- Home
- »
- Medical Devices
- »
-
Biopharmaceutical CMO and CRO Market Size Report, 2030GVR Report cover
![Biopharmaceutical CMO And CRO Market Size, Share & Trends Report]()
Biopharmaceutical CMO And CRO Market Size, Share & Trends Analysis Report By Source (Mammalian, Non-mammalian), By Service (Contract Manufacturing, Contract Research), By Product, And Segment Forecasts, 2024 - 2030
- Report ID: GVR-1-68038-500-7
- Number of Pages: 180
- Format: Electronic (PDF)
- Historical Range: 2018 - 2023
- Industry: Healthcare
Market Size & Trends
The global biopharmaceutical CMO and CRO market size was valued at USD 36.21 billion in 2023 and is anticipated to grow at a compound annual growth rate (CAGR) of 5.6% from 2024 to 2030. There was unprecedented growth in 2020 due to the COVID-19 pandemic. The rising investments in the biopharmaceutical industry by the prominent participants to enhance their productivity and efficiency have driven the bio manufacturers to increase their focus on outsourcing activities. Currently, biopharma companies have begun outsourcing the resource and capital-intensive steps and, in a few cases, the entire chain of biomanufacturing, thereby boosting the demand for contract-based services.
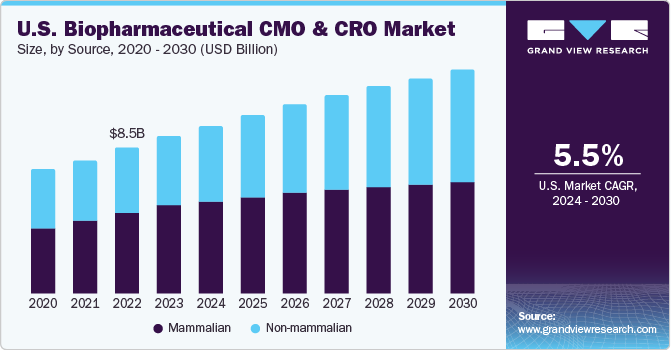
With improvement in outsourced services, biopharmaceutical industry continues to show surge in number of biopharmaceutical firms opting for contract services. CMOs are making enormous investments for expansion of their manufacturing capabilities.For instance, in March 2022, Cambrex, an U.S. based CDMO expanded its biopharmaceutical testing services business with the addition of 11 cGMP laboratories in its U.S. facility. The expansion included the addition of instruments for nanoparticle size analysis, qPCR, imaging, mass spectrometry, immunoblotting, next-generation sequencing (NGS) and for other applications.
In recent years, the biopharmaceutical industry has witnessed a significant number of consolidations. These consolidations were mainly aimed at business expansion and to stay competitive in the biopharmaceutical contract manufacturing and services market. Although the biopharmaceutical CMO & CRO industry itself is relatively developed, the inception of new bioprocessing tools, novel therapeutics, and the priority shifts in the bio/pharmaceutical industry pertaining to products has increased the pressure on the contract biomanufacturers. As a result, CDMOs are adopting different business models for addressing the needs of their clients and stakeholders in the best possible way.
Furthermore, the integration of single-use systems in production facilities helps the CMOs to economically expand the manufacturing capacity. The single-use products offer fast turnaround and limit allied activities, such as cleaning and changeover validation. However, the contract negotiations between CMOs and customers are observed to be difficult owing to the regulatory landscape and complexity of service. Clients and CMOs are facing issues pertaining to the IP rights, warranty & liabilities, prices & timelines, which increases the complexity of negotiations.
Market Characteristics
Market growth stage is medium, and pace of the market growth is accelerating. The biopharmaceutical CMO and CRO market is characterized by a high degree of innovation. Rapid advancements in bioprocessing engineering, including gradual evolution of contract services, have contributed to this massive success of the biologics industry.

The market is also characterized by a medium level of merger and acquisition (M&A) activity by leading players. Merger and acquisition activity in the CRO sector has led to rising competition among the CROs. This, in turn, has pushed the CROs to grow their capabilities to meet the demands of large molecule developers.
Biopharmaceutical CMO and CRO market is characterized by a high impact of regulations. Biopharmaceutical products are highly complex and sensitive. Ensuring these products' quality, safety, and efficacy is paramount. Regulatory frameworks establish strict guidelines for manufacturing processes, quality control, and testing to guarantee that the final products meet rigorous standards.
Biopharmaceutical CMO and CRO market is characterized by a high impact of regional expansion. Regional expansion allows CMOs and CROs to establish a presence in key markets, enabling them to better serve the diverse needs of pharmaceutical companies operating in different regions.
Source Insights
The mammalian segment held the highest market share of more than 56.92% in 2023 owing to the lack of internal expertise in the industry. This is attributed to their capabilities to add human-like post-translational modifications to complex protein therapeutics. Furthermore, the segment greatly benefited from the introduction of novel and enhanced expression systems, improved process monitoring solutions, cell line engineering tools, automated screening methods, and disposable devices.
Non-mammalian segment is projected to witness a rapid growth of 7.7% during the forecast period. Non-mammalian cell lines, such as microbial cell lines, are recognized as potent factories. Innovative strategies are being implemented to identify and explore the potential of various microbes. This, in turn, is anticipated to contribute to the non-mammalian biopharmaceutical manufacturing segment. Moreover, employment of Yeast Saccharomyces cerevisiae for production of several large volume products is aiding revenue generation for this segment.
Services Insights
Contract Manufacturing segment held a dominant share of over 58.0% in 2023. This is attributed to the increasing outsourcing of research activities by biopharmaceutical companies. A substantial number of CMOs and opportunistic CMOs in this space are engaged in providing biopharma entities with plenty of services including end-to-end coverage from cell cultivation to fill/finish services. Furthermore, clients are investing heavily to outsource the manufacturing aspect of their product development program. These factors have resulted in the largest share of this segment.
Contract Research segment is anticipated to witness the fastest growth rate of 6.4% in biopharmaceutical CMO and CRO market over a forecast period. CROs are striving to capitalize on the potential avenues in the industry. New market entrants and small-scale players, which are focused on the development of biopharmaceuticals are anticipated to opt for the contract research services for their discovery programs of new candidates, thereby boosting the segment growth.Furthermore, stringent regulations for approval of biopharmaceuticals are anticipated to drive growth of this segment.
Product Insights
Biologics segment dominated a market with largest revenue share of over 79.91% in 2023. This growth is attributed to the high specificity of biologics, complex manufacturing steps, and a higher success rate as compared to other drug molecules. The use of single-use bioreactors, continuous purification processing, disposable plastic containers, and real-time quality analysis has enabled the CMOs to meet the increasing service demand for biologics production effectively. Moreover, increased outsourcing budget, directed toward biologics development, can be attributed to the large share held by this segment. With the growing biologics market, developers and regulators are struggling to keep up. This is expected to enhance adoption of CMO services, driving the segment.
