- Home
- »
- Market Trend Reports
- »
-
Continuous Glucose Monitoring Ecosystem: Brands, Patients & Adoption
Definition And Scope of Continuous Glucose Monitoring
Continuous glucose monitoring refers to medical technology used for the continuous measurement of glucose levels in interstitial fluid throughout the day and night. Continuous glucose monitoring (CGM) systems typically consist of a small wearable sensor inserted under the skin, a transmitter that sends glucose data, and a receiver or smartphone application that displays real-time readings, trends, and alerts. Unlike traditional self-monitoring of blood glucose (SMBG) methods that rely on intermittent fingerstick testing, CGM provides dynamic glucose data at frequent intervals, enabling patients and healthcare providers to monitor fluctuations, detect hyperglycemia or hypoglycemia, and make timely treatment adjustments. The scope of CGM includes real-time CGM (rtCGM), intermittently scanned CGM (isCGM), and implantable CGM systems, which are widely used in the management of type 1 and type 2 diabetes and are increasingly integrated with digital health platforms and automated insulin delivery systems.
Top Companies Developing Implantable or Long-Wear Glucose Sensors and Their Key Products
Segment
Key Brand
Company
Implantable Glucose Sensors
Eversense/Eversense E3
Senseonics Holdings, Inc.
Eversense XL
Senseonics Holdings, Inc.
Long-Wear CGM Platforms Supporting Implantable Ecosystems
DexCom G7
DexCom, Inc.
Guardian Sensor 4/Synergy Platform
Medtronic plc
FreeStyle Libre Portfolio
Abbott Diabetes Care
Emerging Implantable Sensor Technologies
GlucoSense Optical Implantable Sensor
GluSense Ltd.
Optical Fiber-Based Implantable Sensor Platform
GlucoVista/Academic-Industry Collaborations
Enzyme-free Fluorescent Polymer Implantable Sensor
Several start-ups (Stealth Biotech Firms)
Sources: Company Websites, Investor Presentations, R&D Disclosures
The development of implantable and long-wear glucose sensing technologies represents an important advancement in the continuous glucose monitoring (CGM) landscape, as companies seek to reduce the frequency of sensor replacements while improving long-term glucose monitoring accuracy and patient convenience. Implantable CGM systems, such as the Eversense platform developed by Senseonics, are designed to be placed under the skin for extended periods and can provide continuous glucose readings for several months before replacement is required. In addition to fully implantable sensors, major CGM manufacturers such as Dexcom, Medtronic, and Abbott are advancing long-wear transcutaneous CGM platforms with improved sensor longevity, accuracy, and integration with digital diabetes management ecosystems. At the same time, several emerging technology developers and research collaborations are exploring next-generation implantable sensor platforms, including optical sensing technologies, fiber-based monitoring systems, and enzyme-free fluorescent polymer sensors. These innovations highlight the growing industry focus on extending sensor lifespan, minimizing patient burden, and enabling more seamless long-term glucose monitoring solutions.
Evolution from Self-Monitoring of Blood Glucose (SMBG) to Continuous Monitoring
Glucose monitoring has evolved significantly from traditional self-monitoring of blood glucose (SMBG) methods to advanced continuous glucose monitoring (CGM) technologies. SMBG systems, which rely on intermittent fingerstick blood samples and test strips, have long been the standard approach for diabetes management, allowing patients to measure glucose levels at specific times during the day. However, SMBG provides only snapshot readings and may miss rapid glucose fluctuations between tests. The introduction of CGM systems has transformed glucose monitoring by providing continuous, real-time measurements throughout the day and night, along with trend data and alerts for high or low glucose levels. This shift toward continuous monitoring enables more comprehensive glycemic management, supports proactive clinical decision-making, and improves patient awareness of glucose patterns, particularly in individuals requiring intensive insulin therapy.
