- Home
- »
- Medical Devices
- »
-
Wound Closure Devices Market Size, Industry Report, 2033GVR Report cover
![Wound Closure Devices Market Size, Share & Trends Report]()
Wound Closure Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Sutures, Adhesives, Staples, Strips, Clips, Sealants), By Wound, By End Use, By Distribution Channel, By Mode of Purchase, By Region, And Segment Forecasts
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
- Buy Now
Wound Closure Devices Market Summary
The global wound closure devices market size was estimated at USD 15.39 billion in 2025 and is projected to reach USD 25.19 billion by 2033, growing at a CAGR of 6.31% from 2026 to 2033. The market is primarily driven by the rising number of surgical procedures, increasing incidence of traumatic injuries, and the growing prevalence of chronic conditions such as diabetes.
Key Market Trends & Insights
- North America dominated the wound closure devices industry with the largest revenue share of 39.27% in 2025.
- The U.S. dominated the North America market in 2025, capturing a revenue share of 76.72% of the regional market.
- By product, the suture segment led the market with the largest revenue share in 2025.
- By wound, the acute wounds segment led the market with the largest revenue share in 2025.
- By end use, the acute care hospital segment led the market with the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 15.39 Billion
- 2033 Projected Market Size: USD 25.19 Billion
- CAGR (2026-2033): 6.31%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Advancements in closure technologies that improve healing outcomes and reduce infection risks are further boosting adoption. In addition, expanding healthcare infrastructure and the shift toward minimally invasive procedures continue to support market growth. The growing prevalence of chronic wounds, particularly those associated with diabetes, vascular disorders, and aging populations, is significantly increasing the need for effective wound closure solutions. These wounds require prolonged care and advanced products to support healing and prevent complications.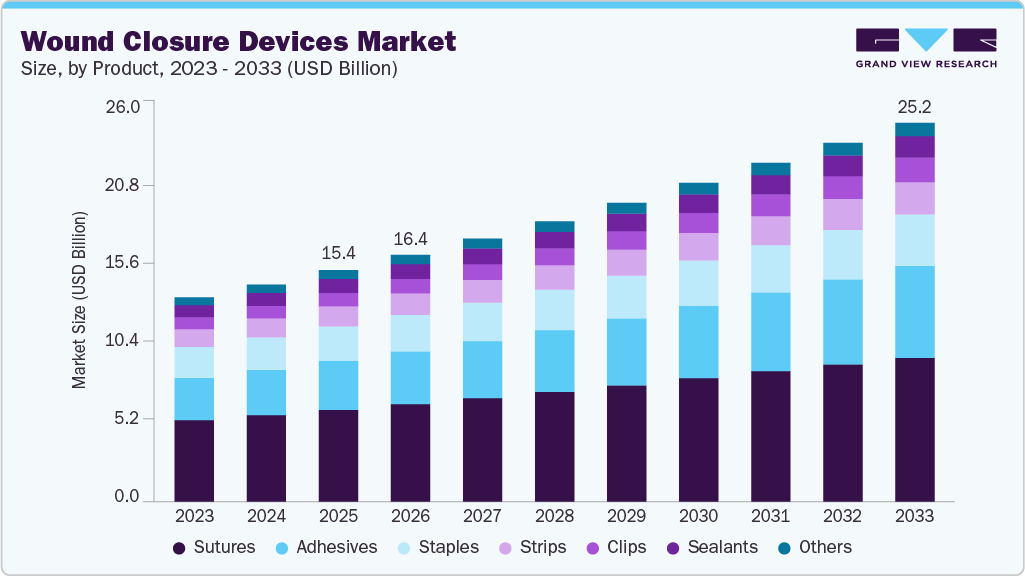
As a result, demand for specialized closure devices and innovative treatment options continues to rise, driving market growth. According to an article published by MDPI in March 2025, diabetic foot remains one of the most severe and complex complications of diabetes, strongly associated with high rates of lower-limb amputation, as well as increased morbidity and mortality. Globally, approximately 18.6 million people are affected by diabetic foot ulcers each year, accounting for nearly 80% of diabetes-related lower limb amputations. It is further estimated that up to 25% of individuals with diabetes may develop diabetic foot, with 19%-34% experiencing foot ulcers during their lifetime.
