The Globalization of Clinical Trials and the Rise of R&D Hubs in the Middle East: Accelerating Innovation, Access, and Investment
Global clinical development is experiencing a structural shift, driven by increasing R&D spending, stricter regulatory pathways in traditional markets, and heightened competition for patient recruitment. As costs rise and timelines extend in the U.S. and Europe, biopharmaceutical sponsors are exploring new geographies that can offer speed, operational excellence, and high-quality data. In this context, the Middle East is quickly emerging as a strategic hub for clinical trials and life sciences R&D. Led by Saudi Arabia, the United Arab Emirates, and Qatar, the region features advanced healthcare infrastructure, digital readiness, and a growing population supported by strong government-backed innovation initiatives. This combination is enabling the Middle East to shift from an underrepresented research destination to a competitive player in the global R&D network.
Historical Evolution of Global Clinical Trial Footprints
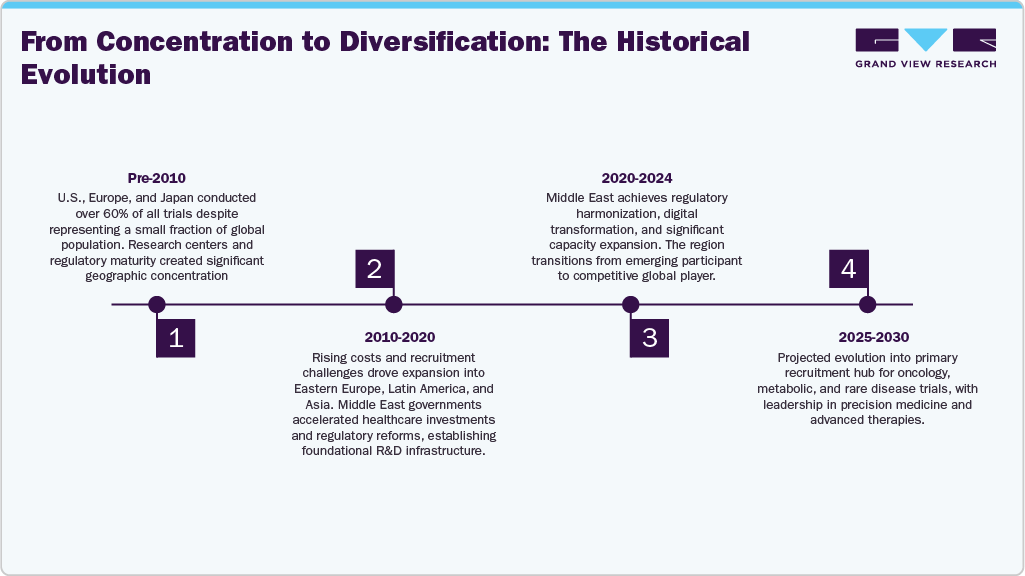
Over the past two decades, the distribution of global clinical trials has expanded significantly beyond traditional regions. Historically, the U.S., Europe, and Japan conducted more than 60 percent of all trials, despite representing only a small part of the world’s population. This imbalance, mainly caused by the concentration of research centers and regulatory maturity, continued into the early 2010s. However, rising development costs estimated at USD 2.6 billion per approved drug and decreasing recruitment rates drove a shift toward emerging markets. Countries in Eastern Europe, Latin America, and Asia began to take larger shares of Phase II and III studies, supported by growing investigator networks and treatment-naive patient pools. This wave of globalization paved the way for the Middle East to join the R&D landscape as governments accelerated investments in healthcare and regulatory reforms.
Historical Development of R&D Capacity in the Middle East (2010–2020)
Between 2010 and 2020, Middle Eastern countries laid the foundation for a highly developed research ecosystem. Regulatory agencies such as the Saudi Food and Drug Authority (SFDA), UAE Ministry of Health and Prevention (MOHAP), and Egypt’s Drug Authority (EDA) were strengthened with structured, internationally aligned frameworks. Clinical trial registries, centralized ethics committees, and the adoption of Good Clinical Practice (GCP) became standard practices across the region. At the same time, significant investments were made to upgrade hospitals, establish oncology and genomic centers, and modernize laboratory networks. The period also saw the rise of regional CROs and the entry of global players, reflecting the region’s increasing attractiveness. These foundational investments laid the groundwork for the current rapid growth of clinical research throughout the Middle East.