- North America
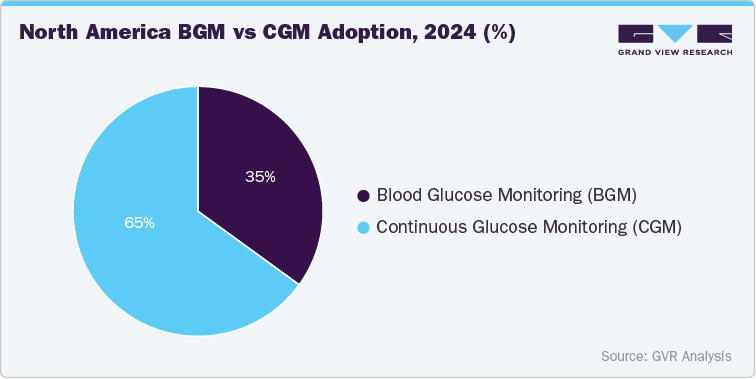
In North America, CGM has become the dominant technology among monitored patients, in market value, and being the most technologically advanced region, supported by strong reimbursement and rapid uptake of wearable CGMs. CGM is increasingly considered near “standard of care” for many people with type 1 diabetes in high-income countries, and rapidly expanding to insulin-treated type 2 diabetes. BGM remains relevant, especially for individuals with mild diabetes not using insulin, those paying out-of-pocket, or as a backup measurement method. Overall, the region is considered one of the most mature CGM markets globally, where CGM has shifted from a niche clinical tool to a mainstream diabetes management approach.
Key Market Drivers and Trends
Rising Prevalence of Diabetes and Prediabetes
Traditional blood glucose testing is often invasive, intermittent, and inconvenient, prompting both patients and providers to adopt CGM systems for better glycemic control and improved quality of life. According to the International Diabetes Federation (IDF), as of 2024, there are approximately 589 million adults living with diabetes worldwide.
Global Diabetes Prevalence, 2024, Age [20-79]
Country
2024 (In Mn)
North America and the Caribbean
56m
South and Central America
35m
Europe
66m
Southeast Asia
107m
Western Pacific
215m
Middle East & North Africa
85m
Africa
25m
Total
589m
Source: International Agency for Research on Cancer (IARC), GVR Analysis
The market is also witnessing a shift toward the increasing demand for personalized, real-time diabetes care. Unlike traditional fingerstick glucose monitoring, CGM devices offer continuous insights into glucose trends, helping patients and healthcare providers make proactive decisions. For instance, in August 2024, Abbott and Medtronic announced a global partnership to integrate Abbott’s FreeStyle Libre CGM technology with Medtronic’s automated insulin delivery systems. This was a critical step towards simplifying diabetes management and reducing the burden of constant self-monitoring. According to a key personnel at Abbott Laboratories’ Diabetes Care Division,
“Libre technology has set the standard for accurate, accessible, easy-to-use, and reliable continuous glucose monitoring. Connecting this CGM built for Medtronic's insulin delivery systems and algorithms makes it easier for people to spend less time thinking about their diabetes and more time living.”
Patients with diabetes are increasingly seeking tools that provide real-time data to prevent both hyperglycemic and hypoglycemic events. CGMs provide this data by measuring interstitial glucose levels daily and at night, offering alerts and predictive analysis that allow for timely interventions. This is especially important in insulin-dependent populations such as those with Type 1 diabetes or advanced Type 2 diabetes.
The global rise in diabetes is one of the strongest demand amplifiers for implantable glucose sensors. With over 530 million individuals currently living with diabetes and another 400+ million in the prediabetes spectrum, there is an increasing need for continuous, accurate metabolic monitoring. Conventional BGM does not provide the granularity or frequency of data required to manage modern diabetes care, while short-wear CGMs, although advancing rapidly face limitations in skin tolerance, wear time, and dropout rates. Implantable sensors bridge the gap by delivering long-term, maintenance-light monitoring suited for growing populations of Type 1 and increasingly Type 2 diabetes patients. As earlier diagnosis and preventive intervention become priorities for health systems globally, implantable platforms are gaining strategic relevance as tools that can detect glycemic variability patterns long before diabetes complications emerge.