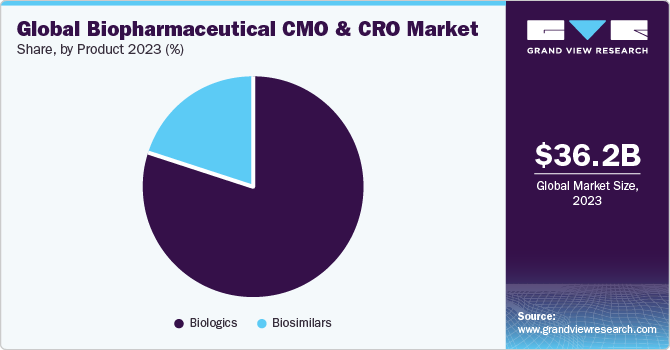
Biosimilars segment is anticipated to witness a fastest growth rate of 8.7% in biopharmaceutical CMO and CRO market over a forecast period.several companies are investing in biosimilar development to outperform the safety, efficacy, disposition, or cost of earlier in-class innovator drugs. This has increased the level of competition amongst innovator manufacturers, which in turn, is likely to benefit the CMOs.
Regional Insights
North America held a largest revenue share of 33.11% in 2023. This can be attributed to a local presence of several service providers in the region. Also, a significant number of approved products in the U.S. are being manufactured by CMOs. The presence of several small- and mid-size biopharmaceutical entities (SMEs) lack the resources and budget to establish facilities with well-equipped resources. This, in turn, has increased an inter-dependency between CMOs and SMEs in the U.S., resulting in the dominance of the U.S. market. Asia Pacific is expected to grow at a significant CAGR during a forecast period.
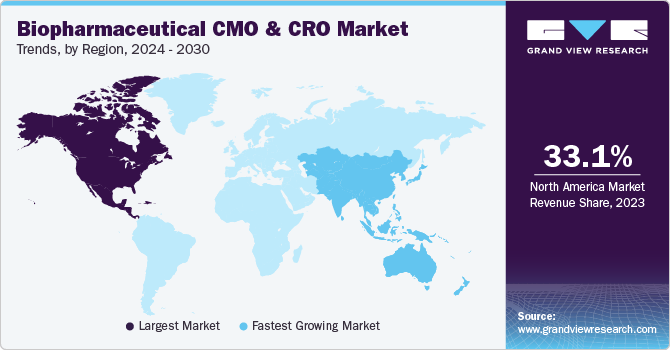
Increasing adoption and investment in advanced research and manufacturing technologies by CMOs & CROs based in the U.S. is propelling the growth of biopharmaceutical CMO & CRO Market in the country. In addition, quality services offered by service providers are driving the biopharma and CMO partnerships in the country. In addition, expansion of foreign CMOs and CROs into the country is expected to propel the market growth.
Asia Pacific biopharmaceuticals CMO & CRO Market is growing at a significant rate and the region is expected to emerge as the fastest-growing market for biopharmaceutical R&D and manufacturing. This growth can be attributed to various factors, such as regulatory changes, improving infrastructure, and presence of a large number of potential study subjects. Several biopharma companies from the U.S. are considering development of pharma products in Asian countries owing to growing cost of R&D in their home country.
China market held a notable share in 2023 due to increasing R&D activities. Increase in outsourcing of operations to Chinese market players by overseas biopharma firms is recognized as a major driver for the Chinese CMO & CRO Market. Global innovator companies outsource a significant portion of their research to restructure R&D and cut costs. Furthermore, biotechnology and its related aspects are significantly funded by the Chinese government. CRO and CMO capabilities are also strengthened by knowledge and quality management skills of a large number of people who have returned to the country after having worked in the U.S. and Europe.
Biopharmaceutical CMO & CRO Market Share Insights
The market is fragmented in nature with a substantial number of established as well as medium- to small-sized CMOs and CROs. Several market participants are privately held or are a part of private equity firms’ portfolios. CMOs are integrating automation and innovative technologies at their plants to improve their capabilities in terms of project throughput and product quality. This has driven the interest of large molecule manufacturers in the CMOs to meet the growing demand for biologics.
Charles River Laboratories International, Inc.; PRA Health Sciences; ICON plc.; Pharmaceutical Product Development, LLC; LabCorp; and Parexel International Corporation are engaged in offering contract research services for large molecule production. Market players focus on various business strategies including mergers, acquisitions, product launches, etc. For instance, in June 2022, FUJIFILM Corporation acquired cell therapy manufacturing facility from Atara Biotherapeutics, Inc. This acquisition is being made for expansion of its CMO business capabilities.
Key Biopharmaceutical CMO & CRO Companies:
The following are the leading companies in the biopharmaceutical CMO & CRO market. These companies collectively hold the largest market share and dictate industry trends. Financials, strategy maps & products of these biopharmaceutical CMO & CRO companies are analyzed to map the supply network.
- Boehringer Ingelheim GmbH
- Lonza Group AG
- Inno Biologics Sdn Bhd
- Rentschler Biopharma SE
- JRS Pharma
- Biomeva GmbH
- ProBioGen AG
- Fujifilm Diosynth Biotechnologies U.S.A., Inc.
- Toyobo Co., Ltd.
- Samsung Biologics
- Thermo Fisher Scientific Inc (Patheon & PPD)
- CMC Biologics
- WuXi Biologics
- AbbVie Inc.
- Binex Co., Ltd.
- Charles River Laboratories International, Inc.
- ICON Plc
- Parexel International Corporation
- Laboratory Corporation of America Holdings
Recent Developments
-
In June 2023, Lonza, a contract manufacturing services (CMO) in the biotechnology and pharmaceutical industries,acquired Synaffix B.V. (Synaffix), a biotechnology company dedicated to advancing its clinical-stage technology platform for Antibody-Drug Conjugate development. The acquisition is set to enhance Lonza's bioconjugates portfolio by incorporating Synaffix's proprietary technology platform and research and development capabilities.
-
In October 2023, Samsung Biologics and Kurma Partners entered in to strategic collaboration for the development and manufacturing of biologics intended for Kurma's portfolio companies.The multi-year collaboration is anticipated to foster the sustainable growth of the biopharmaceutical industry, enabling Samsung Biologics to extend its presence into the European market.
