- Home
- »
- Medical Devices
- »
-
Australia & New Zealand Healthcare CROs Market Report 2030GVR Report cover
![Australia & New Zealand Healthcare CROs Market Size, Share & Trends Report]()
Australia & New Zealand Healthcare CROs Market Size, Share & Trends Analysis Report By Phase Type (Drug Discovery, Preclinical, Clinical), By Service, By Application, By End-use, By Country, And Segment Forecasts, 2023 - 2030
- Report ID: GVR-4-68040-038-8
- Number of Report Pages: 122
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
Report Overview
The Australia & New Zealand healthcare CROs market size was valued at USD 1.0 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 11.5% from 2023 to 2030. Increasing adoption of advanced technologies; rising strategic initiatives such as geographical expansions, mergers & acquisitions, and R&D grants & tax incentives; and cost-effectiveness & favorable environment for clinical trials are a few of the key factors boosting the demand for CROs. The COVID-19 pandemic resulted in every country experiencing a different situation. While some suffered high infection rates and a high number of fatalities, others like Australia & New Zealand did not experience such a severe situation.
Australia and New Zealand had a zero-tolerance attitude toward COVID-19, and it was their governments' strong response to the pandemic that kept both infection and mortality rates relatively low. The governments of the two countries acted swiftly to close their borders and imposed quarantine measures on any returning resident to prevent the spread of the disease. The government also put in place extensive testing and contact tracing.
The revenue growth in the healthcare contract research organizations industry has been boosted by the adoption of virtual clinical trials, which has become one of the major factors post-pandemic. Many CROs are adopting virtual technologies to streamline their clinical research services. The latest Annual CRO Report by the Veeva Unified Clinical Operations Survey suggests that CROs are making significant advancements to speed up and modernize clinical trials.
Data from Veeva Systems indicates that CROs have been implementing cutting-edge digital strategies and technologies to improve trial execution, eliminate information silos, replace manual processes, and promote trial collaborations. For instance, in February 2022, Parexel announced the extension of its partnership with Medidata to enhance the delivery of DCTs for the life sciences industry.
Medidata's DCT solutions support Parexel's efforts in a market where over 75% of submissions include at least a hybrid trial style. It will gain a lot from the partnership because Medidata's DCT solutions will increase the company's product offerings in the DCT sector.
One of the most substantial R&D tax advantages for clinical trials is offered in Australia. Furthermore, as stated by Clinexa Life Sciences on its website's strategic consulting solutions R&D Tax Incentive section, the Australian government offered a cash refund of 43.5% tax credit for companies with an annual turnover of less than AUD 20 million and a 38.5% nonrefundable tax credit for businesses with an annual turnover of over AUD 20 million, for eligible R&D expenditure.
Such tax incentives can help increase the number of industry-sponsored clinical trials, eventually boosting the demand among CRO clients, which have primarily U.S. and Asian sponsors, to perform clinical trials in Australia.
Along with R&D grants and incentives, the Australia and New Zealand CRO market is supported by its cost-effectiveness in performing clinical trials. For instance, cost comparison indicates that Australian clinical trials are 60% less expensive than those conducted in the U.S., especially for companies that can fully utilize R&D incentives.
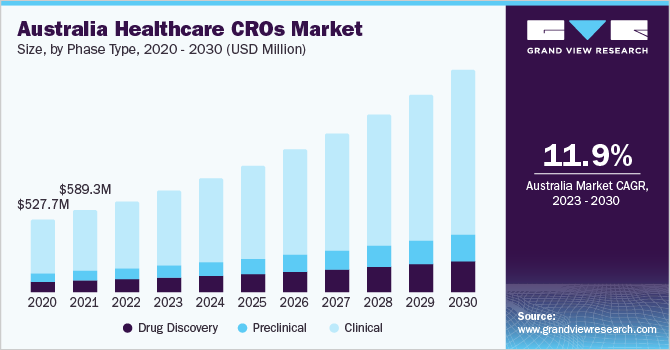
Phase Type Insights
In 2022, the clinical services segment held the highest revenue share of 76.1% in the Australia & New Zealand healthcare contract research organizations market. The growing demand for new treatments for epidemic events, personalized medicines, and orphan drugs, coupled with the increasing number of biologics and the need for advanced technologies, are the primary factors driving this growth.
