- Home
- »
- Market Trend Reports
- »
-
Top 20 Immunology Drugs Market Analysis And Segment Forecasts To 2030
Immunology Drugs Market Size & Trends
The global top 20 immunology drugs market continues to experience strong growth, driven by rising incidence rates, advancements in targeted therapies, and increased awareness and screening efforts worldwide. In 2024, the combined sales of the top 20 immunology drugs was estimated at USD 81.18 billion and is projected to grow at a CAGR of 9.41% from 2025 to 2030. These top-performing drugs span a wide range of therapeutic classes-including Humira, Dupixent, Stelara, Skyrizi, Entyvio, Cosentyx, Enbrel, Xolair, Rinvoq, and Orencia-highlighting the diversity and complexity of modern immunological disease treatment approaches.
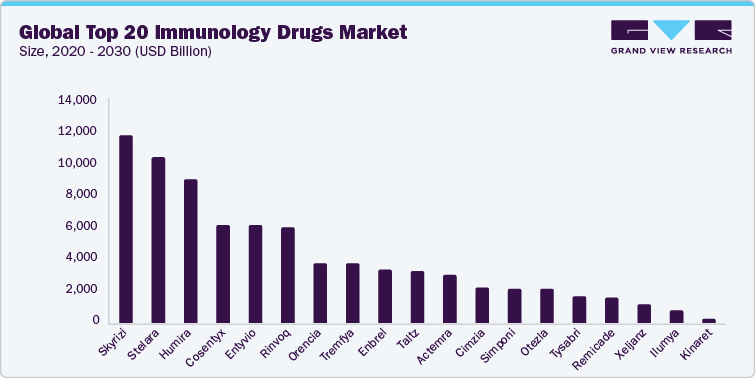
The immunology drug landscape is rapidly evolving, driven by breakthroughs in biologics, improved diagnostic capabilities, and a deeper understanding of immune-mediated diseases. While established therapies such as Humira and Stelara continue to anchor the market, newer entrants like Rinvoq and Skyrizi are gaining traction through favorable clinical profiles, expanded indications, and differentiated mechanisms of action. This shift is further accelerated by the patent expirations of legacy products, fueling a transition towards next-generation therapies with better safety, efficacy, and convenience.
The market is also benefiting from the rising global prevalence of chronic autoimmune conditions such as rheumatoid arthritis, psoriasis, ulcerative colitis, and Crohn’s disease. This trend is especially prominent in developed regions with aging populations and in emerging economies undergoing lifestyle transitions. Meanwhile, advances in personalized medicine, biomarker-driven treatment approaches, and combination immunotherapies are reshaping clinical practice and opening new avenues for drug development.
Between 2025 and 2030, the competitive dynamics of the top 20 immunology drugs will be shaped by factors such as lifecycle management strategies, payer pressures, biosimilar entry, and regulatory shifts. However, ongoing innovation, growing patient access, and evolving treatment paradigms are expected to sustain long-term growth and drive differentiation in this high-value therapeutic area.
Top 20 Immunology Drugs Market, 2024 (USD Million)
Drug Name (Generic Name)
Company
2024 Revenue (USD Billion)
2030 Revenue (USD Billion)
Date of First Regulatory Approval
Approved Indications
Approximate Annual Cost (USD)
Generics/Biosimilars Available
Skyrizi (Risankizumab)
AbbVie Inc.
11.71
39.82
23-Apr-2019 (U.S. FDA)
plaque psoriasis, psoriatic arthritis, Crohn’s disease
~$90,000
No
Stelara (Ustekinumab)
Johnson & Johnson Service Inc.
10.36
8.23
25-Sep-2009 (U.S. FDA)
Plaque psoriasis, psoriatic arthritis, Crohn’s disease, ulcerative colitis
~$120,000
yes
Humira (Adalimumab)
*Similar analysis would be provided for key 20 immunology drugs
Cosentyx (Secukinumab)
Entyvio (Vedolizumab)
Rinvoq (Upadacitinib)
Orencia (Abatacept)
Tremfya (Guselkumab)
Enbrel (Etanercept)
Taltz (Ixekizumab)
Actemra (Tocilizumab)
Cimzia (Certolizumab pegol)
Simponi (Golimumab)
Otezla (Apremilast)
Tysabri (Natalizumab)
Remicade (Infliximab)
Xeljanz (Tofacitinib)
Ilumya (Tildrakizumab)
Kinaret (Anakinra)
Dupixent (Dupilumab)
Several powerful factors are driving sustained growth in the immunology drug market, particularly among the top 20 therapies. Rising global prevalence of chronic inflammatory and autoimmune conditions, such as rheumatoid arthritis, psoriasis, ulcerative colitis, and Crohn’s disease, is expanding the patient base across developed and emerging regions. At the same time, earlier diagnosis and improved disease awareness have facilitated timely therapeutic intervention, increasing long-term treatment uptake.
