postprandial glucose metabolism, while glucagon receptor stimulation promotes fatty acid oxidation and thermogenesis, contributing to improved lipid utilization and energy balance. This multi-pathway modulation drives significant improvements in glycemic control, lipid profile, and cardiometabolic health, resulting in a holistic restoration of metabolic equilibrium. Retatrutide’s once-weekly dosing ensures sustained receptor engagement, leading to consistent therapeutic benefits and enhanced patient adherence compared to existing incretin-based therapies.
Retatrutide’s mechanism of action distinctly differentiates it from traditional single-pathway incretin agonists. Unlike GLP-1-only or dual GIP/GLP-1 agents, its inclusion of glucagon receptor activity extends its metabolic reach, enabling simultaneous regulation of energy expenditure, lipid oxidation, and glycemic control. This unique tri-agonist balance optimizes both central and peripheral metabolic pathways, providing superior efficacy and durability. By addressing the multifactorial nature of metabolic disease, Retatrutide positions itself as a transformative therapeutic platform-one capable of delivering comprehensive metabolic correction, cardiometabolic protection, and long-term clinical benefit. As a result, it stands at the forefront of the next wave of multi-agonist metabolic innovations, redefining treatment possibilities across obesity and related metabolic disorders.
Clinical Development and Trial Insights
Retatrutide (LY-3437943) has demonstrated remarkable clinical progress across multiple studies, underscoring its strong efficacy, safety, and therapeutic potential in the management of obesity, type 2 diabetes, and related cardiometabolic disorders. In a pivotal Phase II trial, Retatrutide exhibited robust metabolic efficacy, achieving clinically meaningful weight reduction, improved glycemic control, and significant enhancements in lipid metabolism. Participants experienced favorable shifts in insulin sensitivity, non-HDL cholesterol, and triglyceride levels, accompanied by reductions in inflammatory biomarkers. Adverse events were predominantly mild to moderate and consistent with the incretin class, affirming a favorable safety and tolerability profile. These outcomes highlight Retatrutide’s potential to surpass existing incretin-based therapies by delivering comprehensive metabolic restoration rather than single-pathway modulation.
Building upon these promising results, Eli Lilly has advanced Retatrutide into an extensive Phase III clinical program designed to confirm its long-term efficacy, durability, and cardiovascular benefits. Ongoing studies are evaluating Retatrutide across diverse patient populations, including those with obesity, type 2 diabetes, and metabolic-associated steatohepatitis (MASH), with endpoints focused on sustained weight management, cardiometabolic improvement, and safety consistency over extended treatment durations. Its once-weekly dosing regimen further enhances convenience and adherence, offering a practical and scalable solution for chronic metabolic disease management.
Collectively, clinical evidence positions Retatrutide as a next-generation metabolic therapy with the potential to redefine obesity and cardiometabolic treatment paradigms. Its multi-agonist mechanism, coupled with consistent efficacy and tolerability, sets a new benchmark for incretin-based innovation. As Lilly progresses toward regulatory submissions and global commercialization, Retatrutide’s strong clinical foundation, strategic pipeline integration, and broad therapeutic scope make it one of the most anticipated metabolic launches of the decade-poised to deliver durable patient outcomes and reshape the future of precision metabolic medicine.