Key Statistics on Diabetic Foot Ulcers in Saudi Arabia
Parameter
Statistic
Lifetime risk of developing DFU in diabetes patients
19-34%
Mortality within 12 months of DFU
~5%
Mortality within 5 years of DFU
~42%
Amputation risk
Higher in patients with diabetes compared to non-diabetics
Source: NCBI
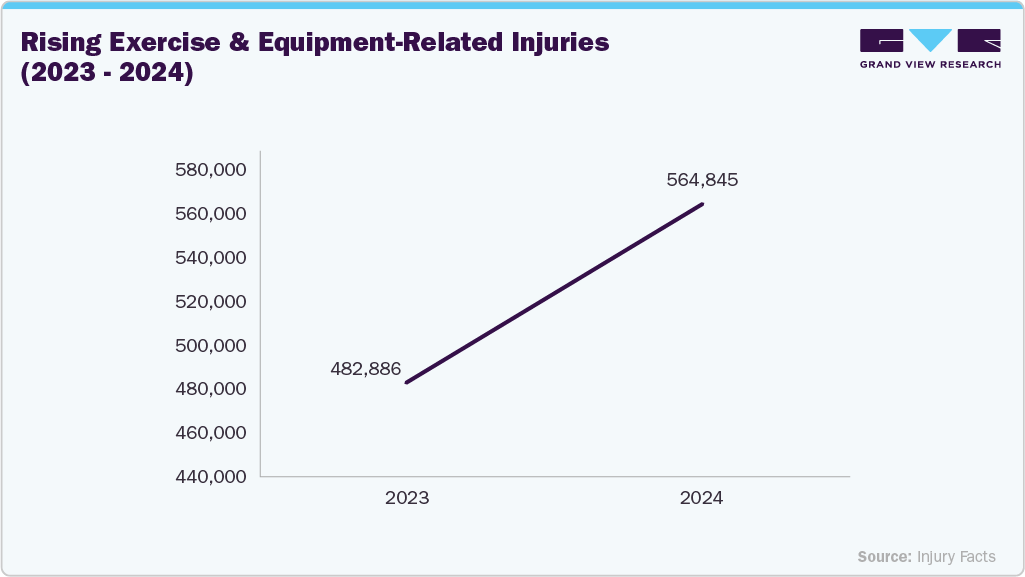
Furthermore, the increasing incidence of sports-related injuries, such as cuts, lacerations, and soft-tissue damage, is significantly contributing to the growth of the market. Rising participation in fitness and recreational activities is driving demand for quick and effective wound management solutions, including sutures, staples, and adhesive closures. This trend further supports market expansion by promoting the adoption of advanced, minimally invasive wound closure technologies. The graph below illustrates the increase in exercise and equipment-related injuries from 2023 to 2024.
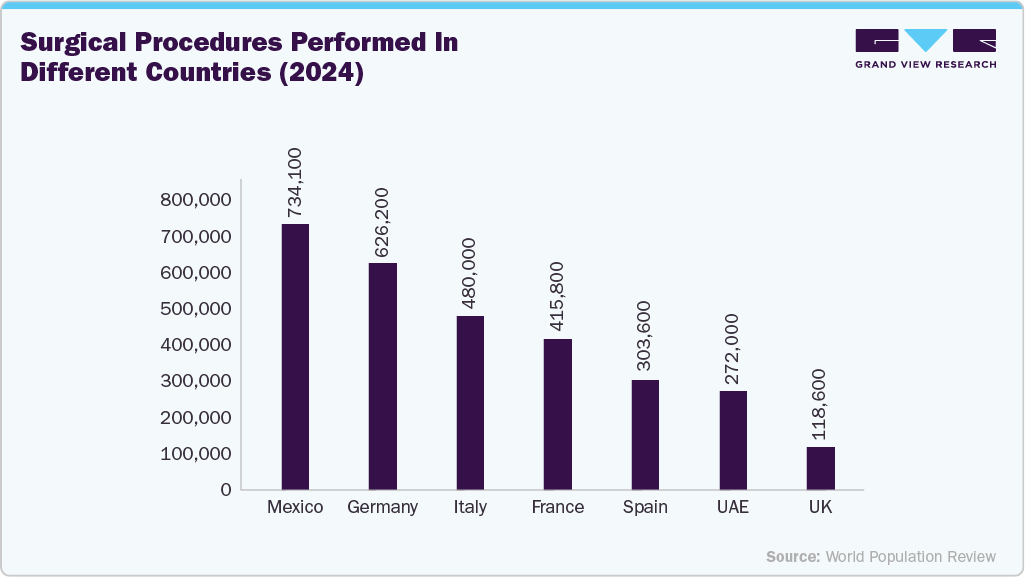
The surgical wound closure devices are the primary growth driver of the overall wound closure devices market, accounting for the largest share of product utilization across hospitals and ambulatory surgical centers. The growing number of surgical procedures is a major driver of demand for wound closure devices (sutures, staplers, and advanced closure technologies), as each operation requires effective, reliable wound management to ensure proper healing. Higher volumes of both elective and emergency surgeries are accelerating the use of sutures, staplers, and adhesives across healthcare settings. This sustained surgical demand continues to boost product utilization and supports consistent market growth.
