Dental Implant Adoption in MEA: Bridging Clinical Need, Policy Shifts, And Market Potential
Executive Summary: The Strategic Pivot
The Middle East & Africa dental implant market is at a critical inflection point. Market growth is no longer driven primarily by implant unit volumes or brand proliferation, but by the depth of digital integration, localization, and clinical enablement delivered to dental providers.
Leading market participants have fundamentally repositioned dental implants from a standalone product sale to a digitally enabled clinical solution, embedded within a broader treatment ecosystem. This shift reflects a structural reality: clinical predictability, workflow efficiency, and patient experience now determine market success, not hardware differentiation alone.
Key Strategic Shift in the Implant Value Proposition
|
Old Model |
New Model |
|
Implant + abutment supply |
End-to-end digital implant ecosystem |
|
Distributor as logistics agent |
Distributor as Clinical Enablement Partner |
|
Imported kits |
Localized assembly + service |
|
Chairside dependency |
Cloud-based AI planning + centralized milling |
Strategic Implication: Dental implants are increasingly treated as workflow anchors, not discrete SKUs. Stakeholders that fail to transition toward service-led, workflow-centric models face accelerated commoditization and sustained margin erosion.
Ecosystem Evolution: What Leading Distributors Are Integrating
Across the UAE and Saudi Arabia, leading distributors have already redefined their role within the dental value chain. Rather than competing on product availability alone, they are embedding capability layers that directly influence clinical outcomes and adoption rates.
Key integrations include:
-
AI-driven treatment planning, improving predictability and case acceptance
-
CBCT imaging and facial scanning, enabling diagnostic precision and aesthetic planning
-
Local 3D milling and CAD/CAM hubs, reducing turnaround times and clinic CAPEX
-
Accredited surgical education and certification, building long-term clinician confidence and loyalty
-
Post-market technical support and repair, ensuring regulatory compliance and operational uptime
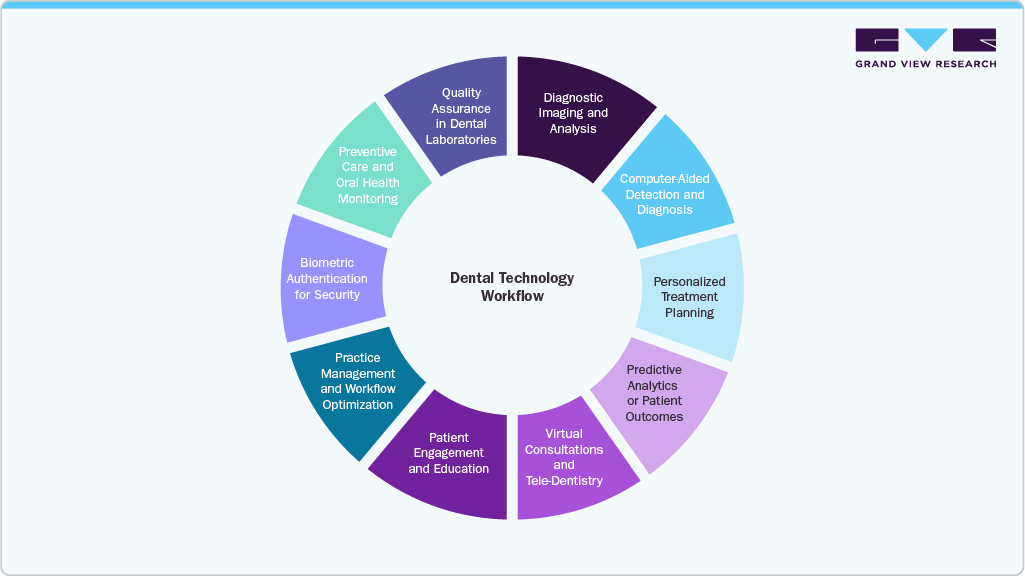
Outcome: Distributors have transitioned from cost centers to strategic clinical partners, capturing recurring value across the implant lifecycle.