Biopharmaceutical CMO & CRO Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 38.94 billion
Revenue forecast in 2030
USD 54.12 billion
Growth rate
CAGR of 5.6% from 2024 to 2030
Historical data
2018 - 2023
Forecast period
2024 - 2030
Quantitative units
Revenue in USD Billion and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Source, service, product, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; U.K.; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; India; Japan; Australia; Thailand; South Korea; Brazil; Mexico; Argentina; Saudi Arabia; UAE; South Africa, Kuwait
Key companies profiled
Boehringer Ingelheim GmbH; Lonza Group AG; Inno Biologics Sdn Bhd; Rentschler Biopharma SE; JRS Pharma; Biomeva GmbH; ProBioGen AG; Fujifilm Diosynth Biotechnologies U.S.A., Inc.; Toyobo Co., Ltd.; Samsung Biologics; Thermo Fisher Scientific Inc, CMC Biologics, WuXi Biologics, AbbVie Inc., Binex Co., Ltd., Charles River Laboratories International, Inc., ICON plc, Parexel International Corporation; Laboratory Corporation of America Holdings
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Biopharmaceutical CMO & CRO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For the purpose of this study, Grand View Research has segmented the global biopharmaceutical CMO & CRO Market report on the basis of source, service, product, and region:
-
Source Outlook (Revenue, USD Billion, 2018 - 2030)
-
Mammalian
-
Non-mammalian
-
-
Service Type Outlook (Revenue, USD Billion, 2018 - 2030)
-
Contract Manufacturing
-
Process Development
-
Downstream
-
Upstream
-
-
Fill & Finish Operations
-
Analytical & QC Studies
-
Packaging
-
-
Contract Research
-
Oncology
-
Inflammation & Immunology
-
Cardiology
-
Neuroscience
-
Others
-
-
-
Product Outlook (Revenue, USD Billion, 2018 - 2030)
-
Biologics
-
Monoclonal antibodies (MAbs)
-
Recombinant Proteins
-
Vaccines
-
Antisense, RNAi, & Molecular Therapy
-
Others
-
-
Biosimilars
-
-
Regional Outlook (Revenue, USD Billion, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
U.K.
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Norway
-
Denmark
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global biopharmaceutical CMO and CRO market size was estimated at USD 32.4 billion in 2022 and is expected to reach USD 36.2 billion in 2023.
b. The global biopharmaceutical CMO and CRO market is expected to grow at a compound annual growth rate of 5.9% from 2023 to 2030 to reach USD 54.1 billion by 2030.
b. North America dominated the biopharmaceutical CMO & CRO market with a share of 33.9% in 2022. This is attributable to the widespread manufacturing of biopharmaceuticals in the region and the presence of a substantial number of biopharmaceutical facilities in the region.
b. Some key players operating in the biopharmaceutical CMO & CRO market include Lonza; Rentschler Biotechnologie GmbH; Boehringer Ingelheim GmbH; JRS Pharma (Celonic GmbH); Inno Biologics Sdn Bhd; ProBioGen; BIOMEVA GmbH; FUJIFILM Diosynth Biotechnologies U.S.A., Inc.; Samsung BioLogics; and TOYOBO CO., LTD.; Patheon, CMC Biologics; WuXi Biologics; AbbVie Inc.; Binex Co., Ltd.; Charles River Laboratories International, Inc.; PRA Health Sciences; Parexel International Corporation.
b. Key factors that are driving the biopharmaceutical CMO and CRO market growth include the rise in investment by CMOs for capacity expansion, the commercial success of biopharmaceuticals, and the consequent increase in demand for biopharmaceuticals, and robust biopharmaceuticals pipelines.
Table of content
Chapter 1 Biopharmaceutical CMO And CRO Market: Research Methodology
1.1 Market Segmentation & Scope
1.2 Market Definition
1.3 Information Procurement
1.3.1 Purchased database
1.3.2 GVR’s internal database
1.3.3 Secondary sources & third party perspectives
1.3.4 Primary research
1.4 Information Analysis
1.4.1 Data analysis models
1.5 Market Formulation & Data Visualization
1.6 Data Validation & Publishing
Chapter 2 Biopharmaceutical CMO And CRO Market: Executive Summary
2.1 Biopharmaceutical CMO & CRO Market Outlook, 2018 - 2030
Chapter 3 Biopharmaceutical CMO And CRO Market: Variables, Trends, & Scope
3.1 Market Liniage
3.1.1 Parent Market
3.1.2 Ancialiary Market
3.2 Biopharmaceutical CMO & CRO Market: Market Dynamics
3.2.1 Market driver analysis
3.2.1.1 Rising investment by CMOs for capacity expansion
3.2.1.2 Commercial success of biopharmaceuticals and consequent increased demand for the biopharmaceuticals
3.2.1.3 Robust biopharmaceuticals pipeline
3.2.1.4 Cost and time saving benefits offered by contract services
3.2.2 Market restraint analysis
3.2.2.1 Limited outsourcing amongst well-stablished biopharmaceutical manufacturer
3.4 Key Opportunity Analysis
3.4.1 Rising funds in biopharmaceutical indutry
3.4.2 Mergers & acquistion for facility expansion
3.4.3 Growing demand for protein therapeutics
3.5 Industry Analysis - Porter’s
3.5.1 Supplier Power: Substantial number of suppliers for bioprocessing equipment contributes to the low supplier power
3.5.2 Buyer Power: Fragmented nature of biopharmaceutical industry has led to moderate bargaining power of buyers
3.5.3 Substitution Threat: High due to presence of several big pharma companies
3.5.4 New Entrants Threat: Presence of established players coupled with need of adequate capacity results in moderate threat of new entrants
3.5.5 Competitive Rivalry: High due to fragmented nature of market
3.6 Biopharmaceuticals CMO & CRO PEST Analysis
3.6.1 Political landscape
3.6.2 Economic landscape
3.6.3 Social landscape
3.6.4 Technology landscape
3.7 COVID-19 Impact and Reformation Analysis
Chapter 4. Biopharmaceuticals CMO & CRO Market: Source Estimates & Trend Analysis
4.1. Segment Dashboard
4.2. Biopharmaceuticals CMO & CRO : Source Movement Analysis, 2023 & 2030 (USD Billion)
4.3. Mammalian Source
4.3.1. Mammalian Source Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Billion)
4.4. Non-Mammalian Source
4.4.1. Non-Mammalian Source Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Billion)
Chapter 5. Biopharmaceuticals CMO & CRO Market: Service Type Estimates & Trend Analysis
5.1. Segment Dashboard
5.2. Biopharmaceuticals CMO & CRO Market: Service Type Movement Analysis, 2023 & 2030 (USD Billion)
5.3. Contract Manufacturing
5.3.1. Contract Manufacturing Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Billion)
5.3.2. Process Development
5.3.2.1. Process Development Market, 2018 - 2030 (USD Billion)
5.3.2.2. Downstream Processing
5.3.2.2.1. Downstream Processing Market, 2018 - 2030 (USD Billion)
5.3.2.3. Upstream Processing
5.3.2.3.1. Upstream Processing Market, 2018 - 2030 (USD Billion)
5.3.3. Fill & Finish Operations
5.3.3.1. Fill & Finish Operations Market, 2018 - 2030 (USD Billion)
5.3.4. Analytical & Qc Testing
5.3.4.1. Analytical & Qc Testing Market, 2018 - 2030 (USD Billion)
5.3.5. Packaging
5.3.5.1. Packaging Market, 2018 - 2030 (USD Billion)
5.4. Contract Research
5.4.1. Contract Research Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Billion)
5.4.2. Oncology
5.4.2.1. Oncology Market, 2018 - 2030 (USD Billion)
5.4.3. Cardiology
5.4.3.1. Cardiology Market, 2018 - 2030 (USD Billion)
5.4.4. Neuroscience
5.4.4.1. Neuroscience Market, 2018 - 2030 (USD Billion)
5.4.5. Others
5.4.5.1.1. Others Market, 2018 - 2030 (USD Billion)
Chapter 6. Biopharmaceuticals CMO & CRO Market: Product Estimates & Trend Analysis
6.1. Segment Dashboard
6.2. Biopharmaceuticals CMO & CRO Market: Product Movement Analysis, 2023 & 2030 (USD Billion)
6.3. Biologics
6.3.1. Biologics Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Billion)
6.3.2. Monoclonal Antibodies
