Clinical Laboratory Services & Supplies in the Middle East: Accelerating the Next Diagnostic Era
Clinical laboratory services and supplies in the Middle East are entering a phase of significant transformation, driven by evolving healthcare priorities, increased demand for diagnostic accuracy, expansion of molecular and genomic capabilities, and increasing opportunities around operational efficiency and supply chain resilience. Laboratory operations, once treated as supporting functions within hospitals, have now become critical enablers of clinical performance, healthcare modernization, and national health objectives. The transformation taking place across the region reflects a structural shift rather than incremental change, and it is reshaping the competitive and operational landscape for laboratories, suppliers, and healthcare providers.
Strategic Importance of Diagnostics in the Regional Healthcare Landscape
The growing application of clinical laboratories in the Middle East is linked directly to the region’s shifting disease burden, modernization agendas, and increased reliance on data-driven care delivery. Rising prevalence of chronic diseases, particularly diabetes, cardiovascular disease, oncology, endocrine disorders, autoimmune conditions, and infectious disease outbreaks, has expanded the volume and complexity of laboratory testing. More than any other healthcare function, laboratory services influence diagnostic timelines, treatment decisions, and patient outcomes.
Governments across the region have embedded diagnostics into broader health transformation programs, giving laboratories a clearly defined role in supporting system-wide efficiency and standardization. As public and private healthcare networks aggregate into cluster structures or integrated delivery systems, laboratories are expected to provide consistent quality, harmonized reporting, and scalable testing capability. This has elevated diagnostics from decentralized units operating independently toward organized, enterprise-wide diagnostic platforms.
Furthermore, the shift toward precision medicine, genomics, biomarker oncology testing, and rare disease identification is introducing diagnostic categories that require specialized laboratory environments and structured supply chain management. Laboratories now sit at the intersection of clinical care, research, regulatory compliance, digital infrastructure, and operational governance positioning them as strategic assets within national health systems.
Market Size and Momentum
The Middle East medical laboratory services & supplies market was valued at USD 7 billion in 2025 and is projected to reach around USD 10 billion by 2033, reflecting a CAGR of more than 5%. A critical growth layer comes from integrating advanced medical diagnostics and imaging services, as countries in the region pivot toward precision medicine, early disease detection, and digital health ecosystems.
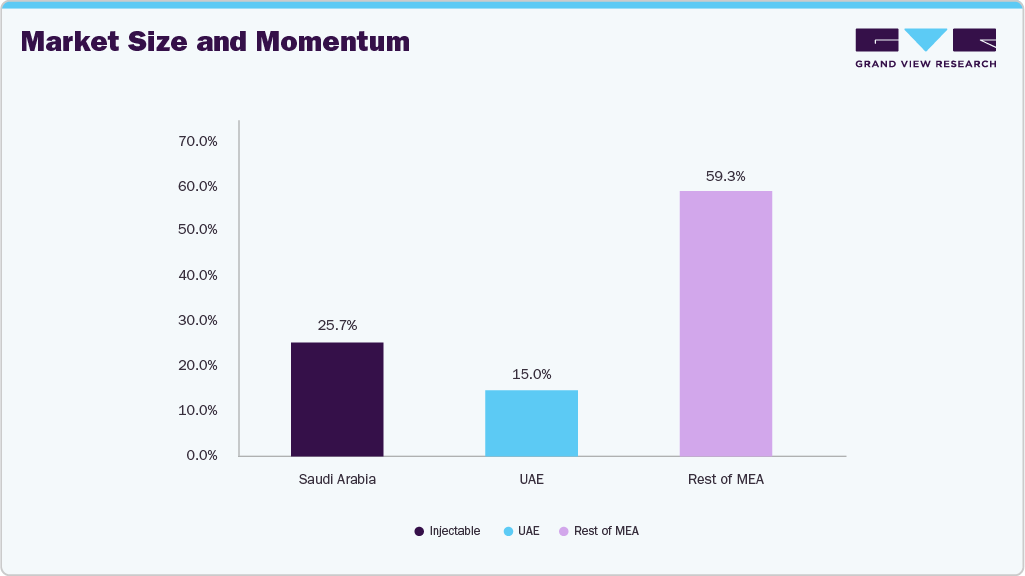
Together, the UAE and Saudi Arabia account for nearly 40% of the Middle East laboratory services market, shaping the region’s future through a mix of public investment, private sector expansion, AI-driven imaging, and digital health integration. Their strategies are increasingly aligned with global trends, positioning the Middle East as a rising hub for clinical excellence, advanced imaging, and diagnostics linked to medical tourism.
