- Home
- »
- Market Trend Reports
- »
-
Nusinersen (Spinraza) Market And Forecast, 2025–2033: Competitive Landscape, Growth Drivers, And Future Outlook
Report Overview
Nusinersen, marketed as Spinraza by Biogen, has significantly impacted the treatment landscape for spinal muscular atrophy (SMA), a rare and severe genetic disorder that leads to motor neuron degeneration. Spinraza's approval in 2016 marked a major milestone in SMA management, providing the first FDA-approved treatment option for this devastating condition. Its market success has been driven by its ability to slow disease progression and improve motor function in both pediatric and adult patients. However, with the expiration of its patent exclusivity on the horizon, the Nusinersen market is entering a transitional phase. This upcoming patent cliff, combined with the emergence of biosimilars and competing treatments, presents both challenges and opportunities for stakeholders in the pharmaceutical industry.
Key Report Deliverables
-
Market Landscape Analysis, Analyze Nusinersen’s current market size and key growth drivers, including the rise of gene therapies and the impact of the upcoming patent expiration in 2029-2030.
-
Market Growth Forecast, forecast market growth considering the potential entry of biosimilars and the impact of competition post-patent expiration.
-
Regulatory and Market Barriers, Assess regulatory challenges and market access barriers for biosimilars and Nusinersen in global markets, particularly in emerging regions.
-
Competitive Landscape, Examine the competitive dynamics, focusing on Biogen’s position against competitors like gene therapies and biosimilars.
-
Regulatory Barriers, Explore the regulatory hurdles for Nusinersen biosimilars, focusing on approval processes and potential delays in market access.
- Strategic Implications, Evaluate Biogen’s strategic moves post-patent expiration, including innovation, geographic expansion, and patient support programs.
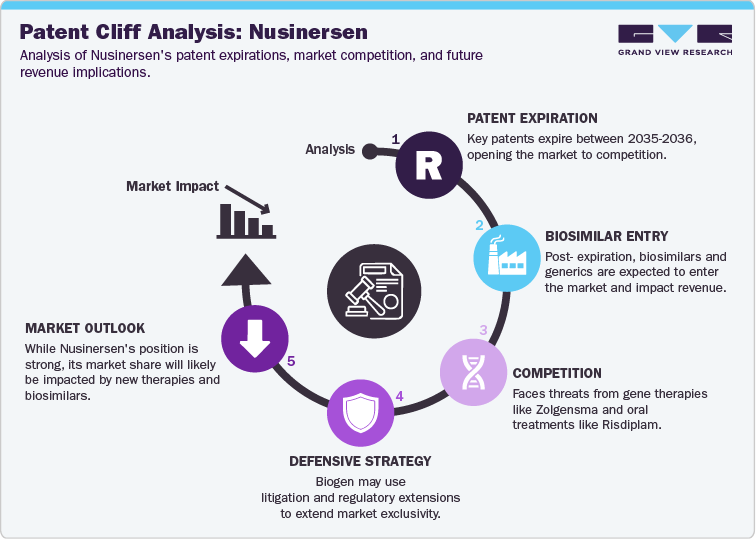
The patent cliff for Nusinersen (Spinraza) is a crucial factor in determining its future market dynamics. The drug is protected by key patents that are set to expire between 2035 and 2036. Specifically, US10436802, which covers methods for treating spinal muscular atrophy (SMA), expires in September 2035, while US12013403, covering compositions and methods for detecting the SMN protein, expires in March 2036. Following these expirations, biosimilars are expected to enter the market, with the potential for generic alternatives to significantly impact Biogen's revenue stream. Additionally, Nusinersen was eligible for patent challenges as of December 2020, which may lead to early entry of generics, depending on legal outcomes. Biogen is likely to pursue strategies to extend its market exclusivity, including patent litigation and seeking regulatory exclusivity extensions.