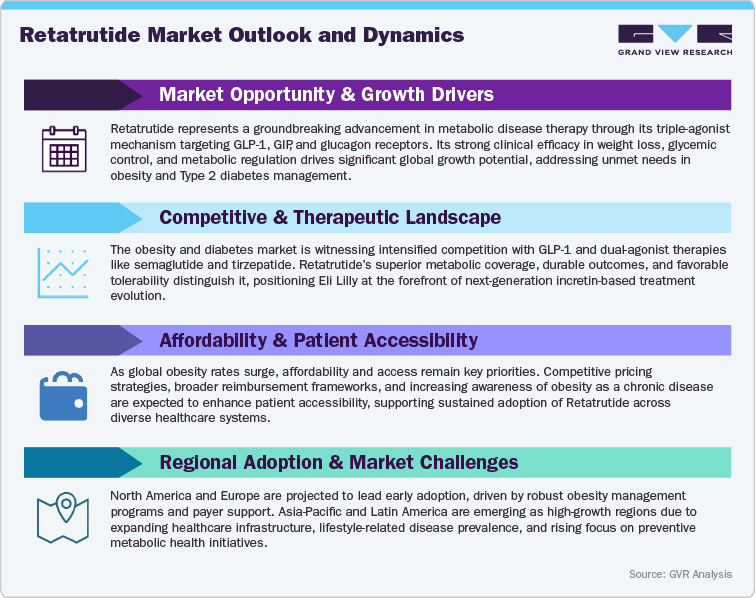
Regulatory Milestones and Commercial Readiness
Retatrutide (LY-3437943) has made substantial regulatory strides, reflecting its strong potential as a first-in-class triple-agonist therapy for obesity, type 2 diabetes, and metabolic-associated disorders. Following robust results from its Phase II clinical program, Eli Lilly has initiated an extensive Phase III trial series designed to validate Retatrutide’s long-term efficacy, cardiovascular safety, and metabolic benefits. These pivotal studies will form the foundation for global regulatory submissions to the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and leading health authorities across Asia-Pacific and Latin America. Given the therapy’s differentiated mechanism and clinically meaningful weight-loss outcomes, regulatory agencies are expected to extend priority or fast-track designations, potentially expediting Retatrutide’s approval timeline and market entry.
Lilly’s regulatory strategy emphasizes a coordinated, global submission framework to streamline approvals and accelerate patient access. The company has engaged in proactive discussions with regulatory bodies to align on key trial endpoints, cardiometabolic biomarkers, and real-world evidence integration. Parallel submissions are being planned across major markets, supported by Retatrutide’s strong safety data and its consistency across diverse populations. These harmonized efforts underscore Lilly’s commitment to delivering a transformative therapeutic option within the rapidly expanding obesity and metabolic care ecosystem.
Commercial readiness for Retatrutide is being bolstered by strategic manufacturing expansion, supply chain optimization, and market access preparation. Eli Lilly is leveraging its established global infrastructure and successful experience with incretin-based therapies such as Tirzepatide (Mounjaro/Zepbound) to ensure rapid scalability and global distribution. Pre-launch initiatives include medical education programs, payer engagement, and healthcare professional outreach to strengthen clinical adoption and formulary inclusion. Retatrutide’s once-weekly dosing, triple-hormone receptor activation, and sustained efficacy make it a compelling proposition for healthcare systems seeking durable, patient-centric metabolic interventions.
Market Dynamics and Growth Drivers
The global Retatrutide (LY-3437943) market is gaining remarkable momentum, propelled by the rising prevalence of obesity, type 2 diabetes, and metabolic-associated disorders, along with increasing clinical focus on multi-hormonal therapeutic strategies. With obesity now recognized as a chronic and multifactorial disease, there is a surging demand for safe, durable, and mechanism-based treatments that extend beyond traditional weight management solutions. Retatrutide, developed by Eli Lilly and Company, is emerging as a transformative triple agonist therapy, simultaneously targeting GLP-1, GIP, and glucagon receptors to deliver unprecedented improvements in weight reduction, glycemic control, and cardiometabolic health. This multifaceted mechanism positions Retatrutide at the forefront of a new generation of incretin-based therapies designed to combat global metabolic dysfunction.
A major growth driver for the Retatrutide market is the unmet need for comprehensive metabolic control in patients inadequately managed by existing GLP-1 or dual-agonist treatments. Current drugs often plateau in efficacy or are associated with tolerability challenges, leaving room for next-generation therapies that achieve deeper and more sustained weight loss. Retatrutide’s unique triple-receptor activation enables simultaneous modulation of appetite, energy expenditure, and glucose metabolism, translating into superior and long-lasting clinical outcomes. Its once-weekly dosing, robust safety profile, and broad metabolic impact make it highly appealing to endocrinologists, obesity specialists, and cardiologists seeking an advanced, mechanism-driven intervention for complex metabolic disorders.
