- Home
- »
- Medical Devices
- »
-
Anti-snoring Devices & Snoring Surgery Market Report, 2030GVR Report cover
![Anti-snoring Devices And Snoring Surgery Market Size, Share & Trends Report]()
Anti-snoring Devices And Snoring Surgery Market Size, Share & Trends Analysis Report By Type (Oral Appliances/ Mouthpieces, Position Control Devices, Nasal Devices, Chin Straps, Tongue Stabilizing Devices), By Region, And Segment Forecasts, 2023 - 2030
- Report ID: 978-1-68038-972-2
- Number of Report Pages: 110
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
Report Overview
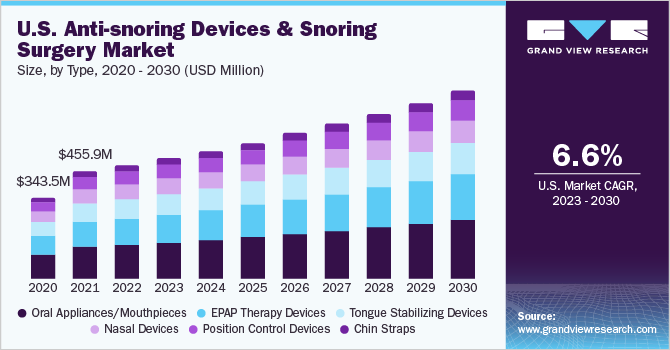
The global anti-snoring devices and snoring surgery market size was estimated at USD 1.52 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 7.3% from 2023 to 2030. Increasing incidence of obesity, the launch of technologically advanced devices, preference for non-invasive treatments, the availability of minimally invasive surgeries, strategic activities by key market players, and easy availability of products through online and offline channels are key factors driving the growth of the market for anti-snoring devices and snoring surgery.
Snoring is an early sign of sleep apnea, an acute condition characterized by disturbed breathing throughout the night, which can result in major long-term health consequences. Obstructive sleep apnea (OSA) elevates the risk of stroke, cardiovascular illness, cardiac arrhythmia, and hypertension. According to the American Medical Association estimates, in 2023 over 30 million people in the U.S. suffer from sleep apnea and OSA is the primary reason for the cause.
The rise in the incidence of obesity has become one of the major concerns, globally. According to the CDC, the U.S. obesity prevalence between the years 2017 and 2020 was 41.9%. It also states that the prevalence of severe obesity increased from 4.7% between 1999-2000 to 9.2% through 2017-2020. Factors, such as a sedentary lifestyle, increasing consumption of alcohol and smoking, and consumption of unhealthy & processed foods are leading to a rise in the number of individuals with obesity problems. Obesity-related problems include stroke, type 2 diabetes, heart disease, and certain types of cancers, which are among the leading cause of premature preventable death.
People suffering from obesity may also suffer from snoring issues and sleep apnea because of the neck fat present. When lying down it causes the upper airway to constrict, which greatly increases the possibility of snoring. Thus, the increasing prevalence of obesity due to bad eating among the population also drives the market growth. As awareness about the health risks associated with snoring and sleep apnea is increasing, more individuals are seeking medical help and getting diagnosed.
This has led to a higher demand for snoring surgeries as a treatment option. There have been advancements in surgical techniques for snoring treatment, including minimally invasive procedures such as radiofrequency ablation (RFA), laser-assisted uvulopalatoplasty (LAUP), and palatal implants. These techniques offer reduced post-operative discomfort, shorter recovery times, and improved outcomes, making surgery a more attractive option for patients.
The COVID-19 pandemic had a slight negative impact on the market. This is because, while many studies concluded that patients with obstructive sleep apnea are vulnerable to adverse post-infection outcomes, such as obesity, hypertension, and idiopathic pulmonary fibrosis, which resulted in a higher number of people being aware and opting for treatment. The pandemic led to a fall in sales of anti-snoring devices. The fall in sales can be attributed to a reduced number of elective surgeries being carried out.
Moreover, severe disruption in the supply chain due to the lockdowns and restrictions, which focused on only necessary movements being carried out. The pandemic also resulted in fewer hospital visits by patients. However, the market began to recover in 2021 with the opening of economies and governments coming up with better initiatives to control the spread of the virus as well as its consequences.
Type Insights
The oral appliances/mouthpieces segment held the largest revenue share of 30.0% in 2022. Factors such as awareness about mouthpieces among dentists & patients, increasing adoption of oral hygiene practices, and the introduction of advanced products are some of the factors driving the market growth. The position-control devices segment is expected to grow at the fastest CAGR of 8.6% over the forecast period.
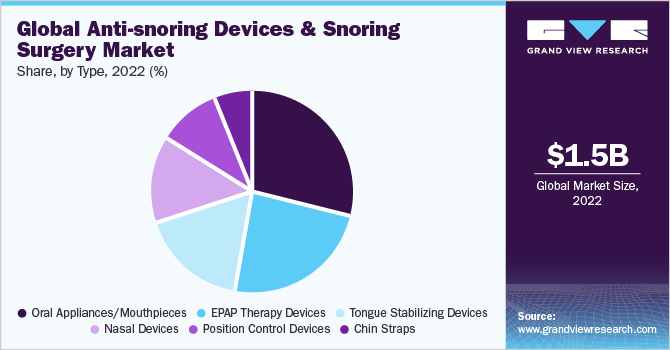
The EPAP therapy devices segment held a considerable market share in terms of revenue, in 2022. Due to the launch of new products such as the Bongo Rx and Smart Nora, which is a smart pillow that helps to adjust your sleep position. In July 2022, SleepRes, LLC. launched a new accessory V-Com. It is inserted in between the CPAP mask and hose and is designed to lower inspiratory pressure and flow, hence enhancing comfort with little to no decrease in expiratory pressure (EPAP).