On the pipeline front, Nusinersen faces increased competition from alternative treatments, especially gene therapies and oral medications. One of the most significant competitors is Zolgensma (Onasemnogene abeparvovec), a one-time gene therapy, which has shown superior outcomes in non-motor functions compared to Nusinersen. Moreover, oral treatments like Risdiplam (Evrysdi) provide patients with a more convenient dosing regimen, making them a viable alternative to Nusinersen’s intrathecal injections. In response, Biogen is seeking regulatory approval for a higher-dose regimen of Nusinersen, which may help maintain its competitive edge. Despite these developments, Nusinersen’s market position remains strong due to its proven efficacy, but its share will likely be impacted as biosimilars and newer therapies gain market traction.

Current Market Scenarios
The Nusinersen (Spinraza) market is currently positioned at a pivotal juncture, with its patent expiration looming, which will significantly alter its market dynamics. Since its approval in 2016, Nusinersen has established itself as a leading biologic therapy for spinal muscular atrophy (SMA), generating substantial revenue due to its efficacy in halting disease progression. The drug has consistently performed well commercially, with annual revenues surpassing USD 1 billion. However, the impending expiration of key patents, scheduled for 2035-2036, is expected to introduce considerable competitive pressures as biosimilars enter the market.
The expiration of Nusinersen’s core patents will open the door for biosimilars, which are expected to directly challenge Biogen’s market position. Several biosimilar candidates are currently under development and are expected to seek approval in the coming years. The introduction of these biosimilars will likely provide more affordable alternatives to the originator product, particularly in price-sensitive markets where cost is a primary barrier to treatment access. Biosimilars typically offer price reductions of 15-30% compared to the original biologic, which will enhance access to SMA therapies, especially in emerging markets where healthcare budgets are constrained.
The regulatory environment surrounding biosimilars is evolving rapidly, with agencies such as the U.S. FDA and the European Medicines Agency (EMA) expediting approval processes to facilitate faster market entry. The approval of biosimilars is expected to contribute to a broader adoption of these therapies, particularly in well-established markets such as the United States and Europe, as well as in emerging markets where access to biologic treatments has traditionally been limited by high costs. These developments will create a more competitive environment, with biosimilars becoming an increasingly significant player in the SMA treatment market.
For Biogen, the challenge will be to maintain its leadership position in the SMA treatment market amidst increasing competition from biosimilars and alternative therapies. In order to preserve market share, Biogen may need to employ strategies such as strengthening brand loyalty through patient support programs, expanding into additional indications, and advancing next-generation treatment options. These efforts will be critical for mitigating the impact of biosimilars and ensuring that Nusinersen retains a competitive edge in the evolving treatment landscape.
Market Dynamics
“Rising Prevalence of Autoimmune Diseases Drives Nusinersen Market Growth”
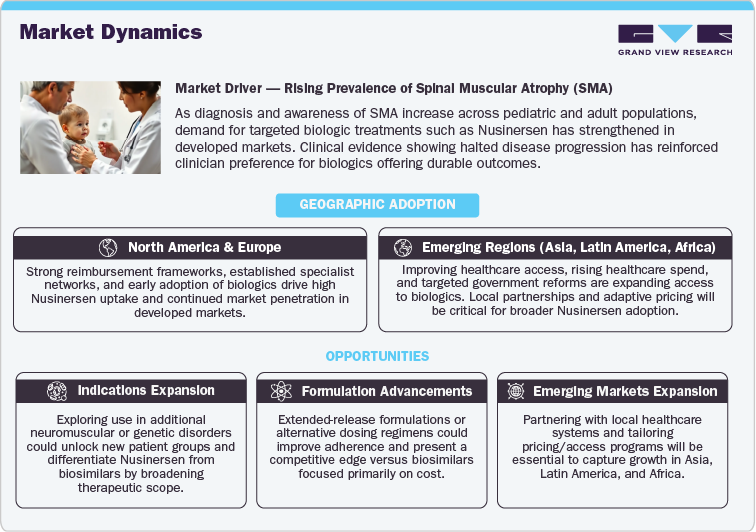
The Nusinersen (Spinraza) market is significantly influenced by the rising prevalence of spinal muscular atrophy (SMA), a rare genetic disorder affecting both pediatric and adult populations. As the incidence of SMA becomes more recognized, the demand for biologic treatments like Nusinersen continues to grow, particularly in developed markets such as North America and Europe. The drug has demonstrated strong clinical efficacy in halting disease progression, consolidating its position as a key treatment for SMA. This demand is supported by healthcare providers' increasing preference for biologics, which offer targeted treatment mechanisms with improved long-term outcomes compared to traditional therapies.