Evolution of Laboratory Service Capabilities and Operating Models
Laboratory services in the Middle East have expanded considerably across technical, operational, and organizational dimensions. Modern laboratories in the region are expected to support both routine and advanced diagnostic workflows with high degrees of accuracy, reproducibility, and throughput.
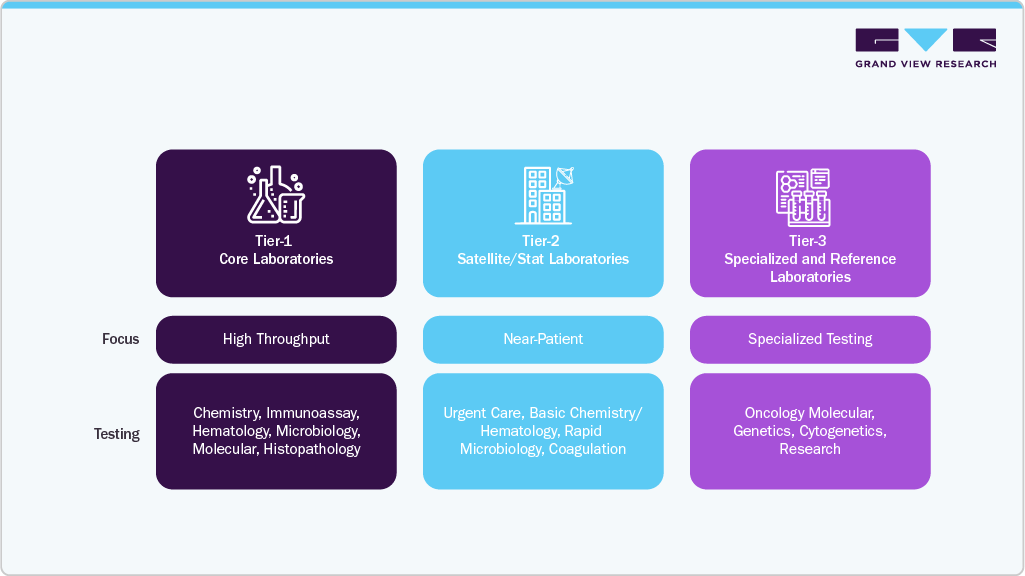
Expansion Across Diagnostic Disciplines: Most large laboratories now operate across a broad diagnostic spectrum that includes clinical chemistry, immunoassay, hematology, coagulation, microbiology, histopathology, cytology, molecular diagnostics, genomics, cytogenetics, and advanced immunology. The shift toward high-complexity diagnostics, such as sequencing-based oncology testing, syndromic panels, AMR testing, and molecular pathology has elevated the scientific and operational sophistication of laboratory environments.
Integrated Multi-Site Laboratory Networks: Public and private systems are consolidating laboratory services across hospitals, outpatient facilities, and specialty centers. These integrated structures support:
-
Unified test menus and workflows
-
Standardized operating procedures
-
Cross-facility resource utilization
-
Centralized quality management and oversight
-
Streamlined clinical reporting and interface consistency
Operational integration also enables laboratories to centralize high-volume testing within core facilities, while maintaining satellite laboratories for urgent or near-patient requirements. This model increases capacity, reduces duplication, and allows laboratories to operate at scale with greater cost efficiency.