Biologic therapies continue to dominate the space, with newer-generation agents like IL-17, IL-23, and JAK inhibitors demonstrating improved efficacy and convenience over legacy TNF inhibitors. Innovation in drug delivery methods, including subcutaneous and oral options, has also enhanced patient adherence. Furthermore, the move toward personalized treatment regimens-guided by immunophenotyping and disease activity scoring has created a more tailored approach to care, increasing the demand for premium-priced therapies that offer sustained remission and safety advantages.
Patent Expiry & Biosimilar Impact
Patent expiries are reshaping the immunology market, with key blockbusters like Humira, Enbrel, Remicade, and soon Stelara, facing growing biosimilar competition. This has led to steep price drops and loss of market share for originator drugs, especially in cost-sensitive and payer-driven markets. While newer therapies like Skyrizi and Rinvoq remain protected, manufacturers are relying on lifecycle strategies and expanded indications to retain value. As biosimilars gain traction, managing affordability while sustaining innovation is becoming crucial.
Table: Key Patent Expiries & Biosimilar Landscape
Drug
Originator
Indication
Patent Expiry
Biosimilar Status
Skyrizi
AbbVie Inc.
Psoriatic Arthritis, Crohn’s Disease
~2031-2034 (U.S./EU)
No biosimilars yet
Stelara
Johnson & Johnson Services Inc.
Psoriatic Arthritis, Crohn’s, Ulcerative Colitis
2023 (EU), 2025 (U.S.)
Biosimilars in late-stage development
Humira
AbbVie Inc.
RA, PsA, UC, CD, Psoriasis, others
Expired (EU 2018, U.S. 2023)
Multiple biosimilars approved & marketed
Cosentyx
Novartis AG
Psoriasis, PsA, AS
~2028-2029
No biosimilars yet
Entyvio
Takeda Pharmaceutical Company Limited
Ulcerative Colitis, Crohn’s Disease
~2026-2027
Biosimilars in development
Rinvoq
AbbVie Inc.
RA, PsA, Atopic Dermatitis, UC, CD
~2030-2033
No biosimilars yet
Orencia
Bristol Myers Squibb
RA, Juvenile Idiopathic Arthritis, PsA
Expired (U.S. 2021, EU earlier)
Biosimilars approved (EU); U.S. under review
Tremfya
Johnson & Johnson Services Inc.
Psoriasis, PsA
~2031
No biosimilars yet
Enbrel
Amgen / Pfizer
RA, PsA, AS, Psoriasis
Expired (EU 2015); U.S. 2029 (disputed)
Biosimilars approved (EU); U.S. pending litigation
Taltz
Eli Lilly and Company
Psoriasis, PsA, Axial Spondyloarthritis
~2026-2028
No biosimilars yet
The immunology drug market is entering a pivotal phase as patent cliffs approach for several high-value biologics. As leading therapies like Stelara, Cosentyx, and Entyvio near the loss of exclusivity, manufacturers are bracing for a new wave of biosimilar entrants. This shift is intensifying competition and driving a transition from volume-driven growth to value-based differentiation. Companies are increasingly adopting proactive lifecycle strategies, such as next-generation biologics, targeted mechanisms (e.g., IL-23 over TNF), and shifting toward oral or less frequent dosing regimens, to defend market share and sustain margins.
Pipeline & Innovation Spotlight
The immunology space continues to witness a robust pipeline of next-generation therapies aimed at enhancing efficacy, safety, and patient convenience. Drug developers are increasingly focused on differentiated mechanisms of action, including selective TYK2 inhibitors, novel interleukin targets (like IL-36 and IL-33), and precision oral immunomodulators that could disrupt the dominance of current biologics. Emerging assets aim to address unmet needs such as non-responders to TNF inhibitors, steroid-sparing treatment goals, and improved long-term disease control.
As regulatory agencies evolve frameworks for faster review of novel immunotherapies and biosimilars, the next five years are expected to bring an influx of differentiated, value-based immunology solutions into the market, reshaping standards of care across chronic autoimmune diseases.