As healthcare access improves in emerging markets, Nusinersen’s adoption is set to rise. These regions, including parts of Asia, Latin America, and Africa, are experiencing enhanced healthcare spending and growing patient awareness, providing a significant opportunity for Nusinersen. Government reforms and increasing healthcare investments are accelerating the adoption of biologics, making Nusinersen more accessible to underserved populations.
“Patent Expiration and Biosimilar Competition Limit Nusinersen Growth”
Despite its strong market presence, Nusinersen faces significant challenges due to the upcoming expiration of its key patents, expected between 2035-2036. Following this, biosimilars will likely enter the market, starting around 2036. These biosimilars will offer cost-effective alternatives, typically priced 15-40% lower than the originator drug, which could drive a shift in market share, particularly in cost-sensitive markets.
The entry of biosimilars will put pressure on Nusinersen’s pricing, especially in regions where healthcare systems are focused on cost containment. This shift will be further amplified by the rapid approval and market entry of biosimilars in emerging markets, where cost is a critical factor in treatment decisions. This competitive dynamic will require Biogen to adjust its pricing and marketing strategies to retain market share and address the growing affordability concerns of healthcare providers and patients.
“Opportunities in Indications, Formulations, and Emerging Markets for Nusinersen Growth”
- Indications Expansion: Nusinersen is currently approved for SMA treatment, but exploring its use in additional neurological or genetic conditions could capture new patient populations. This expansion could further cement Nusinersen’s role in treating rare disorders, differentiating it from emerging biosimilars.
- Formulation Advancements: Development of extended-release formulations or alternative dosing regimens could enhance patient adherence and improve Nusinersen's competitiveness against biosimilars. This strategy would offer a unique value proposition by improving convenience and treatment outcomes for patients.
- Emerging Markets: With increasing healthcare access in regions like Asia, Latin America, and Africa, Nusinersen is well-positioned to capitalize on these markets. Collaborating with local healthcare providers and adapting pricing strategies to meet regional needs will be critical in driving adoption and expanding market reach.
“Post-Patent Competition, Personalized Medicine, and Market Expansion Lift Nusinersen Market”
Post-patent, biosimilars will increase competition in the SMA treatment market, especially in emerging markets where cost plays a significant role in decision-making. Biogen will need to implement robust strategies, including value-based pricing models, to maintain its market leadership.
Additionally, the trend toward personalized medicine presents an opportunity for Nusinersen. Advances in genomic profiling may enable more targeted therapies for SMA, potentially expanding Nusinersen's application in precision medicine. By leveraging companion diagnostics, Biogen can further differentiate Nusinersen from biosimilars, optimizing treatment outcomes for specific patient subgroups.
Overview of Alternative Therapeutics
The Nusinersen (Spinraza) market faces increasing competition from emerging therapies, primarily gene therapies and oral treatments. Zolgensma (Onasemnogene abeparvovec), a one-time gene therapy, offers a potential cure for SMA, presenting a significant competitive challenge to Nusinersen’s ongoing treatment regimen. Additionally, Risdiplam (Evrysdi), an oral treatment for SMA, provides a convenient alternative to Nusinersen’s intrathecal injections, appealing to patients seeking easier administration.