Furthermore, advancements in wound closure products are significantly driving demand in the market by improving clinical outcomes, reducing infection risks, and enhancing ease of use. Innovations such as antimicrobial sutures, bioactive adhesives, and minimally invasive closure systems enable faster healing and better patient comfort. These improvements are encouraging wider adoption across both surgical and outpatient settings, supporting overall market growth. For instance, Ethicon, part of Johnson & Johnson, has developed advanced wound closure solutions such as DERMABOND PRINEO Skin Closure System and DERMABOND ADVANCED Topical Skin Adhesive, along with Mini Topical Skin Adhesives. These products integrate liquid adhesive with mesh or enhanced formulations to ensure strong wound closure and accurate skin edge approximation.
In May 2024, the RECELL GO System, developed by AVITA Medical, Inc., received U.S. Food and Drug Administration approval. This device utilizes the patient’s own skin cells to support treatment of full-thickness wounds and severe burns, promoting faster healing and reduced discomfort compared to conventional grafting methods. Such regulatory approvals for advanced regenerative technologies are expected to accelerate innovation and drive growth in the market. For instance, the SYLKE Adhesive Wound Closure demonstrates key advancements in wound closure technologies by utilizing 99.9% medical-grade silk fibroin, delivering a biocompatible, breathable, and water-resistant solution. It offers 360° stretchability that closely mimics natural skin movement, ensuring secure adhesion for up to 14 days, even in high-mobility areas such as joints.
As a result, the wound closure devices industry is experiencing steady growth due to the increasing prevalence of chronic diseases, which generate a higher demand for long-term wound management solutions. The rising number of surgical procedures further boosts the need for effective closure products, while continuous technological advancements enhance treatment outcomes and efficiency. Together, these factors are driving sustained expansion of the market.
Market Concentration & Characteristics
The market growth stage is moderate, and the pace of growth is accelerating. The wound closure devices industry is characterized by growth owing to rising cases of surgeries and accidental injuries, along with the growing availability of such devices.
The wound closure devices industry is witnessing moderate-to-high levels of innovation, with advancements focused on improving healing outcomes, reducing infection risk, and enhancing ease of use for clinicians. New developments include bioactive sutures, antimicrobial coatings, and tissue adhesives that promote faster tissue regeneration and minimize scarring. Integration of smart technologies, such as sensor-enabled dressings and closure systems that monitor healing conditions, is also emerging. In addition, innovations are aligned with minimally invasive procedures and outpatient care, emphasizing efficiency, patient comfort, and reduced recovery time. For instance, in June 2025, Ethicon (Johnson & Johnson) introduced the ETHICON 4000 Stapler in the U.S., an advanced device engineered to handle complex tissue and deliver strong staple line integrity, helping minimize the risk of surgical leaks and bleeding across various specialties.
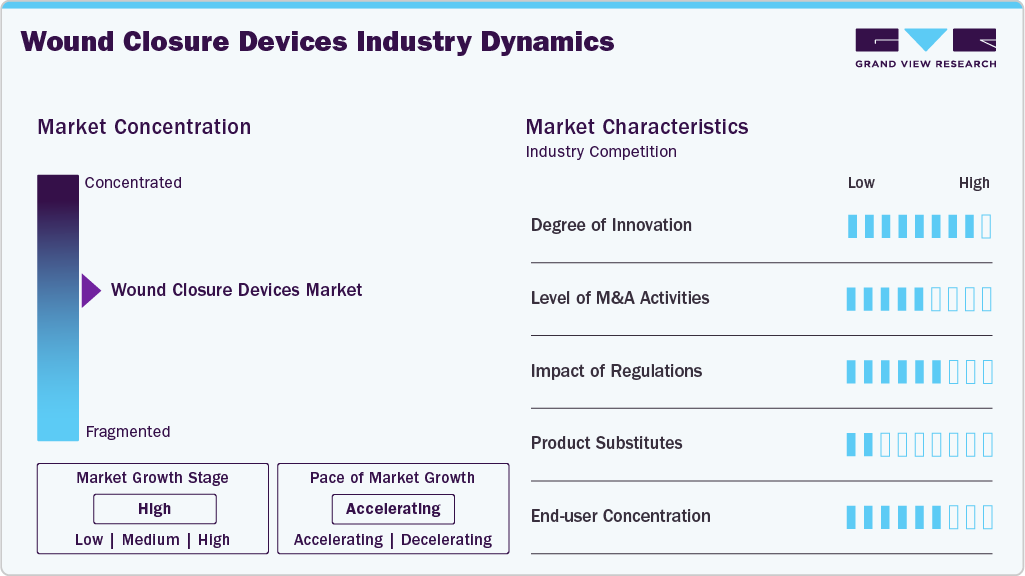
The market for wound closure devices exhibits moderate, strategic-level M&A activity, with companies focusing on acquiring niche technologies and specialized product lines rather than pursuing large-scale consolidation. Deals are typically aimed at strengthening capabilities in advanced closure solutions such as sealants, adhesives, and minimally invasive systems. This selective approach reflects a market driven more by innovation enhancement and portfolio expansion than by aggressive consolidation.