Regulatory & Policy Environment
The regulatory framework governing immunology drugs is evolving to accommodate the growing pipeline of complex biologics, biosimilars, and targeted therapies addressing chronic autoimmune and inflammatory diseases. Regulatory bodies such as the U.S. FDA, EMA, and PMDA (Japan) are increasingly emphasizing scientific rigor and long-term safety in approving immunology drugs, particularly for conditions like rheumatoid arthritis, psoriasis, inflammatory bowel disease (IBD), and atopic dermatitis, where treatment is often lifelong.
Moreover, the biosimilar approval pathway is gaining traction, particularly in the EU and emerging Asia-Pacific markets, where regulatory frameworks for biosimilars are more mature. Agencies are also harmonizing guidelines on interchangeability, extrapolation of indications, and pharmacovigilance to streamline entry and ensure therapeutic equivalence. However, regional disparities remain in biosimilar adoption and regulatory clarity-most notably in parts of the Middle East and Sub-Saharan Africa, where biosimilar frameworks are still under development. Overall, regulatory agencies are striking a careful balance between innovation and sustainability in the immunology drug landscape.
Pricing & Reimbursement Landscape
Immunology drugs, particularly biologics like Humira, Stelara, Dupixent, and Cosentyx, are among the highest-grossing yet cost-intensive therapies in global pharmaceutical markets. In mature healthcare systems, these drugs are subject to intensive health technology assessments (HTAs), where reimbursement decisions are increasingly linked to comparative effectiveness, long-term outcomes, and budget impact. Agencies such as NICE (UK), HAS (France), and G-BA (Germany) are applying strict cost-effectiveness thresholds, while also encouraging competitive tendering and biosimilar uptake to reduce system burden.
In middle- and lower-income markets, affordability challenges persist, prompting tiered pricing, public-private access programs, and localized manufacturing to enhance accessibility. Increasingly, pharmaceutical manufacturers are adopting risk-sharing agreements, outcome-based contracting, and indication-based pricing to align value delivery with reimbursement. With growing fiscal pressure across global health systems, pricing strategies for immunology drugs must now deliver both economic efficiency and sustained therapeutic value.
Route of Administration Insights
The immunology drugs market is segmented into oral, intravenous (IV), and subcutaneous (SC) routes of administration, with each route serving specific clinical needs based on drug formulation, efficacy, patient convenience, and safety profiles. As of 2024, the intravenous (IV) route continues to dominate the market. It has emerged as the most widely adopted route across chronic immunological conditions such as rheumatoid arthritis, psoriasis, and ulcerative colitis, largely due to its balance of efficacy, convenience, and growing support from at-home self-injection solutions.
Leading immunology biologics, such as Humira, Enbrel, Dupixent, Cosentyx, and Stelara are predominantly administered via subcutaneous injection, empowering patients with greater autonomy through pre-filled syringes and auto-injectors. The rising preference for SC delivery reflects the broader industry trend toward decentralized care and patient-friendly regimens, especially in long-term disease management.
Oral immunology therapies are gaining momentum as a transformative option in specific segments of the market. The introduction and uptake of JAK inhibitors (e.g., Xeljanz, Rinvoq, Olumiant) and S1P modulators (e.g., Zeposia) have expanded oral treatment options for diseases like rheumatoid arthritis, ulcerative colitis, and atopic dermatitis. With their ease of use and rapid onset of action, oral drugs are increasingly being integrated into treatment algorithms, particularly in younger or working-age populations seeking low-disruption therapy options.
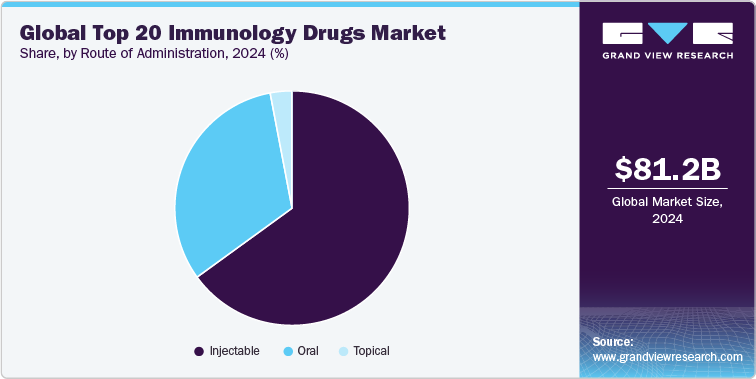
Looking ahead, the shift toward self-administration and home-based care will continue to drive innovation in SC formulations, device-based delivery systems, and patient training support. Meanwhile, oral therapies are set to be the fastest-growing segment from 2025 to 2030, driven by novel small molecules with favorable safety and pharmacokinetic profiles. Overall, the evolving route-of-administration landscape is not only reshaping market share but also redefining the immunology treatment experience across diverse patient populations.