The entry of biosimilars to Nusinersen is also expected, offering lower-cost alternatives to the originator biologic. These biosimilars, when available, will further intensify market competition, particularly in price-sensitive regions. The combined rise of gene therapies, oral treatments, and biosimilars is reshaping the SMA treatment landscape, presenting direct competition to Nusinersen.
Competitive Landscape
The competitive landscape for Nusinersen (Spinraza) is being reshaped by the emergence of gene therapies and oral treatments for spinal muscular atrophy (SMA). The most significant competitor is Zolgensma (Onasemnogene abeparvovec), a one-time gene therapy that addresses the underlying genetic cause of SMA, offering a potential cure with a single administration. Zolgensma’s ability to significantly improve non-motor functions in SMA patients makes it a strong contender, particularly in pediatric populations.
In addition to gene therapies, oral treatments such as Risdiplam (Evrysdi) are increasingly gaining market share. Risdiplam offers a non-invasive, oral administration option that provides a similar therapeutic benefit to Nusinersen. Its ease of use compared to Nusinersen’s intrathecal injections makes it an attractive alternative for both patients and healthcare providers.
With the expiration of Nusinersen's patents expected in the coming years, biosimilars to Nusinersen are likely to enter the market, further intensifying competition. These biosimilars will offer lower-cost alternatives to the originator drug, challenging Nusinersen's pricing structure and market share, especially in cost-sensitive regions. The introduction of these biosimilars will provide a more affordable option for healthcare systems and patients, thereby shifting market dynamics and possibly eroding Biogen's current market leadership.
North America Nusinersen (Spinraza) Market
The North American market for Nusinersen remains a critical revenue driver, supported by strong healthcare infrastructure and reimbursement systems. In the United States, Medicare and private insurers continue to offer favorable coverage for SMA treatments, ensuring broad access to Nusinersen. Despite this, the market faces upcoming competition from emerging gene therapies such as Zolgensma and oral treatments like Risdiplam. As the cost of biologics remains a key issue in U.S. healthcare reforms, the introduction of biosimilars following Nusinersen's patent expiration will likely introduce price competition. However, Nusinersen’s established clinical efficacy and role in treatment guidelines help maintain its market share, though pricing pressures are expected to increase.
Europe Nusinersen (Spinraza) Market
In Europe, Nusinersen continues to be a preferred treatment for SMA, particularly in developed healthcare systems like those in Germany, the UK, and France. The high adoption of biologics for SMA and favorable reimbursement policies support its market presence. However, the expiration of patents and the anticipated entry of biosimilars in the coming years will create competition, particularly in price-sensitive regions. European markets, with a strong focus on cost containment and reimbursement reform, will likely see pricing pressures that could limit Nusinersen’s revenue growth. Despite these challenges, the established clinical outcomes of Nusinersen maintain its position in key markets.
Asia Pacific Nusinersen (Spinraza) Market
The Asia Pacific market is experiencing growth due to improved healthcare infrastructure and increasing awareness of SMA. Developed markets such as Japan show strong demand for Nusinersen, with established regulatory approvals and treatment guidelines. However, emerging markets in China and India face challenges regarding affordability, with the potential for gene therapies and biosimilars to create price competition. Although Nusinersen remains a key treatment in developed regions, the increasing availability of alternative therapies could limit its market share in cost-sensitive areas.
Latin America Nusinersen (Spinraza) Market
In Latin America, the market for Nusinersen is growing, driven by improving healthcare access and rising awareness of SMA. Countries such as Brazil and Mexico are seeing increasing demand for advanced biologic treatments. However, cost remains a significant barrier in many Latin American countries, and the expected introduction of biosimilars could pose a threat to Nusinersen's market share. While the drug’s established efficacy and recognition in SMA treatment support its market position, affordability concerns are likely to influence treatment decisions in cost-sensitive markets.