Regulations have a significant impact on the wound closure devices industry, as stringent approval processes and quality standards directly influence product development timelines and market entry. Compliance with safety, sterilization, and biocompatibility requirements drives manufacturers to invest more in R&D and clinical validation. Evolving regulatory frameworks can both encourage innovation in safer products and create barriers to entry for smaller players.
The wound closure devices industry shows a moderate-to-high end-use concentration, with hospitals accounting for the largest share due to high surgical volumes and access to advanced closure technologies. Ambulatory surgical centers and clinics are gradually increasing their contribution, driven by the rise in outpatient procedures and minimally invasive surgeries. However, the market remains relatively concentrated in institutional healthcare settings compared to home care.
Product Insights
Sutures dominated the market in 2025 due to their wide applicability across a broad range of surgical procedures and strong clinician familiarity. Their cost-effectiveness, availability in both absorbable and non-absorbable forms, and reliability in wound management continue to support high adoption. Despite the emergence of advanced alternatives, sutures remain the preferred choice, particularly in high-volume and resource-sensitive healthcare settings.
Adhesives are expected to witness the fastest growth during the forecast period, driven by their ability to enable quicker application, reduced procedure time, and improved patient comfort. Increasing preference for minimally invasive procedures and outpatient care is accelerating their adoption. In addition, advancements in bio-compatible and antimicrobial adhesive formulations are further supporting their rapid market expansion.
Wound Insights
Acute wounds led the market in 2025 and are expected to register the fastest growth during the forecast period, driven by the high incidence of surgical procedures, trauma cases, and emergency treatments. These wounds require immediate, effective closure, driving strong demand for sutures, staplers, and adhesives. Continuous surgical volumes and hospital-based care further reinforce the segment’s leading position.
The chronic wound segment is expected to grow significantly during the forecast period due to the rising prevalence of conditions such as diabetes, obesity, and an aging population. These factors significantly increase the incidence of chronic injuries, such as diabetic ulcers and venous ulcers, needing innovative medical care solutions. In addition, the difficulty of managing such lesions requires innovative products that promote healing and prevent infections. Furthermore, the growing emphasis on improving patient outcomes and reducing healthcare costs drives investments in advanced medical technologies tailored for chronic wound management.
End Use Insights
Acute care hospitals dominated the market in 2025, driven by the high volume of surgical procedures, trauma cases, and emergency treatments performed in these settings. Their access to advanced infrastructure and skilled professionals supports extensive use of sutures, staplers, and adhesives. The continuous influx of patients requiring immediate, complex care sustains their leading market share.
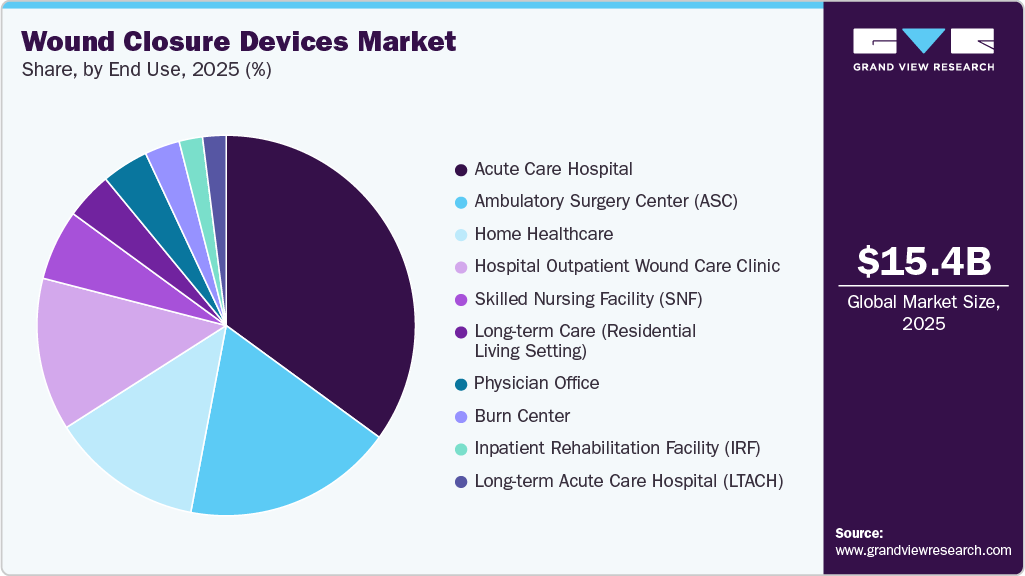
Home healthcare is expected to witness the fastest growth during the forecast period, driven by the rising preference for at-home treatment and post-surgical care. Increasing demand for cost-effective care, shorter hospital stays, and patient convenience is accelerating this shift. In addition, advancements in easy-to-use closure products are supporting greater adoption in home-based settings.