Technological advancements, globalization of clinical trials, and the increasing demand for contract research organizations to conduct clinical trials are expected to further fuel this growth. Additionally, outsourcing Phase II clinical trials to healthcare contract research organizations is generating the highest revenue due to a rise in industry-sponsored and non-industry-sponsored clinical trials in Phase II, along with the complexity associated with these trials.
The drug discovery segment is anticipated to witness a significant growth rate of 12.5% during the forecast period. Australia and New Zealand have well-established pharmaceutical industries with a strong focus on research and development. Additionally, their favorable regulatory frameworks for drug development are likely to attract investment from multinational pharmaceutical companies.
Service Insights
In 2022, the clinical monitoring segment was the leading revenue generator, accounting for a 20.5% share. The growth of this segment can be attributed to the increasing number of clinical trials and the need for monitoring these studies, which has led to a rise in demand for related services.
The trend of outsourcing clinical research to CROs has gained momentum over the past decade due to the cost-effectiveness and technical expertise provided by such services. With the advent of smart analytics and real-time data acquisition devices, clinical monitoring data in the healthcare sector is expected to improve. The early detection of trial errors related to drug safety and toxicity through real-time data acquisition enables timely rectifications such as trial re-design or termination, leading to further growth of this segment.
Over the forecast period, the regulatory/medical affairs segment is expected to experience the highest growth rate of 10.8% in the Australia & New Zealand healthcare contract research organization market. The outsourcing of regulatory affairs has increased significantly due to the rise in R&D activities, clinical trial applications, product registration, and drug pipeline. The segment's growth is supported by the increasing demand to obtain approval for new products, maintain compliance, and optimize resources.
Application Insight
The oncology segment dominated the market with the largest revenue share of 40.2% in 2022. The segment is also anticipated to witness the fastest CAGR over the forecast period. In recent years, the field of oncology has seen a significant increase in R&D activities, with a focus on discovering new and more effective treatments for cancer. This has led to a growing demand for CROs that specialize in oncology research in Australia and New Zealand.
The respiratory diseases segment, categorized based on the application, on the other hand, is anticipated to witness a lucrative growth rate of 12.4% across the forecast period in the Australia & New Zealand healthcare contract research organizations market. The high prevalence of respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), and respiratory infections in Australia and New Zealand is the primary driver of this growth.
End-use Insight
Based on the end-user, the market is classified into pharmaceutical and biopharmaceutical companies and medical device companies. The pharmaceutical & biopharmaceutical companies segment dominated the market with the largest revenue share of 82.83% in 2022. The segment is also expected to experience the fastest CAGR over the forecast period, which can be attributed to the rising R&D expenditures & increasing approvals of new pharmaceuticals and biopharmaceutical products over the past two decades.
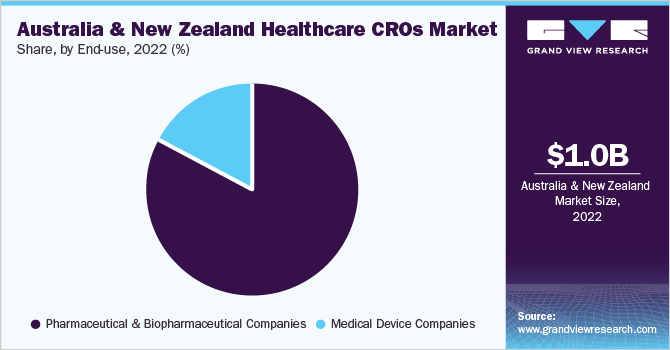
The medical device companies segment, on the other hand, is anticipated to witness a stable growth rate of 10.4% across the forecast period, owing to the increasing focus on developing technologically advanced medical devices, increasing patient preference for minimally invasive surgical procedures, and the growing number of chronic diseases requiring medical devices.