CGM Technology Landscape
Real-Time CGM (rtCGM) Systems
Real-time continuous glucose monitoring (rtCGM) systems are advanced glucose monitoring technologies that continuously measure glucose levels in interstitial fluid and automatically transmit readings to a receiver, smartphone, or connected medical device at frequent intervals, typically every 1–5 minutes. These systems utilize a small wearable sensor and transmitter that provide real-time glucose values, trend graphs, and predictive alerts for hypo- and hyperglycemia. Unlike intermittently scanned CGM systems, rtCGM devices automatically stream glucose data without requiring manual scanning, enabling patients and clinicians to monitor glucose fluctuations continuously and make timely treatment decisions. As a result, rtCGM systems are widely used among individuals with intensive insulin therapy and are increasingly integrated with automated insulin delivery (AID) systems and digital diabetes management platforms.
Recent product developments and industry collaborations between 2024 and 2026 have further strengthened the adoption of rtCGM systems. For example, in 2025, Dexcom received regulatory clearance for the Dexcom G7 15-Day CGM, an upgraded version of its real-time CGM platform offering an extended sensor wear duration of up to 15.5 days, improving convenience and reducing the number of monthly sensor replacements for users. Additionally, Abbott announced a global partnership with Medtronic in 2024 to integrate its FreeStyle Libre CGM technology with Medtronic’s automated insulin delivery systems and smart insulin pens, highlighting the growing trend toward integrated diabetes management ecosystems. In another development, collaborations between CGM companies and digital health platforms are expanding the use of CGM beyond traditional diabetes care, such as the 2026 partnership between Abbott and Withings, which enables integration of Abbott’s over-the-counter CGM devices with the Withings health ecosystem for broader metabolic health monitoring.
Sensor Accuracy Metrics (MARD, Calibration Requirements)
Sensor accuracy is a critical performance parameter for continuous glucose monitoring (CGM) systems, as it determines how closely the sensor readings align with reference blood glucose measurements. One of the most widely used metrics to evaluate CGM accuracy is the Mean Absolute Relative Difference (MARD), which represents the average percentage difference between CGM sensor readings and laboratory or capillary blood glucose values. A lower MARD value generally indicates higher accuracy and reliability of the CGM system. Over the past decade, advancements in sensor chemistry, signal processing algorithms, and data filtering techniques have significantly improved CGM accuracy, with many modern devices achieving MARD values typically in the range of approximately 8–10%. Improved accuracy enables better clinical decision-making, reduces the need for confirmatory fingerstick testing, and supports integration with automated insulin delivery systems.
Another important aspect of CGM performance is calibration requirements, which refer to whether a device requires periodic fingerstick blood glucose measurements to maintain accuracy. Earlier CGM systems often required users to perform one or more daily calibrations using traditional blood glucose meters. However, many newer CGM platforms are designed to be factory-calibrated, eliminating the need for routine fingerstick calibration and improving user convenience. This shift toward calibration-free systems has been enabled by improvements in sensor manufacturing, enzyme stability, and algorithm-based error correction. Reduced calibration requirements enhance patient adherence and usability, making CGM systems more suitable for continuous daily wear and broader adoption among individuals with both type 1 and type 2 diabetes.
Global Patient Base of CGM Users
CGM Companies
Active User Base
Total User Base
Abbott Laboratories
460,000-470,000
2,800,000-2,900,000
DexCom, Inc.
This data will be provided in the final deliverable.
Medtronic plc
Others
GVR Analysis
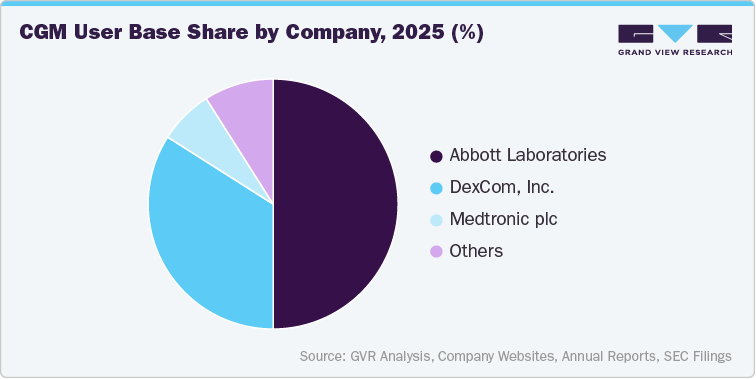
FreeStyle Libre (Abbott Laboratories)
The FreeStyle Libre continuous glucose monitoring (CGM) system, developed by Abbott Diabetes Care, is one of the most widely adopted CGM platforms globally and is designed to provide convenient, continuous glucose monitoring for individuals with diabetes. The system consists of a small wearable sensor applied to the back of the upper arm that measures glucose levels in interstitial fluid and transmits data to a compatible reader or smartphone application.