Distribution Channel Insights
Institutional sales dominated the market in 2025, driven by high procurement volumes from hospitals, surgical centers, and other healthcare facilities. These settings require a consistent supply of sutures, staplers, and advanced closure products for a wide range of procedures. Bulk purchasing agreements and established supplier relationships further strengthen institutional sales' strong position in the market.
Retail sales are expected to grow at the fastest CAGR during the forecast period, driven by increasing consumer awareness and demand for over-the-counter wound care products. The expansion of pharmacies, online platforms, and direct-to-consumer channels is improving product accessibility. In addition, the rising trend of self-care and home management of minor wounds is further accelerating growth in this segment.
Mode of Purchase Insights
Prescribed products dominated the market in 2025, as most advanced closure solutions require clinical supervision and are used in surgical or hospital settings. Healthcare professionals play a key role in selecting appropriate closure methods to ensure safety and effectiveness. This strong reliance on physician-directed treatment continues to drive the dominance of the prescribed segment.
Non-prescribed (OTC) wound closure products are expected to grow at the fastest rate during the forecast period, driven by increasing consumer awareness and preference for self-care in minor wound management. Easy availability through pharmacies and online platforms is enhancing accessibility. In addition, the shift toward home-based care and convenience-oriented solutions is accelerating adoption of OTC products.
Regional Insights
North America Wound Closure Devices Market Trends
The wound closure devices industry in North America held the largest share and accounted for 39.27% of global revenue in 2025. This can be attributed due to evolving with a clear shift toward advanced, patient-centric solutions that improve healing outcomes and reduce procedure time. Healthcare providers prefer minimally invasive closure techniques, such as tissue adhesives and absorbable materials, to support faster recovery and lower complication rates. The region is also seeing growing demand driven by the high burden of surgical procedures and chronic wounds, alongside an aging population with complex care needs. In addition, the expansion of ambulatory surgical centers and emphasis on cost efficiency are encouraging the adoption of easy-to-use, time-saving wound closure technologies.
U.S. Wound Closure Devices Market Trends
The U.S. wound closure devices industry is increasingly shifting toward advanced, efficiency-driven solutions, with strong adoption of absorbable sutures, staples, and tissue adhesives that help reduce procedure time and improve healing outcomes. The growing preference for minimally invasive surgeries and outpatient care is further accelerating demand for easy-to-use, fast-acting closure products. In addition, the high incidence of chronic wounds, along with the rising prevalence of chronic diseases such as diabetes and cardiovascular conditions, continues to drive demand while encouraging innovation and cost-effective care delivery across healthcare settings.
As per 2026 Heart Disease and Stroke Statistics Update Fact Sheet-
-
In 2023, CVD caused 915,973deaths in the U.S., more than all forms of cancer and chronic lower respiratory disease combined.
-
In 2023, coronary heart disease (CHD) was the leading cause of deaths (38.2%) attributable to CVD in the U.S., followed by stroke (17.8%), other CVD (17.1%), hypertensive diseases (14.5%), heart failure (HF, 9.8%), and diseases of the arteries (2.7%).
-
In 2023, stroke was the underlying cause of 162,639 deaths in the U.S., with an age-adjusted mortality rate of 39.0 per 100,000 population.
Europe Wound Closure Devices Market Trends
The Europe wound closure devices industry is evolving with a growing focus on high-quality, biocompatible materials and advanced closure techniques that enhance healing and reduce complications. Increasing surgical procedures and an aging population are driving demand for reliable, efficient wound management solutions. In addition, the expansion of outpatient care and the emphasis on reducing hospital stays are encouraging the adoption of faster, minimally invasive wound-closure technologies across the region.
The UK wound closure devices industry is increasingly oriented toward efficient, cost-conscious solutions, with rising adoption of tissue adhesives and advanced suturing materials that support quicker procedures and recovery. The ongoing shift toward outpatient and community-based care is driving demand for easy-to-apply and minimally invasive closure products. In addition, pressures to reduce surgical backlogs and hospital stays are driving the adoption of faster, more reliable wound closure technologies across healthcare settings.
The France wound closure devices industry is gradually shifting toward advanced, patient-friendly solutions, with increasing use of absorbable sutures and tissue adhesives to improve healing outcomes and reduce procedure time. The growing emphasis on outpatient and ambulatory surgical care is driving demand for quick and efficient closure methods. In addition, the rising elderly population and prevalence of chronic wounds are supporting the need for reliable, easy-to-use wound closure technologies across both hospital and home care settings.