Current Global Clinical Trial Landscape (2024–2025)
The global clinical trial landscape in 2024–2025 is shaped by three main pressures: slowing recruitment in traditional hubs, rising development costs, and increased regulatory scrutiny. Sponsors are experiencing longer enrollment periods and higher dropout rates, particularly in oncology and metabolic disease trials. Meanwhile, regulatory agencies are tightening evidence requirements for accelerated pathways and real-world evidence submissions. These challenges have driven biopharmaceutical companies, particularly early-stage biotechs, to diversify their trial locations and reduce operational risks. As advanced therapies, precision medicine, and rare disease programs grow, sponsors are increasingly recognizing the importance of regions that provide both patient access and quick execution. The Middle East now plays a central role in this strategic realignment.
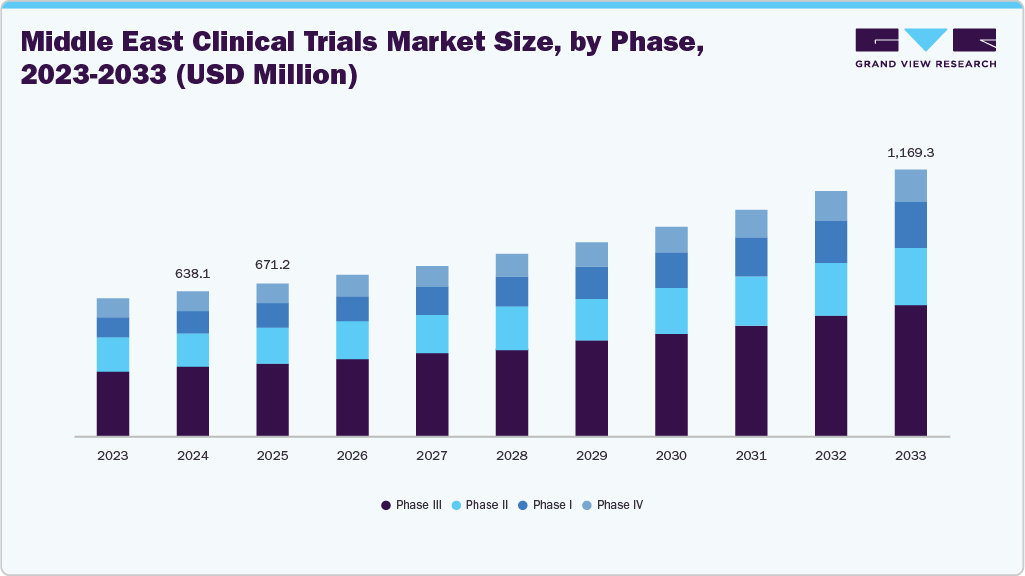
According to Grand View Research's analysis, the Middle East clinical trials market size was estimated at USD 638.11 million in 2024 and is projected to reach USD 1,169.34 million by 2033, growing at a CAGR of 7.18% from 2025 to 2033. The Middle East faces a rising prevalence of chronic diseases such as diabetes, cardiovascular disorders, obesity, and cancer, creating strong demand for new therapies.
Current Stage of Middle East R&D Ecosystem Maturity
The Middle East has evolved from an emerging participant to a more competitive global R&D ecosystem. Saudi Arabia, the UAE, and Qatar now host advanced research hospitals, accredited clinical trial units, and specialized centers for oncology, metabolic diseases, cell therapy, and genomics. Regulatory agencies across the region have digitalized submissions, adopted reliance pathways, and standardized review procedures in alignment with international guidelines. For instance, regulatory harmonization initiatives across the Gulf Cooperation Council (GCC), the Arab Regulators Network, and bilateral reliance frameworks have shortened approval timelines by 30–40% in some markets. Countries such as Saudi Arabia and the UAE now operate digital-first regulatory systems with eCTD submissions, electronic labeling, and AI-enabled review tools. CROs have expanded their capabilities in clinical operations, data management, and biometrics, while governments continue to invest in workforce development and innovation clusters. Collectively, these developments have propelled the region into a phase of rapid maturity, allowing it to support complex Phase I to Phase III studies. Furthermore, increasing collaborations between local and global CROs, academic medical centers, and multinational sponsors have accelerated operational capacity and improved data integrity, enabling the region to compete head-on with established trial hubs.