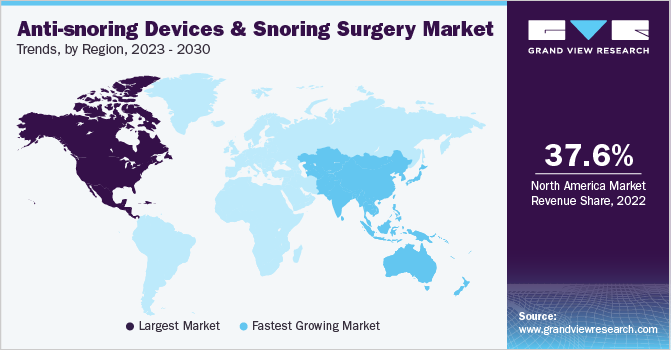
Regional Insights
North America dominated the market and accounted for the largest revenue share of 37.6% in 2022. This can be attributed to several individuals experiencing snoring in the U.S. According to an article published by John Hopkins University in 2022, an estimated 45.0% of adults snore occasionally, while 25.0% snore regularly. Another research article by Sleep Foundation states that in 2020, an estimated 57.0% of men and 40.0% of women in the U.S. were affected by snoring issues. It also stated that even an estimated 27.0% of children were also affected by snoring problems.
A major factor leading to a rise in the number of individuals being affected with snoring issues in the region is growing obesity, it is a major concern, especially in the U.S., caused mainly due to the growing adoption of a sedentary lifestyle and an increase in the consumption of processed foods. The presence of market players in this region and the availability of products on e-commerce sites as well as in retail stores is expected to boost market penetration and product reach.
Asia Pacific is expected to grow at the fastest CAGR of 8.3% during the forecast period due to the rising awareness regarding this condition and the presence of a large number of patients with untreated snoring issues. According to an article released by the International Journal of Health Sciences and Research in September 2022, the overall prevalence of obesity in India was 40.3%, indicating that more than two-thirds of obese people have OSA. Growing disposable income levels are also expected to boost product demand in Asia Pacific.
Key Companies & Market Share Insights
Key players are focusing on expanding their reach in the market through the introduction of new products. For instance, in June 2022, Illusion Aligners introduced a new anti-snoring device, which is an extremely lightweight and simple-to-use oral anti-snoring device that alleviates OSA symptoms as well as improves sound sleep, lowers fatigue, and improves the overall quality of life.
In February 2021, Signifier Medical Technologies, LLC, received FDA approval for the marketing of a device to reduce mild obstructive sleep apnea and snoring in patients aged 18 years & above. Some prominent players in the global anti-snoring devices and snoring surgery market include:
-
Apnea Sciences Corp.
-
SomnoMed Ltd.
-
Airway Management, Inc.
-
ResMed Inc.
-
Fisher & Paykel Healthcare Corp. Ltd.
-
Koninklijke Philips NV
-
Sleep Well Enjoy Life, Ltd.
-
Tomed Dr. Toussaint GmbH
-
MEDiTAS Ltd.
Anti-snoring Devices And Snoring Surgery Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 1.62 billion
Revenue forecast in 2030
USD 2.65 billion
Growth rate
CAGR of 7.3% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Report updated
August 2023
Quantitative units
Revenue in USD million/billion, and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Type, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Apnea Sciences Corp.; SomnoMed Ltd.; Airway Management, Inc.; ResMed Inc.; Fisher & Paykel Healthcare Corp. Ltd.; Koninklijke Philips NV; Sleep Well Enjoy Life, Ltd.; Tomed Dr. Toussaint GmbH; MEDiTAS Ltd
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Anti-snoring Devices And Snoring Surgery Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global anti-snoring devices and snoring surgery market report based on type, and region:
-
Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Oral Appliances/Mouthpieces
-
Nasal Devices
-
Position Control Devices
-
Chin Straps
-
Tongue Stabilizing Devices
-
EPAP Therapy Devices
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Norway
-
Denmark
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East and Africa
-
Saudi Arabia
-
South Africa
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global anti-snoring devices and snoring surgery market size was estimated at USD 1.52 billion in 2022 and is expected to reach USD 1.62 billion in 2023
b. The global anti-snoring devices and snoring surgery market is expected to grow at a compound annual growth rate of 7.3% from 2023 to 2030 to reach USD 2.65 billion by 2030.
b. North America dominated the anti-snoring devices and snoring surgery market with a share of 37.6% in 2022. This is attributable to large share are the rising awareness regarding the detrimental effects of this condition, presence of a large number of players in this sector, and an untapped population suffering from this condition.
b. Some key players operating in the anti-snoring devices and snoring surgery market include Apnea Sciences Corporation, SomnoMed Ltd., Airway Management, Inc., ResMed Inc., Fisher & Paykel Healthcare Corp. Ltd., Koninklijke Philips nv, Sleep Well Enjoy Life, Ltd., Tomed Dr. Toussaint GmbH and MEDiTAS Ltd.
b. Key factors that are driving the anti-snoring devices and snoring surgery market growth include increasing awareness about the ill effects of snoring, soaring geriatric and obese population, presence of an untapped population pool characterized with a snoring condition, and the increasing consumption of alcohol and cigarettes.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