Asia Pacific Wound Closure Devices Market Trends
The Asia Pacific wound closure devices industry is witnessing strong growth driven by expanding healthcare infrastructure, rising surgical procedures, and increasing awareness of advanced wound care solutions. There is a growing shift toward cost-effective and easy-to-use products such as absorbable sutures and tissue adhesives, particularly in emerging economies. In addition, the increasing prevalence of chronic wounds, improving access to healthcare, and rising demand for minimally invasive treatments are further shaping market expansion across the region.
China’s wound closure devices industry is evolving with a strong push toward efficiency and cost optimization, as hospitals increasingly prefer faster, easy-to-use solutions that reduce procedure time and hospital stays. Domestic manufacturers are gaining traction by offering competitively priced alternatives, while demand is gradually shifting from traditional sutures to advanced adhesives and staplers. The growing burden of chronic conditions and expanding access to surgical care in lower-tier cities are supporting consistent market expansion.
Latin America Wound Closure Devices Market Trends
The Latin America wound closure devices industry is experiencing significant growth, driven by rising surgical procedures, a growing burden of chronic diseases, and gradual improvements in healthcare access across key countries. There is a noticeable shift from conventional sutures toward advanced solutions such as tissue adhesives and absorbable materials, driven by the need for faster healing and better patient outcomes. In addition, cost-sensitive healthcare systems are encouraging the adoption of efficient, value-based products, shaping a balanced but innovative-driven market expansion.
MEA Wound Closure Devices Market Trends
The MEA wound closure devices industry is advancing rapidly, driven by improving healthcare infrastructure and a gradual increase in surgical procedures across urban centers. Demand is shifting toward more efficient, infection-reducing solutions, such as advanced sutures and adhesives, particularly in private healthcare settings. However, growth remains uneven due to cost sensitivity and limited access in rural areas, creating a mixed adoption landscape across the region.
Key Wound Closure Devices Company Insights
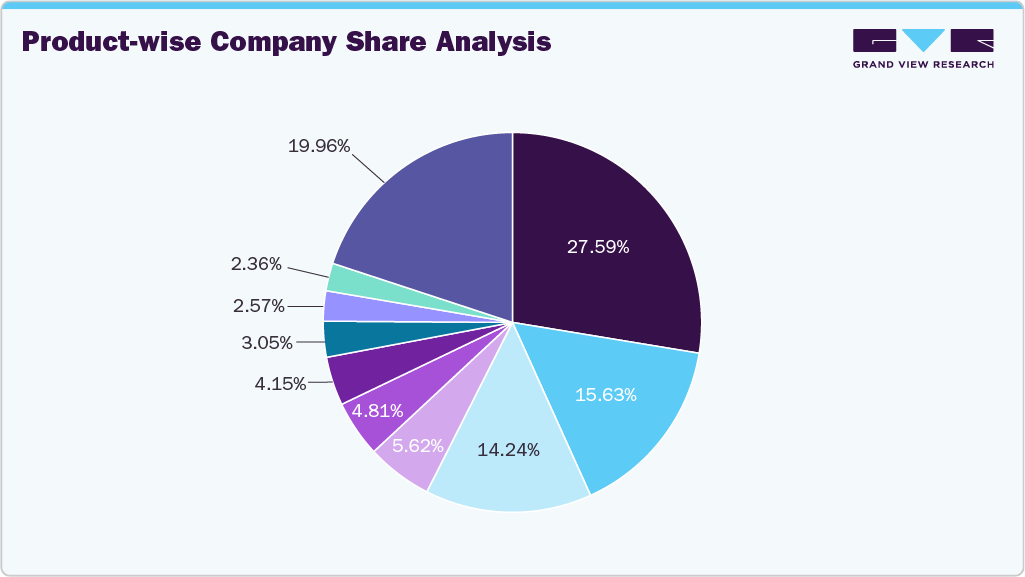
3M, Ethicon (Johnson & Johnson Services, Inc.), Medtronic, Baxter, Smith+Nephew, Stryker, Advanced Medical Solutions Group plc, among others are some of the major players in the wound closure devices industry. Companies are launching products to gain a competitive advantage in the coming years.
Key Wound Closure Devices Companies:
The following key companies have been profiled for this study on the wound closure devices market.
- Solventum
- 3M
- DeRoyal Industries, Inc.
- DermaRite Industries, LLC.
- B. Braun SE
- Cardinal Health
- Baxter
- Stryker
- Smith+Nephew
- Medtronic
- Ethicon (Johnson & Johnson)
- Riverpoint Medical
- DermaClip
- Medline Industries, LP
- Gentell
- Dynarex Corporation
- Corza Medical
- Dukal, LLC
- Aspen Surgical Products, Inc.
- Essity Health & Medical (Essity)
- Aero Healthcare
- Healthium Medtech Ltd.
- KitoTech Medical, Inc.