6.3.2.1. Monoclonal Antibodies Market, 2018 - 2030 (USD Billion)
6.3.3. Recombinant Proteins
6.3.3.1. Recombinant Proteins Market, 2018 - 2030 (USD Billion)
6.3.4. Vaccines
6.3.4.1. Vaccines Market, 2018 - 2030 (USD Billion)
6.3.5. Antisense, RNAi, & Molecular Therapy
6.3.5.1. Antisense, Rnai, & Molecular Therapy Market, 2018 - 2030 (USD Billion)
6.3.5.2. Others
6.3.6. Others Market, 2018 - 2030 (USD Billion)
6.4. Biosimilars
6.4.1. Biosimilars Market Revenue Estimates and Forecasts, 2018 - 2030 (USD Billion)
Chapter 7. Biopharmaceuticals CMO & CRO Market: Regional Estimates & Trend Analysis
7.1. Biopharmaceuticals CMO & CRO Market Share, By Region, 2023 & 2030, USD Billion
7.2. North America
7.2.1. North America Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.2.2. U.S.
7.2.2.1. Key Country Dynamics
7.2.2.2. Regulatory Landscape/Reimbursement Scenario
7.2.2.3. Competitive Insights
7.2.2.4. U.S. Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.2.3. Canada
7.2.3.1. Key Country Dynamics
7.2.3.2. Regulatory Landscape/Reimbursement Scenario
7.2.3.3. Competitive Insights
7.2.3.4. Canada Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3. Europe
7.3.1. Europe Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.2. U.K.
7.3.2.1. Key Country Dynamics
7.3.2.2. Regulatory Landscape/Reimbursement Scenario
7.3.2.3. Competitive Insights
7.3.2.4. U.K. Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.3. Germany
7.3.3.1. Key Country Dynamics
7.3.3.2. Regulatory Landscape/Reimbursement Scenario
7.3.3.3. Competitive Insights
7.3.3.4. Germany Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.4. France
7.3.4.1. Key Country Dynamics
7.3.4.2. Regulatory Landscape/Reimbursement Scenario
7.3.4.3. Competitive Insights
7.3.4.4. France Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.5. Italy
7.3.5.1. Key Country Dynamics
7.3.5.2. Regulatory Landscape/Reimbursement Scenario
7.3.5.3. Competitive Insights
7.3.5.4. Italy Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.6. Spain
7.3.6.1. Key Country Dynamics
7.3.6.2. Regulatory Landscape/Reimbursement Scenario
7.3.6.3. Competitive Insights
7.3.6.4. Spain Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.7. Sweden
7.3.7.1. Key Country Dynamics
7.3.7.2. Regulatory Landscape/Reimbursement Scenario
7.3.7.3. Competitive Insights
7.3.7.4. Sweden Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.8. Denmark
7.3.8.1. Key Country Dynamics
7.3.8.2. Regulatory Landscape/Reimbursement Scenario
7.3.8.3. Competitive Insights
7.3.8.4. Denmark Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.9. Norway
7.3.9.1. Key Country Dynamics
7.3.9.2. Regulatory Landscape/Reimbursement Scenario
7.3.9.3. Competitive Insights
7.3.9.4. Norway Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.3.10. Netherland
7.3.10.1. Key Country Dynamics
7.3.10.2. Regulatory Landscape/Reimbursement Scenario
7.3.10.3. Competitive Insights
7.3.10.4. Netherland Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.4. Asia Pacific
7.4.1. Asia Pacific Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.4.2. China
7.4.2.1. Key Country Dynamics
7.4.2.2. Regulatory Landscape/Reimbursement Scenario
7.4.2.3. Competitive Insights
7.4.2.4. China Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.4.3. Japan
7.4.3.1. Key Country Dynamics
7.4.3.2. Regulatory Landscape/Reimbursement Scenario
7.4.3.3. Competitive Insights
7.4.3.4. Japan Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.4.4. India
7.4.4.1. Key Country Dynamics
7.4.4.2. Regulatory Landscape/Reimbursement Scenario
7.4.4.3. Competitive Insights
7.4.4.4. India Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.4.5. South Korea
7.4.5.1. Key Country Dynamics
7.4.5.2. Regulatory Landscape/Reimbursement Scenario
7.4.5.3. Competitive Insights
7.4.5.4. South Korea Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.4.6. Australia
7.4.6.1. Key Country Dynamics
7.4.6.2. Regulatory Landscape/Reimbursement Scenario
7.4.6.3. Competitive Insights
7.4.6.4. Australia Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.4.7. Thailand
7.4.7.1. Key Country Dynamics
7.4.7.2. Regulatory Landscape/Reimbursement Scenario
7.4.7.3. Competitive Insights
7.4.7.4. Thailand Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.5. Latin America
7.5.1. Latin America Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.5.2. Brazil
7.5.2.1. Key Country Dynamics
7.5.2.2. Regulatory Landscape/Reimbursement Scenario
7.5.2.3. Competitive Insights
7.5.2.4. Brazil Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.5.3. Mexico
7.5.3.1. Key Country Dynamics
7.5.3.2. Regulatory Landscape/Reimbursement Scenario
7.5.3.3. Competitive Insights
7.5.3.4. Mexico Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.5.4. Argentina
7.5.4.1. Key Country Dynamics
7.5.4.2. Regulatory Landscape/Reimbursement Scenario
7.5.4.3. Competitive Insights
7.5.4.4. Argentina Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.6. Middle East and Africa
7.6.1. Middle East and Africa Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.6.2. Saudi Arabia
7.6.2.1. Key Country Dynamics
7.6.2.2. Regulatory Landscape/Reimbursement Scenario
7.6.2.3. Competitive Insights
7.6.2.4. Saudi Arabia Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.6.3. UAE
7.6.3.1. Key Country Dynamics
7.6.3.2. Regulatory Landscape/Reimbursement Scenario
7.6.3.3. Competitive Insights
7.6.3.4. UAE Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.6.4. South Africa
7.6.4.1. Key Country Dynamics
7.6.4.2. Regulatory Landscape/Reimbursement Scenario
7.6.4.3. Competitive Insights
7.6.4.4. South Africa Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
7.6.5. Kuwait
7.6.5.1. Key Country Dynamics
7.6.5.2. Regulatory Landscape/Reimbursement Scenario
7.6.5.3. Competitive Insights
7.6.5.4. Kuwait Biopharmaceuticals CMO & CRO Market Estimates and Forecasts, 2018 - 2030 (USD Billion)
Chapter 8. Competitive Landscape
8.1. Recent Developments & Impact Analysis by Key Market Participants
8.2. Company Categorization
8.3. Company Market Share Analysis
8.4. Company Heat Map Analysis
8.5. Strategy Mapping
8.5.1. Expansion
8.5.2. Mergers & Acquisition
8.5.3. Partnerships & Collaborations
8.5.4. New Product Launches
8.5.5. Research And Development
8.6. Company Profiles
8.6.1. Boehringer Ingelheim GmbH
8.6.1.1. Participant’s Overview
8.6.1.2. Financial Performance
8.6.1.3. Product Benchmarking
8.6.1.4. Recent Developments
8.6.2. LONZA
8.6.2.1. Participant’s Overview
8.6.2.2. Financial Performance
8.6.2.3. Product Benchmarking
8.6.2.4. Recent Developments
8.6.3. Inno Biologics Sdn Bhd
8.6.3.1. Participant’s Overview
8.6.3.2. Financial Performance
8.6.3.3. Product Benchmarking
8.6.3.4. Recent Developments
8.6.4. Rentschler Biotechnologie GmbH
8.6.4.1. Participant’s Overview
8.6.4.2. Financial Performance
8.6.4.3. Product Benchmarking
8.6.4.4. Recent Developments
8.6.5. JRS PHARMA
8.6.5.1. Participant’s Overview
8.6.5.2. Financial Performance
8.6.5.3. Product Benchmarking
8.6.5.4. Recent Developments
8.6.6. BIOMEVA GmbH
8.6.6.1. Participant’s Overview
8.6.6.2. Financial Performance
8.6.6.3. Product Benchmarking
8.6.6.4. Recent Developments
8.6.7. ProBioGen
8.6.7.1. Participant’s Overview
8.6.7.2. Financial Performance
8.6.7.3. Product Benchmarking
8.6.7.4. Recent Developments
8.6.8. FUJIFILM Diosynth Biotechnologies U.S.A., Inc.
8.6.8.1. Participant’s Overview
8.6.8.2. Financial Performance
8.6.8.3. Product Benchmarking
8.6.8.4. Recent Developments
8.6.9. TOYOBO CO., LTD.
8.6.9.1. Participant’s Overview
8.6.9.2. Financial Performance
8.6.9.3. Product Benchmarking
8.6.9.4. Recent Developments
8.6.10. Thermo Fisher Scientific Inc
8.6.10.1. Participant’s Overview