Furthermore, the global paradigm shift toward obesity as a treatable metabolic disease is accelerating market expansion. Governments, payers, and healthcare providers are increasingly investing in chronic weight management programs, supporting reimbursement policies for advanced incretin therapies. Strategic collaborations among pharmaceutical innovators, academic researchers, and public health institutions are strengthening evidence-based adoption, while public awareness campaigns emphasize the long-term health and economic benefits of effective weight control.
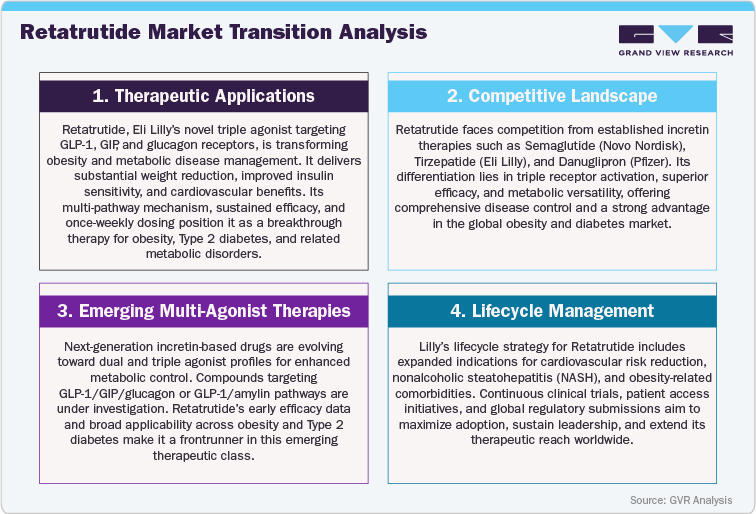
Competitive Landscape: Retatrutide vs. Existing Obesity Therapies and Emerging Incretin Agonists
Retatrutide (LY-3437943) is redefining the competitive landscape of obesity and metabolic disease therapeutics through its unique triple-agonist mechanism, exceptional clinical efficacy, and superior tolerability profile. Developed by Eli Lilly and Company, Retatrutide simultaneously activates GLP-1, GIP, and glucagon receptors, orchestrating a comprehensive modulation of appetite regulation, energy expenditure, and glucose metabolism. This triple-receptor synergy addresses multiple facets of obesity, type 2 diabetes, and cardiometabolic dysfunction, distinguishing Retatrutide as a next-generation therapy that goes beyond the scope of existing incretin-based treatments such as semaglutide (Wegovy, Ozempic) and tirzepatide (Mounjaro, Zepbound), which primarily target one or two hormonal pathways.
In head-to-head comparisons and clinical observations, Retatrutide has demonstrated superior weight reduction, improved glycemic control, and enhanced lipid metabolism, achieving deeper and more sustained therapeutic responses. Unlike earlier GLP-1 or dual agonists, Retatrutide’s engagement of the glucagon receptor promotes increased energy expenditure and fat oxidation, resulting in greater total body weight loss and metabolic restoration. Its once-weekly administration offers high patient convenience, ensuring consistent compliance while maintaining potent, long-duration efficacy. Early-phase clinical data highlight significant reductions in body mass index (BMI), HbA1c, and visceral fat, underscoring its transformative potential across both obese and non-diabetic metabolic populations.
As the incretin therapy market becomes increasingly competitive, several next-generation agents are entering development, including danuglipron (Pfizer), orforglipron (Eli Lilly), and CT-388 (Cytokinetics). However, few have demonstrated the triple-pathway synergy and metabolic versatility seen with Retatrutide. Its broad applicability across obesity, nonalcoholic steatohepatitis (NASH), and cardiometabolic disorders creates a strong clinical and commercial advantage. With a favorable safety profile, high patient adherence, and multi-system efficacy, Retatrutide is well positioned to lead the next wave of metabolic innovation, shaping the competitive future of incretin-based and multi-agonist therapies.