Key Companies & Market Share Insights
To achieve a competitive edge, the industry players implement various strategic measures. Vital factors that affect competitiveness are product launches, mergers and acquisitions, geographic expansions, and partnerships. An example of such a strategic move is the acquisition of Model Answers, a Brisbane-based consultancy firm, by Parexel in February 2020. Model Answers delivers pharmacokinetic (PK) and pharmacodynamic (PD) modeling, simulation, and analysis services to life sciences customers, boosting the probability of successful drug development. Some prominent players in the Australia and New Zealand healthcare CROs market include:
-
PAREXEL International Corporation
-
Thermo Fischer Scientific, Inc.
-
SGS SA
-
ICON plc
-
Medpace Australia Pty. Ltd
-
Charles River Laboratories International, Inc.
-
IQVIA
-
Novotech
-
Laboratory Corporation of America Holdings
-
Syneos Health
Australia & New Zealand Healthcare CROs Market Report Scope
Report Attribute
Details
Market Size value in 2023
USD 1.1 billion
Revenue forecast in 2030
USD 2.4 billion
Growth Rate
CAGR 11.5% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Quantitative units
Revenue in USD million and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase type, service, application, end-use, country
Country scope
Australia; New Zealand
Key companies profiled
Thermo Fisher Scientific, Inc. (PPD); SGS; ICON plc; Medpace Australia Pty. Ltd.; Charles River Laboratories; IQVIA; Novotech; Labcorp Drug Development; Syneos Health
Customization scope
If you need specific market information, which is not currently within the scope of the report, we will provide it to you as a part of the customization.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Australia & New Zealand Healthcare CROs Market Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the Australia & New Zealand healthcare CROs market report based on phase type, service, application, end-use, and country:
-
Phase Type Outlook (Revenue, USD Million, 2018 - 2030)
-
Drug Discovery
-
Target Validation
-
Lead Identification
-
Lead Optimization
-
-
Preclinical
-
Clinical
-
Phase I Trial Services
-
Phase II Trial Services
-
Phase III Trial Services
-
Phase IV Trial Services
-
-
-
Service Outlook (Revenue, USD Million, 2018-2030)
-
Project Management/Clinical Supply Management
-
Data Management
-
Regulatory/Medical Affairs
-
Medical Writing
-
Clinical Monitoring
-
Quality Management/ Assurance
-
Bio-statistics
-
Investigator Payments
-
Laboratory
-
Patient and site Recruitment
-
Technology
-
Others
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Oncology
-
Cardiovascular
-
Autoimmune/Inflammation
-
Central nervous system (CNS)
-
Dermatology
-
Infectious diseases
-
Respiratory
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2018 - 2030)
-
Pharmaceutical & Biopharmaceutical Companies
-
Medical Device Companies
-
-
Country Outlook (Revenue, USD Million, 2018 - 2030)
-
Australia
-
New Zealand
-
Frequently Asked Questions About This Report
b. The Australia & New Zealand healthcare CROs market size was estimated at USD 1.0 billion in 2022 and is expected to reach USD 1.1 billion in 2023.
b. The Australia & New Zealand healthcare CROs market is expected to grow at a compound annual growth rate of 11.5% from 2023 to 2030 to reach USD 2.4 billion by 2030.
b. On the basis of type, the clinical segment contributed for 76.14% of the Australia and New Zealand healthcare contract research organizations market in 2022. The complexity of clinical trials has led to an increase in the use of CROs. One key element causing greater complexity is the need for biopharmaceutical companies to address critical disease threats, such as oncology and orphan diseases. This has led to an increase in the demand for CROs to run clinical trials.
b. Some of the key players in the Australia & New Zealand healthcare CROs market are Thermo Fisher Scientific, Inc. (PPD), SGS SA, ICON plc, Medpace Australia Pty. Ltd, Charles River Laboratories, IQVIA, Novotech, Labcorp Drug Development, and Syneos Health.
b. Increasing adoption of advanced technologies; rising strategic initiatives such as geographical expansion, mergers & acquisitions, & R&D grants & tax incentives; and cost-effectiveness & favorable environment for clinical trials are a few of the key factors boosting the demand for contract research organizations in Australia & New Zealand.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