8.6.10.2. Financial Performance
8.6.10.3. Product Benchmarking
8.6.10.4. Recent Developments
8.6.11. CMC Biologics
8.6.11.1. Participant’s Overview
8.6.11.2. Financial Performance
8.6.11.3. Product Benchmarking
8.6.11.4. Recent Developments
8.6.12. Binex Co., Ltd.
8.6.12.1. Participant’s Overview
8.6.12.2. Financial Performance
8.6.12.3. Product Benchmarking
8.6.12.4. Recent Developments
8.6.13. WuXi Biologics
8.6.13.1. Participant’s Overview
8.6.13.2. Financial Performance
8.6.13.3. Product Benchmarking
8.6.13.4. Recent Developments
8.6.14. AbbVie, Inc.
8.6.14.1. Participant’s Overview
8.6.14.2. Financial Performance
8.6.14.3. Product Benchmarking
8.6.14.4. Recent Developments
8.6.15. Charles River Laboratories International, Inc
8.6.15.1. Participant’s Overview
8.6.15.2. Financial Performance
8.6.15.3. Product Benchmarking
8.6.15.4. Recent Developments
8.6.16. ICON plc.
8.6.16.1. Participant’s Overview
8.6.16.2. Financial Performance
8.6.16.3. Product Benchmarking
8.6.16.4. Recent Developments
8.6.17. Laboratory Corporation of America Holdings
8.6.17.1. Participant’s Overview
8.6.17.2. Financial Performance
8.6.17.3. Product Benchmarking
8.6.17.4. Recent Developments
8.6.18. Parexel International Corporation
8.6.18.1. Participant’s Overview
8.6.18.2. Financial Performance
8.6.18.3. Product Benchmarking
8.6.18.4. Recent Developments
List of Tables
Table 1 List of secondary sources
Table 2 List of abbreviations
Table 3 Global biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 4 Global biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 5 Global biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 6 Global biopharmaceutical CMO & CRO market, by region, 2018 - 2030 (USD Billion)
Table 7 North America biopharmaceutical CMO & CRO market, by country, 2018 - 2030 (USD Billion)
Table 8 North America biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 9 North America biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 10 North America biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 11 U.S. biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 12 U.S. biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 13 U.S. biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 14 Canada biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 15 Canada biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 16 Canada biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 17 Europe biopharmaceutical CMO & CRO market, by country, 2018 - 2030 (USD Billion)
Table 18 Europe biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 19 Europe biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 20 Europe biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 21 Germany biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 22 Germany biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 23 Germany biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 24 UK biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 25 UK biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 26 UK biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 27 France biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 28 France biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 29 France biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 30 Italy biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 31 Italy biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 32 Italy biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 33 Spain biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 34 Spain biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 35 Spain biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 36 Denmark biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 37 Denmark biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 38 Denmark biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 39 Sweden biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 40 Sweden biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 41 Sweden biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 42 Norway biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 43 Norway biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 44 Norway biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 45 Asia Pacific biopharmaceutical CMO & CRO market, by country, 2018 - 2030 (USD Billion)
Table 46 Asia Pacific biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 47 Asia Pacific biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 48 Asia Pacific biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 49 Japan biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 50 Japan biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 51 Japan biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 52 China biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 53 China biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 54 China biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 55 India biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 56 India biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 57 India biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 58 Australia biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 59 Australia biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 60 Australia biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 61 South Korea biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 62 South Korea biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 63 South Korea biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 64 Thailand biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 65 Thailand biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 66 Thailand biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 67 Latin America biopharmaceutical CMO & CRO market, by country, 2018 - 2030 (USD Billion)
Table 68 Latin America biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 69 Latin America biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 70 Latin America biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 71 Brazil biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 72 Brazil biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 73 Brazil biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 74 Mexico biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 75 Mexico biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 76 Mexico biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 77 Argentina biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 78 Argentina biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 79 Argentina biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 80 Colombia biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 81 Colombia biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 82 Colombia biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 83 Middle East & Africa biopharmaceutical CMO & CRO market, by country, 2018 - 2030 (USD Billion)
Table 84 Middle East & Africa biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 85 Middle East & Africa biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 86 Middle East & Africa biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 87 South Africa biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 88 South Africa biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 89 South Africa biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 90 Saudi Arabia biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 91 Saudi Arabia biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 92 Saudi Arabia biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 93 UAE biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 94 UAE biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 95 UAE biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