Opportunities and Strategic Challenges
The Retatrutide (LY-3437943) market presents immense opportunities amid the global push for innovative obesity and metabolic disease treatments. As a first-in-class triple agonist targeting GLP-1, GIP, and glucagon receptors, Retatrutide introduces a new paradigm in metabolic restoration, offering deeper and more durable outcomes than current incretin-based therapies. Rising obesity prevalence, coupled with increasing recognition of obesity as a chronic, treatable condition, is fueling strong clinical and commercial interest. The therapy’s broad metabolic impact spanning weight loss, glycemic control, lipid regulation, and energy expenditure enhancement positions Retatrutide as a transformative candidate for both diabetic and non-diabetic populations. Backed by Eli Lilly’s proven expertise in incretin therapeutics, Retatrutide is strategically positioned to lead the next generation of multi-agonist therapies redefining obesity and cardiometabolic care.
Despite its breakthrough potential, Retatrutide faces notable strategic challenges on its path to commercialization. Regulatory alignment, long-term cardiovascular safety validation, and manufacturing scalability will be key to sustaining growth and ensuring global readiness. While clinical results show exceptional efficacy, robust real-world evidence will be critical for establishing payer confidence and long-term adoption. Additionally, the expanding competition from dual agonists (tirzepatide, orforglipron) and emerging pan-incretin modulators necessitates clear differentiation in clinical positioning and value communication. Pricing and reimbursement strategies must balance premium efficacy with accessibility, particularly in regions facing obesity-related healthcare burdens.
To maintain leadership, Eli Lilly must execute a focused strategy emphasizing clinical superiority, global scalability, and patient-centered innovation. Leveraging Retatrutide’s triple-receptor mechanism and once-weekly convenience, the company can strengthen its position as a frontrunner in metabolic therapeutics. Expansion into adjacent indications such as type 2 diabetes, NASH, and cardiometabolic disorders-combined with strategic partnerships for manufacturing, distribution, and medical education, will further enhance reach and adoption. By aligning data-driven commercialization with global healthcare priorities, Retatrutide is poised to become a cornerstone therapy in the evolving multi-agonist obesity market, driving sustained growth and reshaping the future of metabolic medicine.
Market Forecast and Future Outlook (2025-2033)
The Retatrutide (LY-3437943) market is poised for robust expansion between 2025 and 2033, fueled by the surging global prevalence of obesity, type 2 diabetes, and related cardiometabolic disorders. With pivotal Phase 3 clinical trials currently advancing, topline data expected in 2026-2027 are anticipated to support regulatory submissions across key markets including the United States, Europe, and Asia-Pacific. Based on current development milestones, the commercial launch of Retatrutide is projected around 2027-2028, positioning it as a flagship therapy within the emerging class of multi-agonist incretin drugs and a potential game-changer in the global metabolic therapeutics landscape.
Following its introduction, rapid adoption is expected among endocrinologists, obesity specialists, and cardiometabolic clinicians, driven by its triple-agonist mechanism, once-weekly dosing, and unprecedented efficacy in achieving weight reduction, glycemic control, and metabolic restoration. Its favorable safety and tolerability profile, combined with consistent clinical outcomes, will support strong uptake as healthcare systems prioritize disease-modifying treatments and long-term cost savings. Post-approval data and real-world evidence will further reinforce Retatrutide’s clinical value, expanding confidence among prescribers, payers, and patients worldwide.
By 2033, Retatrutide is expected to capture a significant share of the global obesity and metabolic therapeutics market, potentially outpacing earlier incretin agents. Strategic alliances, regional commercialization partnerships, and value-based reimbursement models will accelerate access and adoption across major geographies. Moreover, ongoing pipeline expansion into adjacent indications such as NASH, type 2 diabetes, and cardiovascular risk reduction is set to broaden its clinical and commercial footprint. As healthcare shifts toward integrated metabolic care and mechanism-driven therapies, Retatrutide is strategically positioned to redefine the standard of obesity management-delivering durable efficacy, improved patient quality of life, and shaping the next era of metabolic innovation worldwide.