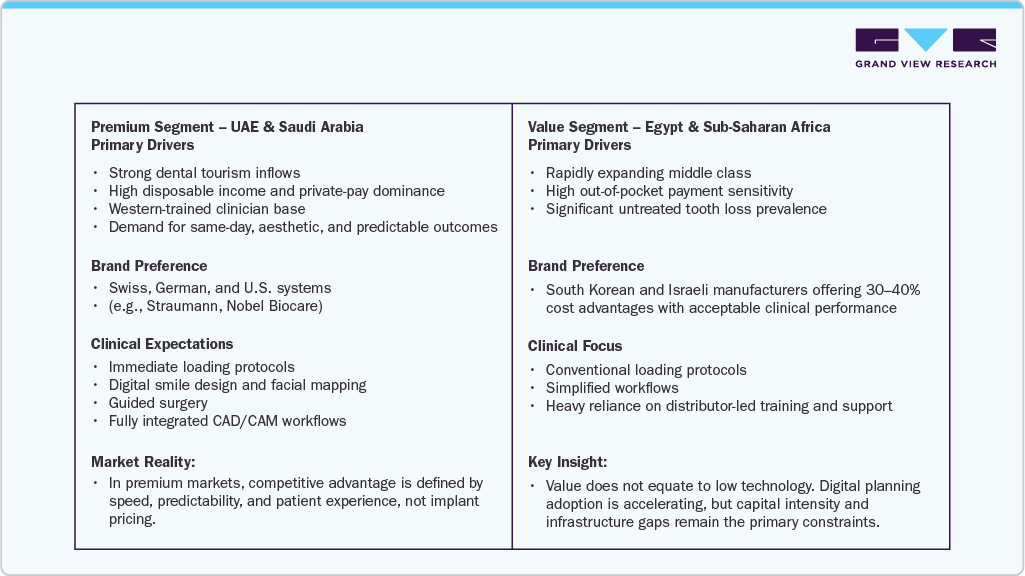
Market Dynamics: The Dual-Market & Digital Revolution: The MEA implant market functions as a two-speed ecosystem, defined by income stratification, reimbursement realities, and digital maturity
UAE
Digital Leadership & Market Modernization
- The UAE has positioned itself as a global benchmark for digitally enabled dentistry, driven by regulation, tourism, and private-sector investment.
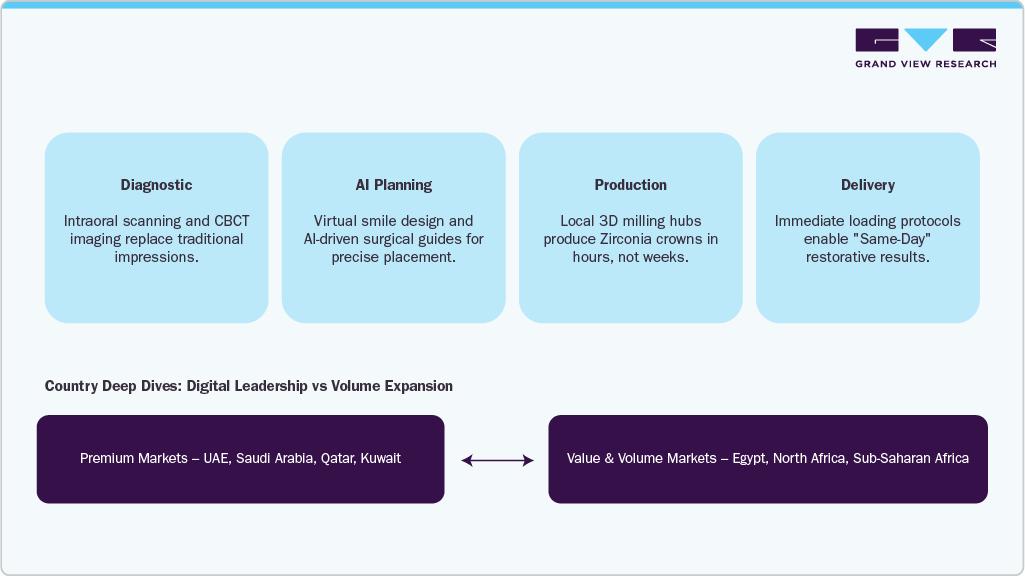
Digital Dentistry Framework
CBCT & Facial Scan → AI Planning → Guided Surgery → Same-Day Prosthetics → Virtual Follow-Up
This closed-loop workflow has materially improved case acceptance rates, clinical outcomes, and patient satisfaction.