Table 96 Kuwait biopharmaceutical CMO & CRO market, by source, 2018 - 2030 (USD Billion)
Table 97 Kuwait biopharmaceutical CMO & CRO market, by service type, 2018 - 2030 (USD Billion)
Table 98 Kuwait biopharmaceutical CMO & CRO market, by product, 2018 - 2030 (USD Billion)
List of Figures
Fig. 1 Market research process
Fig. 2 Information Procurement
Fig. 3 Primary research pattern
Fig. 4 Market research approaches
Fig. 5 Value chain based sizing & forecasting
Fig. 6 QFD modelling for market share assessment
Fig. 7 Market segmentation & scope
Fig. 8 Biopharmaceutical CMO & CRO market outlook, 2018 - 2030
Fig. 9 Market trends & outlook
Fig. 10 Market driver relevance analysis (Current & future impact)
Fig. 11 Cumulative success rate comparison of biologics with small molecules over time
Fig. 12 Number of biologics investigational new drugs, 2005-2021
Fig. 13 Number of Reference Products by U.S. Biosimilars Launchable Dates
Fig. 14 Market restraint relevance analysis (Current & future impact)
Fig. 15 Risk associated with outsourcing services
Fig. 16 PEST Analysis (political & legal, economic and technological)
Fig. 17 Porter’s Five Forces Analysis
Fig. 18 Company market share analysis
Fig. 19 Strategy framework
Fig. 20 Participant categorization
Fig. 21 CMO selection process
Fig. 22 Trends in the biopharmaceutical industry
Fig. 23 R&D process of biopharmaceutical development
Fig. 24 Development phases where outsourcing partners are engaged
Fig. 25 Global biopharmaceuticals CMO & CRO market: Source outlook key takeaways
Fig. 26 Global Biopharmaceuticals CMO & CRO market: Source movement analysis
Fig. 27 Global mammalian source market, 2018 - 2030 (USD Billion)
Fig. 28 Global mammalian source market, 2018 - 2030 (USD Billion)
Fig. 29 Global biopharmaceuticals CMO & CRO market: Service type outlook key takeaways
Fig. 30 Global biopharmaceuticals CMO & CRO market: Service movement analysis
Fig. 31 Global contract manufacturing market, 2018 - 2030 (USD Billion)
Fig. 32 Percentage of biomanufacturers outsourcing some activity of upstream and downstream processing, 2011-2016
Fig. 33 Global process development market, 2018 - 2030 (USD Billion)
Fig. 34 Global downstream processing market, 2018 - 2030 (USD Billion)
Fig. 35 Global upstream processing market, 2018 - 2030 (USD Billion)
Fig. 36 Global fill & finish operations market, 2018 - 2030 (USD Billion)
Fig. 37 Global analytical & QC testing market, 2018 - 2030 (USD Billion)
Fig. 38 Global contract packaging market, 2018 - 2030 (USD Billion)
Fig. 39 Breakdown of the general CRO industry by type of service
Fig. 40 Initial attributes that play key role into CRO engagement
Fig. 41 Post-engagement attributes that play key role into CRO engagement
Fig. 42 Global contract research market, 2018 - 2030 (USD Billion)
Fig. 43 Global oncology market, 2018 - 2030 (USD Billion)
Fig. 44 Global inflammation & immunology market, 2018 - 2030 (USD Billion)
Fig. 45 Global cardiology market, 2018 - 2030 (USD Billion)
Fig. 46 Global neuroscience market, 2018 - 2030 (USD Billion)
Fig. 47 Global other CRO services market, 2018 - 2030 (USD Billion)
Fig. 48 Global biopharmaceuticals CMO & CRO market: Product outlook key takeaways
Fig. 49 Global biopharmaceuticals CMO & CRO market: Product movement analysis
Fig. 50 Global biologics market, 2018 - 2030 (USD Billion)
Fig. 51 Year wise annual approvals for MAbs products which are currently marketed in U.S./EU
Fig. 52 Share of MAbs-related R&D programs at different pipeline phases, 2015
Fig. 53 Global monoclonal antibodies market, 2018 - 2030 (USD Billion)
Fig. 54 Comparison of R&D biotech pipeline expansion for recombinant products
Fig. 55 Global recombinant proteins market, 2018 - 2030 (USD Billion)
Fig. 56 Global vaccines market, 2018 - 2030 (USD Billion)
Fig. 57 Global antisense, RNAi, &molecular therapy market, 2018 - 2030 (USD Billion)
Fig. 58 Global other product market, 2018 - 2030 (USD Billion)
Fig. 59 Global biosimilars market, 2018 - 2030 (USD Billion)
Fig. 60 Regional market place: Key takeaway
Fig. 61 Comparison of biopharmaceutical manufacturing facilities region and country wise
Fig. 62 Biopharmaceuticals CMO & CRO regional outlook, 2020 & 2028
Fig. 63 North America biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 64 Number of U.S. pharmaceutical outsourcing M&A transactions
Fig. 65 Biopharmaceuticals goods export growth comparison between 2011 and 2014
Fig. 66 U.S. biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 67 Pros and cons of Canadian pharmaceutical & biopharmaceutical industry
Fig. 68 Canada biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 69 Europe biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 70 Revenue growth of Germany pharmaceutical industry
Fig. 71 UK biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 72 Germany biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 73 France biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 74 Italy biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 75 Spain biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 76 Norway biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 77 Denmark biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 78 Sweden biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 77 Factors supporting contract manufacturing/research in Asian countries
Fig. 78 Asia Pacific biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 79 China biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 80 India biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 81 Japan biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 82 South Korea biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 83 Thailand biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 84 Australia biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 85 Latin America biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 86 Brazil biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 87 Mexico biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 88 Colombia biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 89 Argentina biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 90 Middle East & Africa biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 91 South Africa biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 92 Saudi Arabia biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 93 UAE biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)
Fig. 94 Kuwait biopharmaceuticals CMO & CRO market, 2018 - 2030 (USD Billion)What questions do you have? Get quick response from our industry experts. Request a Free ConsultationMarket Segmentation
- Biopharmaceutical CMO & CRO Source Outlook (Revenue, USD Billion, 2018 - 2030)
- Mammalian
- Non-mammalian
- Biopharmaceutical CMO & CRO Service Outlook (Revenue, USD Billion, 2018 - 2030)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Biopharmaceutical CMO & CRO Product Outlook (Revenue, USD Billion, 2018 - 2030)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Biopharmaceutical CMO & CRO Market: Regional Outlook (Revenue, USD Billion, 2018- 2030)
- North America
- North America Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- North America Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- North America Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- U.S.