Technological Modernization and Digital Enablement: Laboratories across the region are upgrading instrumentation, automation platforms, LIS/LIMS systems, and analytics tools. Investments in automated pre-analytical systems, integrated analyzers, microbiology automation, and digital pathology platforms are improving turnaround times, reducing error rates, and enabling sustained performance under growing demand.
Digital enablement is also reshaping laboratory operations. Modern LIS architectures support rule-based verification, real-time monitoring, cumulative result trending, clinical analytics, accreditation compliance tracking, and integration with national EHR systems. Laboratories are increasingly expected to provide operational transparency and clinical intelligence, reinforcing their role within broader digital health strategies.
Strengthened Quality, Accreditation, and Regulatory Frameworks: Accreditation is now standard practice among leading laboratories. ISO 15189 frameworks, along with international accreditation schemes, are driving improvements in personnel competency, method validation, QC management, traceability, and continual improvement. Regulatory authorities across the GCC have increased oversight of molecular testing, genetic laboratories, proficiency testing participation, and data governance, reinforcing diagnostic quality and patient safety expectations.
Transformation of the Laboratory Supplies Ecosystem
While service capabilities define laboratory output, the supply-side environment determines the system’s stability, performance, and scalability. The Middle East’s clinical supply ecosystem is undergoing a substantial restructuring characterized by modernization, strategic sourcing, increased technical complexity, and a shift toward localization.
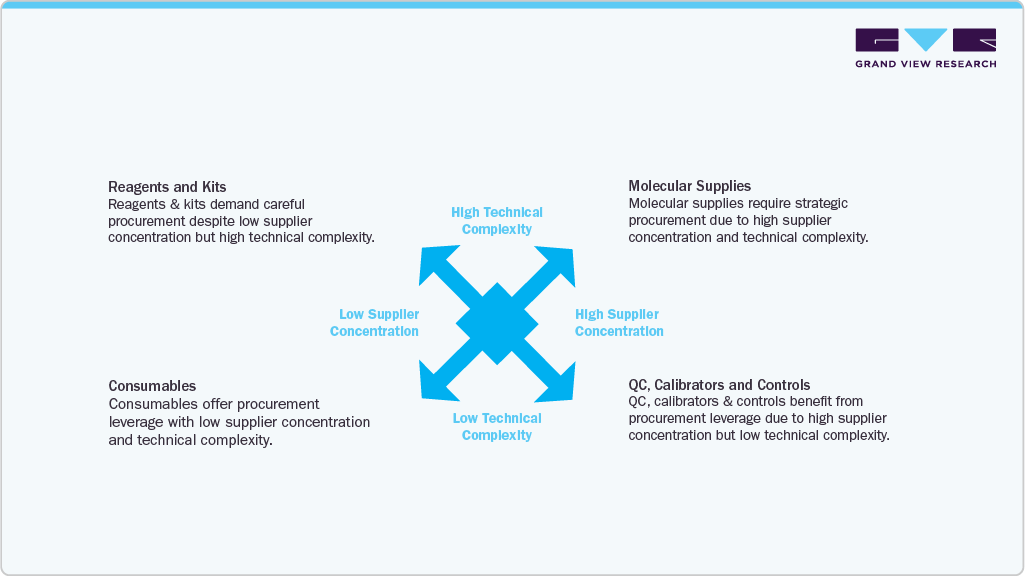
Supply Chain Maturity and Organizational Structure: Over the years, laboratory supplies were procured at a facility level, with fragmented vendor portfolios and inconsistent contract management. This model is no longer adequate for laboratories performing large volumes of high-complexity tests. Healthcare organizations are moving toward centralized procurement models that leverage enterprise-level contracts, structured supplier evaluations, consumption analytics, and governance frameworks to manage reagent performance and supply continuity.