Key Institutional & Commercial Initiatives
|
Entity |
Timeline |
Strategic Action |
Market Impact |
|
Dubai Health Authority |
Feb 2025 |
Smart Dental Initiative mandating AI-assisted planning |
Standardized digital care |
|
Zahrawi Group / Straumann |
May 2025 |
Same-day implant workflows |
Faster patient conversion |
|
Mediclinic Middle East (Mediclinic Middle East operates six hospitals in the UAE with over 900 inpatient beds) |
Aug 2025 |
Virtual smile design via mobile apps |
Improved patient engagement |
|
Metromed |
Q3 2025 |
Centralized zirconia milling hub |
Reduced clinic CAPEX |
Strategic Meaning: UAE distributors are monetizing speed, aesthetics, and digital confidence, not just implants.
Saudi Arabia
Vision 2030, Localization & Volume Leadership
- Saudi Arabia is the largest MEA implant market by volume, shaped by Vision 2030, localization mandates, and public-sector scale.
Localization & Digital Health Stack
Global OEM → Local Assembly → SFDA Compliance → Distributor-Led Training → Rural Tele-Dentistry
|
Entity |
Timeline |
Strategic Action |
Vision 2030 Link |
|
Saudi Food & Drug Authority |
Jan 2025 |
Mandatory local training & repair |
Workforce localization |
|
Tamer Group / Nobel Biocare |
Jun 2025 |
Local surgical kit assembly |
Local content targets |
|
SEHA Virtual Hospital |
Sep 2025 |
5G-enabled tele-dentistry |
Rural access expansion |
|
Al Jeel Medical |
Oct 2025 |
CBCT rollout in 50 centers |
Diagnostic modernization |
Strategic Meaning:
In Saudi Arabia, regulatory compliance capability combined with a local operational footprint has become a durable competitive moat.
Egypt
Price-Sensitive Scale & Training-Led Adoption
-
Largest patient pool in North Africa
-
Strong demand for mid-priced implant systems
-
Distributor-led education is the primary adoption lever
-
Government price controls slow premium brand penetration
Strategic reality: Egypt is a volume multiplier, not a margin leader—success depends on training density and portfolio breadth.
South Africa
Private-Sector Anchor for Sub-Saharan Africa
-
Concentrated private dental chains in urban centers
-
Strong regulatory discipline (SAHPRA)
-
Acts as a gateway market for regional expansion
Strategic reality: Moderate growth, high clinical rigor, strong aftermarket opportunity.
Qatar & Kuwait
Premium, Low-Volume, High-ARPU Markets
-
High per-procedure spend
-
Rapid adoption of digital workflows
-
Limited local training → dependence on distributor-OEM partnerships
Strategic reality: Ideal for pilot launches and premium protocol validation.
Regulatory & Compliance Quick View
|
Country |
Avg. Approval (Months) |
Local Mandates |
Post-Market Burden |
|
Saudi Arabia (SFDA) |
9–12 |
High (Localization Focus) |
Intensive Clinical Data |
|
UAE (MoHAP) |
4–6 |
High (Digital Compliance) |
Moderate |
|
Egypt (EDA) |
10–14 |
High (Price Controls) |
High |
|
South Africa (SAHPRA) |
8–10 |
Moderate |
Moderate |
Major Dental Implant Distributors in MEA
|
Country |
Distributor |
Product Focus |
Key Partnerships |
|
|
Saudi Arabia |
Tamer Group |
1922 |
Biomaterials, Surgical Kits |
Nobel Biocare, Geistlich |
|
Saudi Arabia |
Al Jeel Medical |
1975 |
Imaging, Diagnostics |
GE Healthcare, Carestream |
|
UAE |
Zahrawi Group |
1989 |
Surgical, Aesthetic |
Straumann, Medtronic |
|
UAE |
Metromed |
1990 |
Restorative, Lab Tech |
50+ Global Brands |
|
Kuwait |
ATCO Technology |
2006 |
Consumables, Equipment |
Global Restorative Brands |
|
Pan-Africa |
Dental Warehouse |
Legacy |
Full Restorative Suite |
Dentsply Sirona, 3M |
Conclusion: From Hardware to Healthcare Platforms
The MEA dental implant market is no longer about selling screws—it is about enabling outcomes.
Winning Formula
-
Digital-first workflows
-
Localized manufacturing & service
-
Regulatory readiness
-
Continuous clinical education
-
Integrated patient experience
Distributors that evolve into Clinical Enablement Partners—bridging technology, training, and localization—will define the next decade of dental innovation in MEA.
The future of MEA dental implants is not imported—it is digitally built, locally enabled, and clinically empowered.