Middle East And Africa Nusinersen (Spinraza) Market
The Middle East and Africa market for Nusinersen is expanding, with rising awareness of SMA and improving healthcare systems in developed regions like the UAE and Saudi Arabia. The demand for biologic therapies is increasing, but in emerging markets, affordability remains a challenge. Biosimilars and alternative therapies could create competitive pressure on Nusinersen in regions with cost-sensitive healthcare systems. Despite these challenges, Nusinersen's established clinical outcomes and ongoing improvements in healthcare infrastructure support its continued presence in the region.
Analyst Perspective
The Nusinersen (Spinraza) market is expected to face significant challenges due to increasing competition from gene therapies like Zolgensma, oral treatments such as Risdiplam, and the eventual entry of biosimilars following patent expiration. While Nusinersen maintains strong market presence due to its proven efficacy, the rise of one-time gene therapies offering potential cures and the convenience of oral treatments may shift patient preferences. Biosimilars, expected to be priced 15-40% lower than Nusinersen, will put further pressure on its market share, particularly in price-sensitive regions. Biogen must explore strategic approaches, including differentiated dosing regimens, expanding indications, and enhancing patient support, while addressing regional pricing challenges to retain market leadership in an increasingly competitive landscape.
Case Study (Recent Engagement): Keytruda Patent-Cliff & Price- Erosion Impact Model
PROJECT OBJECTIVE
To evaluate the potential revenue, price, and patient access implications of Keytruda’s 2028 patent cliff, incorporating biosimilar entry dynamics, country-specific adoption curves, and Merck’s lifecycle defense strategies (remarkably the subcutaneous formulation). The goal was to provide the client with a transparent, scenario-based model to anticipate outcomes and inform strategy
GVR SOLUTION
-
Built a bottom-up commodity-flow and analogue-based model, anchored on Merck’s $29.5B Keytruda sales in 2024.
-
Integrated jurisdictional LOE timelines (EU mid-2028, U.S. 2028-2029 pending litigation outcomes).
-
Modeled biosimilar adoption S-curves calibrated to oncology antibody analogues (EU faster via tenders, U.S. slower via contracting).
-
Applied price-erosion benchmarks (EU −15-30% Yr-1, deepening to −45-60% by Yr-3; U.S. −10-25% net decline over same horizon).
-
Layered lifecycle defenses (SC uptake assumptions of 25-40% of innovator units, combo refresh, contracting) to quantify buffers.
-
Delivered outputs as a dynamic Excel scenario tool and a management-ready PPT deck with revenue bridges, sensitivity tornadoes, and SC migration visuals.
IMPACT FOR CLIENT
-
Enabled the client to quantify downside vs. defense-optimized revenue trajectories:
-
Base case: 30-40% global revenue decline by Year-3 post-LOE.
-
Downside: 45-55% decline in tender-heavy markets.
-
Defense-optimized: Contained erosion to ~−20-25% with strong SC adoption.
-
-
Gave the client a clear view of which markets drive early erosion (EU) and where strategic contracting or SC migration can preserve share (U.S.).
-
Equipped decision-makers with a playbook of watch-points (tender concentration, litigation outcomes, SC IP coverage, combo pipeline) to guide commercial strategy.
-
Provided a transparent methodology that could be presented to boards/investors with evidence-backed assumptions
WHY THIS MATTERS
-
Keytruda is the world’s best-selling cancer drug, representing nearly one-third of Merck’s revenue.
-
Patent expiry will reshape both Merck’s earnings profile and global oncology access dynamics.
-
Payers and governments stand to benefit from biosimilar entry through lower costs, but manufacturers need to manage cliff risk while capturing upside from lifecycle innovations.
-
Understanding how quickly revenues erode and how patient access expands post-biosimilar is critical for:
-
Biopharma companies (strategic planning, pipeline prioritization).
-
Investors (valuing Merck’s cash flows beyond 2028).
-
Payers and policymakers (budgeting for oncology drug spend).
-
A robust patent cliff model helps clients navigate the dual challenge of price erosion and patient expansion, ensuring strategies are grounded in real-world benchmarks.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