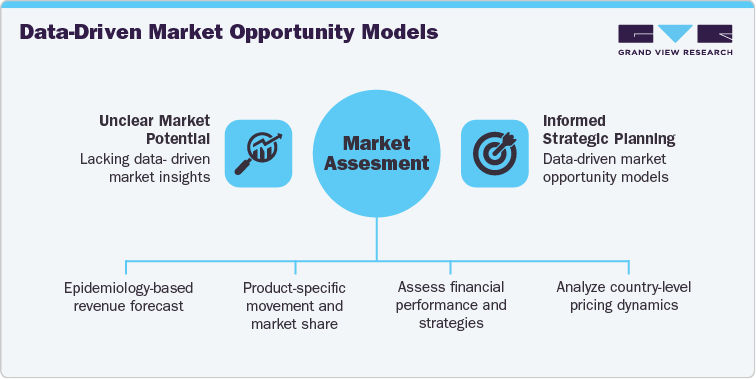
Case Study (Recent Engagement): GLP-1 Receptor Agonist Market Opportunity Assessment
Project Objective
A leading global life sciences client approached us to assess the market potential and commercialization strategy for GLP-1 receptor agonist therapies across type 2 diabetes and obesity indications. The project aimed to support strategic planning for a novel, oral GLP-1 pipeline candidate, with a focus on launch timing, competitive positioning, and regional expansion.
GVR Solution
-
Conducted an epidemiology-based revenue forecast (2021-2036) using patient flow and analogue modeling approaches across North America, Europe, Asia Pacific, and the Middle East.
-
Delivered product-specific movement and market share analysis for:
-
Tirzepatide (Mounjaro)- used as a reference analogue for uptake modeling
-
Orforglipron (pipeline)- projected using analogue-based scenarios from comparable oral GLP-1 launches
-
-
Benchmarked key players such as Eli Lilly and Novo Nordisk across financial performance, product pipeline, and global rollout strategies.
-
Assessed country-level pricing, regulatory, and reimbursement dynamics, supported by a custom launch timeline and uptake forecast for Orforglipron, modeled analogously to prior GLP-1 innovations.
-
Provided outputs (Excel, PPT, dashboard) and ongoing strategic support tailored to the client’s internal planning and commercialization team needs.
Impact for Client
-
Created market models for launch planning and portfolio prioritization.
-
Guided product strategy with pricing, uptake, and competitor insights.
-
Identified growth markets and shaped regulatory and launch plans.
Why this Matters
-
Build analogue-based forecasts for emerging therapies
-
Provide product insights for pipeline drugs with no historical sales
-
Offer strategic guidance on market entry, launch, and clinical-commercial integration
We bring the same level of analytical rigor, therapeutic market expertise, and consultative flexibility to your assessment of the pancreatic cancer microbubble-based therapy market.
Analyst Perspective
Retatrutide (LY-3437943) is emerging as a groundbreaking innovation in obesity and metabolic disease treatment, introducing a first-in-class triple agonist mechanism that activates GLP-1, GIP, and glucagon receptors. This unique approach enhances satiety, boosts energy expenditure, and improves metabolic regulation, addressing the core drivers of obesity, insulin resistance, and dyslipidemia. Unlike single-pathway incretin therapies, Retatrutide offers comprehensive metabolic benefits with superior weight reduction, glycemic control, and cardiometabolic improvements. Its once-weekly dosing and favorable safety and tolerability profile make it a next-generation therapy poised to transform obesity management and redefine standards in metabolic care.
Clinical results reinforce Retatrutide’s potential as a best-in-class therapy. Promising findings from Phase 2 studies demonstrated remarkable improvements in body weight, HbA1c, and lipid parameters, showcasing its broad therapeutic impact. With Phase 3 trials advancing rapidly, Eli Lilly’s strong development strategy, regulatory planning, and global launch initiatives are expected to drive rapid commercialization across key markets including the United States, Europe, and Asia-Pacific. Retatrutide is well-positioned to serve both treatment-naïve individuals and those with inadequate responses to GLP-1 or dual-agonist therapies, strengthening its leadership in the expanding incretin-based therapy segment.
With the global burden of obesity and metabolic disorders rising sharply, Retatrutide’s value extends beyond weight management to achieving holistic metabolic restoration. Analysts project strong market uptake following approval, supported by its exceptional efficacy, durable outcomes, and broad applicability across obesity, type 2 diabetes, and cardiometabolic disease. As the most advanced triple agonist therapy in development, Retatrutide is set to establish a new era in metabolic medicine, delivering sustained benefits, improved patient outcomes, and reshaping the future of global metabolic health.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