Clinical Operations Strength and Execution Advantage
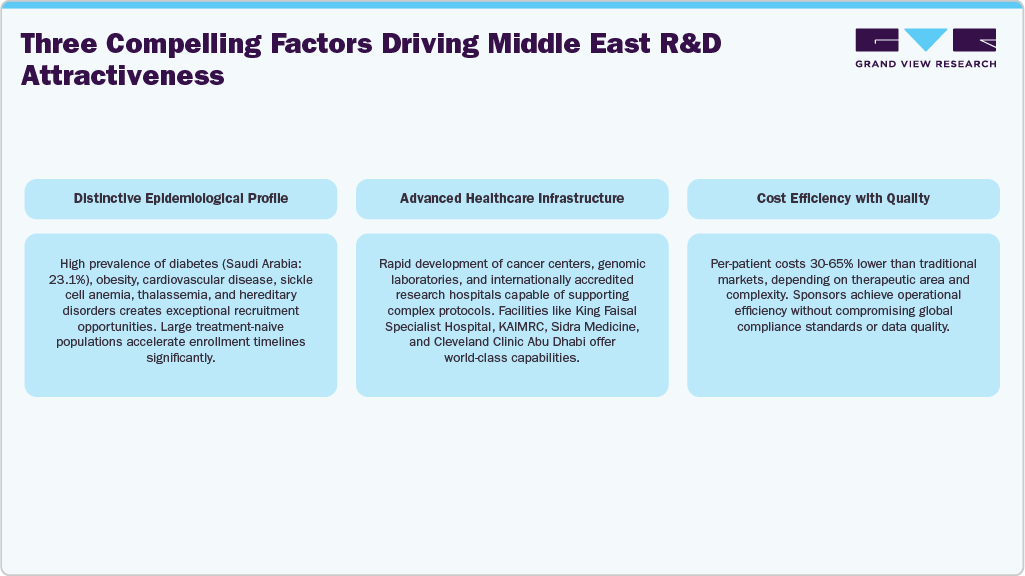
Clinical operational strength has emerged as a key differentiator in the Middle East. The region is dominated by interventional studies, backed by active investigator involvement and large pools of treatment-naive patients. Facilities such as King Faisal Specialist Hospital & Research Centre, KAIMRC, Sidra Medicine, and Cleveland Clinic Abu Dhabi boast top-tier infrastructure and specialized research resources. Site activation times are significantly shorter compared to many Western and Asian markets, aided by digital hospital systems such as electronic health records, AI-powered diagnostics, and patient registries that facilitate targeted recruitment. Both regional and local CROs offer strong execution capabilities, transforming the Middle East into a highly effective area for Phase I, II, and III clinical trials.
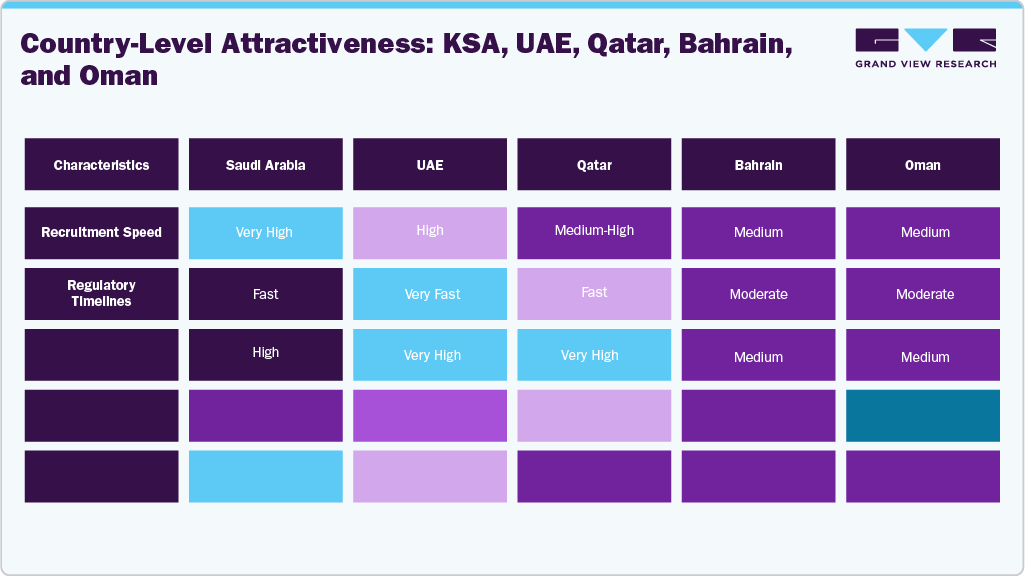
Saudi Arabia serves as the primary market for regional R&D, with the largest population, the strongest government investment, and the most determined clinical research initiatives. The UAE is the fastest-growing ecosystem, supported by world-class private hospitals, strong academic institutions, and innovation-friendly regulatory systems. Qatar has positioned itself as a leader in genomics and precision medicine, offering specialized capabilities for complex therapeutic areas. Bahrain and Oman, though smaller, are modernizing their regulatory systems and expanding research capacity, emerging as efficient secondary markets for multi-country studies. Together, these countries form a diverse yet interconnected clinical research corridor capable of supporting large-scale and high-complexity trials.