These procurement models support:
-
Standardization of reagents and consumables across multi-site networks
-
Superior negotiating power with global diagnostic companies
-
Reduction of variability in test performance
-
Improved cost-per-test analysis and budgeting discipline
-
Enhanced traceability, inventory turnover, and demand forecasting
Increasing Technical Complexity of Supplies: The modernization of diagnostic capabilities has increased the technical specificity of supplies required to support laboratory operations. Reagents, controls, calibrators, molecular kits, sequencing consumables, and microbiology automation cards must meet stringent performance specifications and instrument compatibility requirements. Unlike general medical supplies, laboratory supplies must maintain precise quality stability, batch consistency, and environmental sensitivity, making supply reliability a critical operational factor.
The expansion of molecular and genomic testing has also elevated cold-chain requirements, batch validation processes, supplier quality monitoring, and technical support expectations. Laboratories now require structured reagent acceptance protocols, advanced QC materials, technical documentation, and software-driven tracking to support test integrity.
Localization and Regional Manufacturing Initiatives: Recognizing vulnerabilities associated with global supply chains, several Middle Eastern governments are promoting local manufacturing of fundamental laboratory consumables. This includes specimen tubes, transport media, swabs, basic reagents, and selected POCT materials. While highly specialized molecular reagents will remain globally sourced in the near term, growth in local assembly and reagent packaging is expected to enhance resilience and reduce lead times.
Supplier Partnerships and Performance Management: Laboratories increasingly select suppliers based on long-term partnership value rather than price alone. Supplier evaluation now considers:
-
Technical performance metrics
-
Instrument compatibility and reagent longevity
-
Delivery reliability and logistics capability
-
Local technical support availability
-
Responsiveness to QC issues and recalls
-
Total cost-of-ownership rather than unit pricing
-
Integration with automation and LIS platforms
These requirements are reinforcing a more structured, partnership-oriented supplier landscape across the region.
Technology Acceleration and Its Impact on Laboratory Services and Supplies
Technology is redefining both how laboratory services are delivered and how supplies are consumed, monitored, and sourced. Several areas are undergoing accelerated deployment:
Automation and High-Throughput Systems: Automated systems have become fundamental to laboratories facing increased demand and limited workforce availability. Automation platforms improve consistency, reduce manual touchpoints, and allow laboratories to scale operations while controlling labor costs. Automation also provides more predictable reagent consumption patterns, improving inventory management and procurement planning.
Molecular Diagnostics and Next-Generation Testing: The rapid expansion of PCR-based testing, targeted sequencing panels, and comprehensive multi-omics workflows is reshaping laboratory infrastructure and supply dependency. Molecular workflows require specialized consumables with strict performance attributes, influencing procurement strategies and technical support frameworks.
Digital Pathology and AI Integration: Digital pathology is gaining relevance as hospitals expand oncology services. Transitioning from manual microscopy to digitized slide workflows requires new categories of consumables, staining reagents, slide storage, and image management systems. AI algorithms integrated into digital pathology platforms provide quantification, classification, and triage assistance, improving diagnostic confidence and efficiency.
Data-Driven Operations and Predictive Analytics: Diagnostic data systems are evolving from simple report-generation tools to enterprise intelligence platforms. Through integration with LIS, inventory systems, automation platforms, and procurement portals, laboratories can forecast reagent consumption, predict equipment utilization, model turnaround times, and proactively manage resource allocation. This analytical capability reduces waste, improves performance reliability, and enhances financial transparency.
Future Outlook
Clinical laboratory services and supplies in the Middle East have progressed from operational necessities to strategic components of healthcare transformation. As the region invests in advanced diagnostics, integrated laboratory networks, supply chain resilience, and digital infrastructure, laboratories are assuming greater influence over clinical performance, cost efficiency, and system-wide quality.
The next phase of evolution will be defined by technological maturity, structured procurement and supply chain models, expanded diagnostic complexity, and elevated expectations for quality management. Laboratories will continue to drive national healthcare agendas through improved diagnostic accuracy, advanced scientific capabilities, and integration with digital health ecosystems.
The Middle East is well positioned to become a leader in next-generation diagnostics, backed by sustained investment, modernization initiatives, and a growing recognition of laboratories as essential pillars of clinical excellence and long-term healthcare sustainability.