Key Companies & Market Share Insights
The top 20 immunology drugs market is dominated by a select group of multinational pharmaceutical giants, many of whom have established leadership positions through their innovative therapies, extensive clinical research, and robust marketing strategies. The market remains highly competitive, with companies striving to enhance patient outcomes through targeted immunotherapy treatment.
Leading Companies by Market Share
- AbbVie Inc.
- Johnson & Johnson Inc.
- Eli Lilly and Company
- Novartis AG
- Pfizer Inc.
- Sanofi
- Amgen Inc.
- Takeda Pharmaceutical Company Limited.
- Bristol-Myers Squibb
- GSK plc.
- F. Hoffmann-La Roche Ltd
- AstraZeneca
- Biogen
- UCB Pharma
- Sun Pharma (Ilumya)
Market Share Insights
-
F. Hoffmann-La Roche Ltd continues to lead the global breast cancer therapeutics market, particularly in the HER2-positive segment, driven by established blockbusters like Orencia, Humira, Entyvio, and the fast-growing Dupixent.
-
Pfizer remains a dominant player with Stelara, the top-selling CDK4/6 inhibitor, while Novartis and Eli Lilly follow closely with Cosentyx and Skyrizi, respectively.
-
AstraZeneca is rapidly gaining market share through Dupixent (in collaboration with Daiichi Sankyo) and Actemra, targeting HER2-low and BRCA-mutated breast cancer patients.
-
Merck & Co. and Bristol-Myers Squibb are actively expanding their presence in breast cancer with immunotherapy agents (Enbrel, Opdivo).
-
Amgen and Johnson & Johnson hold strong positions in bone metastasis management, with Tysabri and Cimzia being widely adopted.
Key Top 20 Immunology Drugs Market Companies:
The following are the leading companies in the top 20 immunology drugs market. These companies collectively hold the largest market share and dictate industry trends.
- F. Hoffmann-La Roche Ltd
- Novartis AG
- Pfizer Inc.
- AstraZeneca
- Merck & Co., Inc.
- Johnson & Johnson
- Bristol-Myers Squibb Company
- Sanofi
- GSK plc.
- AbbVie Inc.
- Takeda Pharmaceutical Co.
- Biogen
- Eli Lilly and Company
- Novo Nordisk A/S
- Amgen Inc.
Recent Developments
-
In April, 2025, the U.S. Food and Drug Administration approved a prefilled syringe formulation of Argenx SE's immune disorder drug Vyvgart. This new version allows for at-home self-administration, enhancing convenience for patients with generalized myasthenia gravis (gMG) and chronic inflammatory demyelinating polyneuropathy (CIDP). The approval is expected to broaden Vyvgart's market reach and improve patient adherence
-
In April 2025, AstraZeneca's subsidiary, Alexion Pharmaceuticals, is facing a class-action lawsuit filed by EmblemHealth, alleging that the company manipulated the patent system to unlawfully extend its monopoly on the high-cost drug Soliris. The lawsuit claims these actions delayed the introduction of cheaper biosimilars, potentially leading to overpayments exceeding USD 2 billion
-
In May 2024, Novartis AG announced new clinical data confirming the long-term efficacy and safety of remibrutinib, a highly selective Bruton’s tyrosine kinase (BTK) inhibitor, in the treatment of chronic spontaneous urticaria (CSU).
Segments Covered In The Report
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global top 20 immunology drugs market report based on product, route of administration, and region:
-
Product Outlook (Revenue, USD Million, 2018 - 2030)
-
Skyrizi
-
Stelara
-
Humira
-
Cosentyx
-
Entyvio
-
Rinvoq
-
Orencia
-
Tremfya
-
Enbrel
-
Taltz
-
Actemra
-
Cimzia
-
Simponi
-
Otezla
-
TYSABRI
-
Remicade
-
Xeljanz
-
Ilumya
-
Kinaret
-
Dupixent
-
-
Route of Administration Outlook (Revenue, USD Million, 2018 - 2030)
-
Topical
-
Injectable
-
Oral
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