Saudi Arabia’s rapid transformation under Vision 2030 has reshaped its role in global clinical research. With over 1,000 interventional trials conducted from 2009 to 2022, the country is now the most active clinical research market in the Middle East. The country’s population is projected to reach 50–60 million by 2030, which offers unparalleled potential for patient recruitment. High disease rates, robust early-phase infrastructure, and internationally aligned regulatory pathways boost its competitiveness. Subsequently, sponsors increasingly view Saudi Arabia as the ideal gateway for trial expansion, Phase III implementation, rare disease initiatives, and the development of precision medicine.
Key Therapeutic Areas of Focus
Therapeutic opportunity clusters in the Middle East reflect regional disease burdens and government priorities. Oncology remains the largest and fastest-growing area, driven by increasing incidence rates and the expansion of cancer center capacity. Cardiometabolic diseases, especially diabetes, present a significant opportunity due to the region’s high prevalence. Rare genetic diseases, influenced by hereditary factors, facilitate rapid recruitment and offer significant scientific value to global sponsors. Immunology, infectious diseases, dermatology, and respiratory conditions comprise the therapeutic portfolio, which is bolstered by robust investigator networks and increasing public awareness of clinical research.
Investment Landscape and Infrastructure Development
Continuous investment in research infrastructure has been key to the region’s progress. Governments have funded new oncology centers, Phase I units, and clinical research clusters, while private healthcare groups contribute advanced facilities and global accreditations. CROs and CDMOs are establishing regional hubs, providing end-to-end support for sponsors and strengthening local capabilities in biometrics, regulatory affairs, data management, and project execution. Investments in genomics, precision medicine, and digital health platforms further improve the depth and scalability of the region’s R&D ecosystem.
Emerging Role of Real-World Evidence and Digital Health
Digital health is redefining the conduct of clinical trials across the Middle East. Electronic health records, national health information exchanges, digital registries, and AI-enabled analytics provide a robust foundation for generating real-world data. Hospitals are increasingly adopting remote monitoring tools, wearable integration, and decentralized trial models to expand patient reach. These capabilities are particularly relevant for oncology, chronic disease, and rare disease studies, where longitudinal datasets and multi-year follow-up are essential. As regulatory agencies begin integrating real-world data into submissions, the Middle East is well-positioned to become a leader in digital-enabled clinical research.
Collaboration Models with Global Pharma and Biotech
Collaboration models in the region now go far beyond traditional CRO engagements. Global pharmaceutical companies are establishing long-term partnerships, research alliances, and functional service provider (FSP) models with regional CROs. Public–private partnerships, especially in Saudi Arabia and the UAE, unite governments, hospitals, and sponsors to boost research capacity. Integrated agreements with technology providers support data management, AI-driven trial optimization, and real-world evidence platforms. These models foster a more resilient, scalable, and sustainable environment for conducting clinical trials.
Risks, Challenges, and Capacity Gaps
Despite rapid growth, challenges persist in areas such as regulatory fragmentation, workforce depth, and readiness for early-phase research. Differences in approval processes, ethics committees, and documentation across Middle Eastern markets can delay multi-country trial activation. Talent shortages in advanced therapeutics and specialized domains require continuous investment in workforce development. Public awareness of clinical trials is inconsistent. Addressing these gaps through targeted investments, workforce development, and regional harmonization will be essential to sustaining the momentum of clinical trial activities.
Forward Outlook: The Middle East on the 2030 Global Clinical R&D Map
By 2030, the Middle East is expected to become a leading contributor to global clinical development. The region is poised to become a primary recruitment hub for oncology, metabolic, and rare disease trials, as well as a preferred destination for Phase I and precision medicine programs. The integration of digital health, reliance-based regulatory frameworks, and expanding research clusters will strengthen its role in supporting complex therapies, including cell and gene treatments. As global sponsors diversify their portfolios to manage geopolitical and operational risks, the Middle East will play a key role in advancing the next generation of medical innovation.