- U.S. Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- U.S. Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- U.S. Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- U.S. Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Canada
- Canada Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Canada Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Canada Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Canada Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- North America Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Europe
- Europe Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Europe Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Europe Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- U.K.
- U.K.Biopharm aceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- U.K. Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- U.K. Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- U.K.Biopharm aceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Germany
- Germany Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Germany Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Germany Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Germany Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- France
- France Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- France Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- France Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- France Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Italy
- Italy Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Italy Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Italy Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Italy Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Spain
- Spain Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Spain Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Spain Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Spain Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Sweden
- Sweden Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Sweden Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Sweden Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Sweden Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Norway
- Norway Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Norway Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Norway Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Norway Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Denmark
- Denmark Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Denmark Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Denmark Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Denmark Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Europe Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Asia Pacific
- Asia Pacific Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Asia Pacific Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Asia Pacific Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Japan
- Japan Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Japan Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Japan Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Japan Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- China
- China Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- China Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- China Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- China Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- India
- India Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- India Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- India Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- India Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- South Korea
- South Korea Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- South Korea Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- South Korea Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- South Korea Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Australia
- Australia Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Australia Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Australia Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Australia Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Thailand
- Thailand Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Thailand Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Thailand Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Thailand Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Asia Pacific Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Latin America
- Latin America Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Latin America Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Latin America Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Brazil
- Brazil Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Brazil Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Brazil Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Brazil Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mexico
- Mexico Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Mexico Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Mexico Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Mexico Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Argentina
- Argentina Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Argentina Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Argentina Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Argentina Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Latin America Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Middle East & Africa
- Middle East & Africa Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Middle East & Africa Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Middle East & Africa Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- South Africa
- South Africa Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- South Africa Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- South Africa Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- South Africa Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Saudi Arabia
- Saudi Arabia Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Saudi Arabia Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Saudi Arabia Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Saudi Arabia Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- UAE
- UAE Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- UAE Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- UAE Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- UAE Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Kuwait
- Kuwait Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Mammalian
- Non-mammalian
- Kuwait Biopharmaceutical CMO & CRO Market, By Service, 2018- 2030 (USD Billion)
- Contract Manufacturing
- Process Development
- Downstream
- Upstream
- Fill & Finish Operations
- Analytical & QC studies
- Packaging
- Process Development
- Contract Research
- Oncology
- Inflammation & Immunology
- Cardiology
- Neuroscience
- Others
- Contract Manufacturing
- Kuwait Biopharmaceutical CMO & CRO Market, By Products, 2018- 2030 (USD Billion)
- Biologics
- Monoclonal antibodies (MAbs)
- Recombinant Proteins
- Vaccines
- Antisense, RNAi, & Molecular Therapy
- Others
- Biosimilars
- Biologics
- Kuwait Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- Middle East & Africa Biopharmaceutical CMO & CRO Market, By Source, 2018- 2030 (USD Billion)
- North America
Biopharmaceutical CMO And CRO Market Dynamics
Driver: Rising investments for capacity expansion by CMOs
With improvements in outsourcing services, the biopharmaceutical industry continues to show surge in number of biopharmaceutical firms opting for contract services. CMOs are making enormous investments for expanding their manufacturing capabilities. For instance, in March 2022, Cambrex, a U.S.-based CDMO, expanded its biopharmaceutical testing services business with the addition of 11 cGMP laboratories in its U.S. facility. This expansion included the addition of instruments for nanoparticle size analysis, qPCR, imaging, immunoblotting, mass spectrometry, next-generation sequencing, and other applications. In another development, in February 2022, Piramal Pharma, an Indian CDMO announced an investment of USD 74.4 million to expand its active pharmaceutical ingredient and antibody-drug conjugate (ADC) manufacturing facilities in the UK. Smaller companies are also expanding their manufacturing facilities. For instance, in April 2019, Lithuania-based Biotechpharma UAB expanded its facility in the country with the addition of a new line of bioreactors with 5,000 liters capacity. Many entities are also observed to add capacity through both internal expansions and acquisitions to address the rising demand for such services. For instance, in February 2022, Recipharm acquired Arranta Bio, a provider of advanced therapy medicinal products, to expand its biologics offerings in the U.S. Such actions by market players are expected to promote market growth.
Driver: Commercial success and consequent strong demand for biopharmaceuticals
Biopharmaceuticals manufacturing is recognized as a leading sector in terms of growth potential. Many healthcare entities believe that this sector would be one of the top 10 revenue generators in the coming years. Moreover, market players are making continuous efforts for developing this sector to meet the increasing demand for bio-based pharmaceuticals, which is expected to boost the number of CMOs/CROs for biopharmaceutical production. This growth of biopharmaceuticals has resulted in the development of a large product pipeline. By understanding the cost-effectiveness due to contract manufacturing, many firms have begun to outsource parts of their biopharmaceutical manufacturing to contract manufacturers. In 2019, the FDA approved 36 biopharmaceutical products, including recombinant proteins, monoclonal antibodies, and others. According to the International Federation of Pharmaceutical Manufacturers & Associations, biopharmaceutical R&D spending generated USD 179 million in 2018 and this number is expected to grow to USD 213 million by 2025, owing to the rising demand for biopharmaceuticals. The number of pipeline drugs have significantly increased in recent years. As per Pharma Intelligence, in 2018, 15,267 drugs were in the pipeline and in 2021, 18,582 drugs were in the pipeline. This is expected to drive the growth of the market. The biologics industry has grown substantially in recent years to include novel product types including rDNA, nanobodies, synthetic vaccines, soluble receptors, fusion proteins, immunoconjugates, and immunotherapeutics. Because of revolutionization of disease therapy through biologics, this sector provides many opportunities to explore hybrid structures that combine biologics with well-defined chemical entities.