- Trusetal Verbandstoffwerk GmbH
- Advanced Medical Solutions Group plc
- H.B. Fuller Medical Adhesive Technologies, LLC
- AVITA Medical, Inc.
- Meril
- Terumo Corporation
- CliniSut
- Synthecon
- Dolphin Sutures (Futura Surgicare Pvt Ltd)
- Unisur Lifecare
- AdvaCare Pharma
- Suzhou Sunmed Co., Ltd.
- Samyang Biopharm Corporation
- Baihe Medical
- MUB Medical.
- BRIJ Medical
- Southmedic Inc.
- Sylke, Inc.
- Clozex Medical, Inc.
- DemeTECH
- Teleflex (incl. Weck)
- Lotus Surgicals
- Internacional Farmaceutica
- Serag-Wiessner
- Lux Sutures
- Chemence Medical
- Intuitive Surgical
- Frankenman
- Reach Surgical (Genesis MedTech)
- Purple Surgical
- Grena Ltd
- Ackermann Surgical
- CSL Behring
- Integra LifeSciences
- Artivion (formerly CryoLife)
- Takeda
- McKesson Medical-Surgical
- Mölnlycke Health Care
- Paul Hartmann AG
- Lohmann & Rauscher
Recent Developments
-
In June 2025, Ethicon, part of Johnson & Johnson, announced the U.S. launch of the ETHICON 4000 Stapler, an advanced surgical stapler designed to manage complex tissue and ensure strong staple line integrity, thereby reducing the risk of surgical leaks and bleeding complications across multiple specialties.
-
In April 2025, Henry Schein, Inc. entered into a distribution agreement with Health Future, enabling access to its Disposable Skin Stapler and Skin Staple Remover across the consortium’s network. The collaboration supports the integration of these wound closure products across 22 hospitals, clinics, and care centers, aiming to improve patient outcomes, streamline procurement, and enhance operational efficiency.
-
In April 2025, AVITA Medical, Inc. announced the presentation of 16 abstracts at the American Burn Association 2025 Annual Meeting, highlighting real-world and clinical data from its wound care portfolio, including RECELL, Cohealyx, and PermeaDerm. The research presentations emphasize the company’s expanding clinical evidence base and its commitment to improving outcomes in burn and wound care.
-
In October 2024, Mölnlycke Health Care collaborated with Transdiagen for advanced research in wound care. This partnership will give rise to innovative wound closure devices using expertise from both companies.
-
In July 2024, AVITA Medical, Inc. collaborated with Regenity Biosciences to strengthen the company's wound care portfolio with Regenity’s bioresorbable technology and expand its wound care product distribution.
-
In July 2024, Vernacare Ltd (H.I.G. Capital, LLC), a manufacturer of wound closure strips, transferred USD 4.54 million in surgical tool operations from China to the UK. This plan would allow the company to extend its manufacturing capacity at Worksop, Newtown, Wales, and England establishments and save about USD 324.60 million.
Wound Closure Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 16.41 billion
Revenue forecast in 2033
USD 25.19 billion
Growth Rate
CAGR of 6.31% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends, and volume analysis
Segments covered
Product, wound, end use, distribution channel, mode of purchase, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country Scope
U.S.; Canada; Mexico; UK; Germany; Italy; France; Spain; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Solventum; 3M; DeRoyal Industries, Inc.; DermaRite Industries, LLC.; B. Braun SE; Cardinal Health; Baxter; Stryker; Smith+Nephew; Medtronic; Ethicon (Johnson & Johnson); Riverpoint Medical; DermaClip; Medline Industries, LP; Gentell; Dynarex Corporation; Corza Medical; Dukal, LLC; Aspen Surgical Products, Inc.; Essity Health & Medical (Essity); Aero Healthcare; Healthium Medtech Ltd.; KitoTech Medical, Inc.; Trusetal Verbandstoffwerk GmbH; Advanced Medical Solutions Group plc; H.B. Fuller Medical Adhesive Technologies, LLC; AVITA Medical, Inc.; Meril; Terumo Corporation; CliniSut; Synthecon; Dolphin Sutures (Futura Surgicare Pvt Ltd); Unisur Lifecare; AdvaCare Pharma; Suzhou Sunmed Co., Ltd.; Samyang Biopharm Corporation; Baihe Medical; MUB Medical; BRIJ Medical; Southmedic Inc.; Sylke, Inc.; Clozex Medical, Inc.; DemeTECH; Teleflex (incl. Weck); Lotus Surgicals; Internacional Farmaceutica; Serag-Wiessner; Lux Sutures; Chemence Medical; Intuitive Surgical; Frankenman; Reach Surgical (Genesis MedTech); Purple Surgical; Grena Ltd.; Ackermann Surgical; CSL Behring; Integra LifeSciences; Artivion (formerly CryoLife); Takeda; McKesson Medical-Surgical; Mölnlycke Health Care; Paul Hartmann AG; Lohmann & Rauscher