Earlier versions operated as an intermittently scanned CGM (isCGM), requiring users to scan the sensor to view glucose readings, while newer generations such as FreeStyle Libre 3 offer real-time glucose monitoring with automatic data transmission and optional alerts. The platform is known for its relatively longer sensor wear duration, factory calibration, and user-friendly design, which have contributed to its widespread adoption across both type 1 and type 2 diabetes populations. Continuous product improvements and expanding regulatory approvals have strengthened Abbott’s position in the CGM market, making the FreeStyle Libre system a key driver of global CGM adoption.
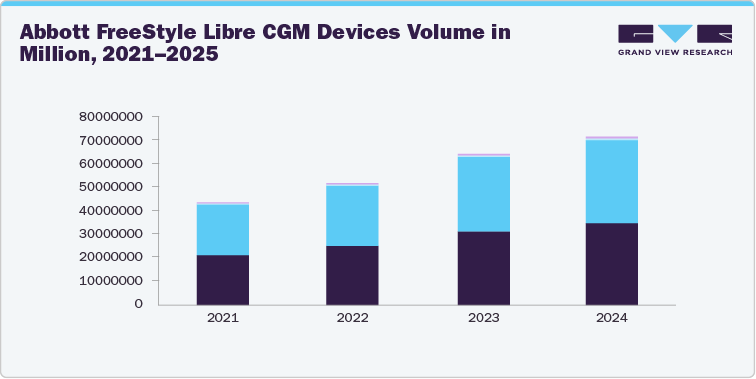
- Global CGM User Base, 2021-2025
Year
Global CGM Users
Major Products
2021
Total CGM User Base of the Company
FreeStyle Libre 2/3
2022
2023
2024
2025
Company Websites, Annual Reports, SEC Filings
Product Lifecycle and Upgrade Trends
Transition from DexCom G6 to G7
The transition from Dexcom G6 to Dexcom G7 represents a significant product upgrade within Dexcom’s continuous glucose monitoring (CGM) portfolio, reflecting advancements in sensor design, usability, and data connectivity. The Dexcom G7 system integrates the sensor and transmitter into a single, smaller wearable device, reducing overall size and simplifying the application process for users. In addition, the G7 offers a shorter warm-up period, improved accuracy, and enhanced mobile connectivity features compared to the G6 platform. Dexcom has been actively transitioning its installed user base from the G6 to the G7 system through expanded regulatory approvals, broader geographic launches, and increased integration with digital diabetes management platforms and automated insulin delivery systems. This upgrade cycle is expected to gradually shift the majority of Dexcom users toward the newer G7 platform as the company continues to expand its product ecosystem and improve patient convenience.
Product Market Share Analysis (%), 2024
The CGM devices market is characterized by a mix of well-established multinational players and emerging specialty brands, each occupying distinct positions based on product portfolio, regional reach, and innovation capabilities. Companies like Abbott Laboratories, DexCom, Inc. and Medtronic plc dominate the premium segment, leveraging strong brand recognition, extensive clinical evidence, and global distribution networks to secure leading market shares.
Share this report with your colleague or friend.
Pricing & Purchase Options
Service Guarantee
-
Insured Buying
This report has a service guarantee. We stand by our report quality.
-
Confidentiality
We are in compliance with GDPR & CCPA norms. All interactions are confidential.
-
Custom research service
Design an exclusive study to serve your research needs.
-
24/5 Research support
Get your queries resolved from an industry expert.
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


