- Home
- »
- Market Trend Reports
- »
-
Next-Generation Wound Dressings Trends, Technologies, And Insights
Executive Summary
The next generation wound dressings are evolving with the increasing prevalence of chronic wounds, and rising demand for effective home-based care. Emerging technologies such as hydrocolloid, foam, hydrogel, alginate, antimicrobial, sensor-integrated dressing, AI, and cloud-based platforms are transforming wound management by enabling real-time monitoring, personalized treatment, and more efficient care delivery.
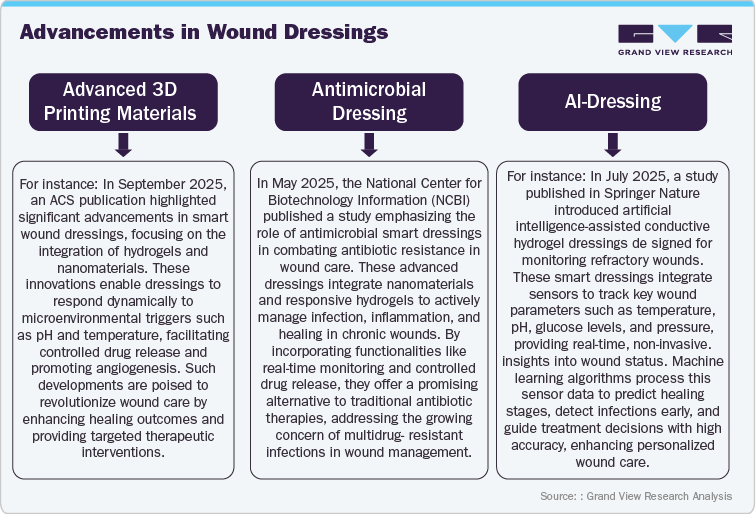
Market Drivers
- Rising Prevalence of Chronic Wounds
The rising prevalence of chronic wounds, including diabetic foot ulcers, pressure ulcers, venous leg ulcers, and arterial ulcers, is driving significant growth in the wound dressing market. In October 2023, the Advances in Wound Care publication reported that chronic wounds affect 10.5 million U.S. Medicare beneficiaries, an increase of 2.3 million since 2014. These wounds impact the quality of life of nearly 2.5% of the total U.S. population, highlighting a significant public health concern. According to the American Diabetes Association, an estimated 537 million people worldwide have diabetes, and 19% to 34% of these individuals are expected to develop a diabetic foot ulcer in their lifetime.
This growing demand encourages innovation in developing innovative, antimicrobial, bioactive dressings, and sensor-integrated and AI-enabled technologies. As a result, companies are investing in research and clinical trials to enhance healing outcomes, reduce infection risks, and improve the quality of life for patients with chronic wounds.
- Prevalence and Prevention of Hospital-Acquired Pressure Injuries in Omani Critical Care Units (2024)
Variable
Prevalence
Overall HAPI (including Stage I)
21.8%
HAPI (excluding Stage I)
19.2%
Most affected site
Sacrum
Predominant stage
Stage II
At-risk patients receiving adequate prevention
73.3%
- Increasing Focus on Clinical Trials
Increasing focus on clinical trials is a key driver of advancements in wound dressings, as it provides robust evidence of safety and efficacy, supports regulatory approvals, and accelerates the adoption of innovative materials and smart technologies in clinical practice. Ongoing trials enable the optimization of dressings formulations and personalized therapies, ensuring better healing outcomes for diverse patient populations.
Below is a table summarizing a few of the recently completed or ongoing studies in this field.
Study Title
Conditions
Interventions
Sponsor
Enrollment
Completion Date
Exciflex for Chronic Wound Therapy
Chronic Wound|Diabetes|Spinal Cord Injury|Ischemic Wound
DEVICE: exciflex
VA Office of Research and Development
16
9/30/2029
Smart Bandage With App-Based Telemonitoring Versus Standard Wound Care for Venous Leg Ulcer: A Randomized Controlled Trial
Venous Leg Ulcer (VLU)|Chronic Wound
DEVICE: Smart Bandage with Telemonitoring|DEVICE: Conventional Bandage
Asmat Burhan
110
11/30/2025
Bioactive Smart Dressings for Diabetic Foot Ulcers: Randomized Controlled Trial
Diabetic Foot Ulcer
COMBINATION_PRODUCT: PRP gel and SOC-treatment|COMBINATION_PRODUCT: EPO/ISDN/UFH cryogel dressing|PROCEDURE: Standard of Care
Mansoura University
30
1/1/2023
Source: ClinicalTrials.gov
Competitive Scenario
Some of the key players operating in the next generation wound dressings include:
-
Scapa
-
Axio Biosolutions Pvt Ltd
-
Smith+Nephew
-
AVERY DENNISON CORPORATION
-
MEDCu
-
Mölnlycke AB
-
Convatec Inc.
-
3M
-
Triage Meditech Pvt. Ltd
-
Advamedica Inc
Recent Developments
Some of the strategies undertaken by key industry players operating in the advancements in wound dressings market are mentioned below:
-
In March 2025, Mölnlycke Health Care launched Mepilex Border Flex, a multi-layered silicone-coated foam dressing designed to improve chronic wound care. Dressing enhances conformability and exudate management, reduces dressing-related complications, and promotes faster healing and patient comfort.
-
In September 2025, Advamedica Inc. received FDA 510(k) clearance and HCPCS code approval for MaxioCel, its advanced wound care dressing made from 100% chitosan. The approval expands its availability in the U.S., complementing over five years of use in Europe and other countries. MaxioCel is designed to manage moderate to heavily exuding wounds, maintaining a moist healing environment and supporting easy removal.
In June 2023, JeNaCell, an Evonik company, launched the epicite balance wound dressing in Germany. Made from biosynthetic cellulose, it supports healing of chronic wounds with low to medium exudate by maintaining a moist environment, and is suitable for conditions like venous leg ulcers, diabetic foot ulcers, and pressure ulcers.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