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Wound Closure Devices Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global wound closure devices market report based on product, wound, end use, distribution channel, mode of purchase, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Sutures
-
Surgical Suture
-
Absorbable
-
Non-Absorbable
-
-
Barbed Suture (only Absorbable)
-
Uni-directional barbed suture
-
Bi-directional barbed suture
-
-
Gut Suture
-
Microsutures
-
-
Adhesives
-
Staples
-
Powered
-
Manual
-
-
Strips
-
By Sterility
-
Sterile
-
Non-Sterile
-
-
By Product
-
Reinforced Strips
-
Flexible Strips
-
-
-
Clips
-
Sealants
-
Non-Synthetic
-
Collagen Based
-
Synthetic
-
Topical Cyanoacrylates
-
Others
-
-
-
Others
-
-
Wound Outlook (Revenue, USD Million, 2021 - 2033)
-
Acute Wounds
-
Surgical/ Procedure
-
Obstetrics / Gynecology
-
Colorectal
-
Cardiovascular
-
Bariatric and Upper GI
-
Plastic and Reconstructive Surgery
-
Urology
-
Hepato-pancreato-biliary (HPB)
-
Dermatology
-
Others
-
-
Lacerations & Minor Cuts
-
Burns
-
-
Chronic Wounds
-
Diabetic Foot Ulcers
-
Pressure Ulcers
-
Venous Leg Ulcers
-
-
Other Chronic Wounds
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Acute Care Hospital
-
Ambulatory Surgery Center (ASC)
-
Hospital Outpatient Wound Care Clinic
-
Home Healthcare
-
Skilled Nursing Facility (SNF)
-
Physician Office
-
Burn Center
-
Inpatient Rehabilitation Facility (IRF)
-
Long-term Acute Care Hospital (LTACH)
-
Long-term Care (residential living setting)
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Institutional Sales
-
Retail Sales
-
-
Mode of Purchase Outlook (Revenue, USD Million, 2021 - 2033)
-
Prescribed
-
Non-prescribed (OTC)
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
Italy
-
France
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
MEA
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
Key factors driving the market growth include the prevalence of chronic wounds, increasing cases of accidents, and the rising number of surgeries.
The global wound closure devices market size was estimated at USD 15.39 billion in 2025 and is expected to reach USD 16.41 billion in 2025.
The global wound closure devices market is expected to grow at a compound annual growth rate of 6.31% from 2026 to 2033 to reach USD 25.19 billion by 2033.
The sutures segment held the dominant market share in 2025 owing to the rising number of surgeries and burn cases globally.
Some key players operating in the wound closure devices market include Solventum; 3M; DeRoyal Industries, Inc.; DermaRite Industries, LLC.; B. Braun SE; Cardinal Health; Baxter; Stryker; Smith+Nephew; Medtronic; Ethicon (Johnson & Johnson); Riverpoint Medical; DermaClip; Medline Industries, LP; Gentell; Dynarex Corporation; Corza Medical; Dukal, LLC; Aspen Surgical Products, Inc.; Essity Health & Medical (Essity); Aero Healthcare; Healthium Medtech Ltd.; KitoTech Medical, Inc.; Trusetal Verbandstoffwerk GmbH; Advanced Medical Solutions Group plc; H.B. Fuller Medical Adhesive Technologies, LLC; AVITA Medical, Inc.; Meril; Terumo Corporation; CliniSut; Synthecon; Dolphin Sutures (Futura Surgicare Pvt Ltd); Unisur Lifecare; AdvaCare Pharma; Suzhou Sunmed Co., Ltd.; Samyang Biopharm Corporation; Baihe Medical; MUB Medical; BRIJ Medical; Southmedic Inc.; Sylke, Inc.; Clozex Medical, Inc.; DemeTECH; Teleflex (incl. Weck); Lotus Surgicals; Internacional Farmaceutica; Serag-Wiessner; Lux Sutures; Chemence Medical; Intuitive Surgical; Frankenman; Reach Surgical (Genesis MedTech); Purple Surgical; Grena Ltd.; Ackermann Surgical; CSL Behring; Integra LifeSciences; Artivion (formerly CryoLife); Takeda; McKesson Medical-Surgical; Mölnlycke Health Care; Paul Hartmann AG; Lohmann & Rauscher
About the Author(s)
Medical Devices Research Team
Healthcare · Medical DevicesThis report was authored by the medical devices research team at Grand View Research - comprising two research analysts, one senior research analyst, and one industry expert - with specialized expertise in the medical devices segment of the healthcare industry. All findings are based on proprietary healthcare databases, executive interviews, and regulatory analysis, subject to internal peer review prior to publication.
Last Updated:
Speak to Analyst
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.