Restraint: Limited outsourcing among major biopharmaceutical manufacturers
Presence of well-established international players in the market, which are capable of performing in-house production, is expected to hamper growth of the market to a certain extent. Several big pharma clients are involved in conducting in-house R&D and clinical drug testing through a well-established infrastructure. Large firms consider outsourcing perilous due to loss of strategic control and limited management oversight. Owing to this, large pharma companies opt to maintain their manufacturing operations in-house. Furthermore, firms are using their in-house capabilities as a strategy to maintain secrecy of their product development processes. Owing to these factors, several biopharmaceutical entities have chosen to maintain in-house biologics development. For instance, in March 2022, IASO Biotherapeutics, a biopharmaceutical company involved in the manufacturing of drugs, stated that it had received a drug manufacturing license by the Medical Products Administration of Province Jiangsu to produce CAR-T Cell Therapy Products. Big pharma companies are investing significant amounts in their in-house manufacturing facilities, which is, consequently, anticipated to impede market growth.
What Does This Report Include?
This section will provide insights into the contents included in this biopharmaceutical CMO and CRO market report and help gain clarity on the structure of the report to assist readers in navigating smoothly.
Biopharmaceutical CMO And CRO market qualitative analysis
-
Industry overview
-
Industry trends
-
Market drivers and restraints
-
Market size
-
Growth prospects
-
Porter’s analysis
-
PESTEL analysis
-
Key market opportunities prioritized
-
Competitive landscape
-
Company overview
-
Financial performance
-
Product benchmarking
-
Latest strategic developments
-
Biopharmaceutical CMO And CRO market quantitative analysis
-
Market size, estimates, and forecast from 2018 to 2030
-
Market estimates and forecast for product segments up to 2030
-
Regional market size and forecast for product segments up to 2030
-
Market estimates and forecast for application segments up to 2030
-
Regional market size and forecast for application segments up to 2030
-
Company financial performance
What questions do you have? Get quick response from our industry experts. Request a Free ConsultationResearch Methodology
A three-pronged approach was followed for deducing the biopharmaceutical CMO and CRO market estimates and forecasts. The process has three steps: information procurement, analysis, and validation. The whole process is cyclical, and steps repeat until the estimates are validated. The three steps are explained in detail below:
Information procurement: Information procurement is one of the most extensive and important stages in our research process, and quality data is critical for accurate analysis. We followed a multi-channel data collection process for biopharmaceutical CMO and CRO market to gather the most reliable and current information possible.
- We buy access to paid databases such as Hoover’s and Factiva for company financials, industry information, white papers, industry journals, SME journals, and more.
- We tap into Grand View’s proprietary database of data points and insights from active and archived monitoring and reporting.
- We conduct primary research with industry experts through questionnaires and one-on-one phone interviews.
- We pull from reliable secondary sources such as white papers and government statistics, published by organizations like WHO, NGOs, World Bank, etc., Key Opinion Leaders (KoL) publications, company filings, investor documents, and more.
- We purchase and review investor analyst reports, broker reports, academic commentary, government quotes, and wealth management publications for insightful third-party perspectives.
Analysis: We mine the data collected to establish baselines for forecasting, identify trends and opportunities, gain insight into consumer demographics and drivers, and so much more. We utilized different methods of biopharmaceutical CMO and CRO market data depending on the type of information we’re trying to uncover in our research.
-
Market Research Efforts: Bottom-up Approach for estimating and forecasting demand size and opportunity, top-down Approach for new product forecasting and penetration, and combined approach of both Bottom-up and Top-down for full coverage analysis.
-
Value-Chain-Based Sizing & Forecasting: Supply-side estimates for understanding potential revenue through competitive benchmarking, forecasting, and penetration modeling.
-
Demand-side estimates for identifying parent and ancillary markets, segment modeling, and heuristic forecasting.
-
Qualitative Functional Deployment (QFD) Modelling for market share assessment.
Market formulation and validation: We mine the data collected to establish baselines for forecasting, identify trends and opportunities, gain insight into consumer demographics and drivers, and so much more. We utilize different methods of data analysis depending on the type of information we’re trying to uncover in our research.
-
Market Formulation: This step involves the finalization of market numbers. This step on an internal level is designed to manage outputs from the Data Analysis step.
-
Data Normalization: The final market estimates and forecasts are then aligned and sent to industry experts, in-panel quality control managers for validation.
-
This step also entails the finalization of the report scope and data representation pattern.
-
Validation: The process entails multiple levels of validation. All these steps run in parallel, and the study is forwarded for publishing only if all three levels render validated results.
Biopharmaceutical CMO And CRO Market Categorization:
The biopharmaceutical CMO and CRO market was categorized into four segments, namely source (Mammalian, Non-mammalian), service tpye (Contract Manufacturing, Contract Research), product (Biologics, Biosimilars), and regions (North America, Europe, Asia Pacific, Latin America, Middle East & Africa).
Segment Market Methodology:
The biopharmaceutical CMO and CRO market was segmented into source, service, product, and region. The demand at a segment level was deduced using a funnel method. Concepts like the TAM, SAM, SOM, etc., were put into practice to understand the demand. We at GVR deploy three methods to deduce market estimates and determine forecasts. These methods are explained below:
Market research approaches: Bottom-up
-
Demand estimation of each product across countries/regions summed up to from the total market.
-
Variable analysis for demand forecast.
-
Demand estimation via analyzing paid database, and company financials either via annual reports or paid database.
-
Primary interviews for data revalidation and insight collection.
Market research approaches: Top-down
-
Used extensively for new product forecasting or analyzing penetration levels.
-
Tool used invoice product flow and penetration models Use of regression multi-variant analysis for forecasting Involves extensive use of paid and public databases.
-
Primary interviews and vendor-based primary research for variable impact analysis.
Market research approaches: Combined
- This is the most common method. We apply concepts from both the top-down and bottom-up approaches to arrive at a viable conclusion.
Regional Market Methodology:
The biopharmaceutical CMO and CRO market was analyzed at a regional level. The globe was divided into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa, keeping in focus variables like consumption patterns, export-import regulations, consumer expectations, etc. These regions were further divided into twenty-four countries, namely, the U.S.; Canada; the UK; Germany; France; Italy; Spain; Norway; Sweden; Denmark; China; Japan; India; South Korea; Thailand; Australia; Brazil; Argentina; Colombia; Mexico; South Africa; Saudi Arabia; UAE; and Kuwait.
All three above-mentioned market research methodologies were applied to arrive at regional-level conclusions. The regions were then summed up to form the global market.
Biopharmaceutical CMO and CRO market companies & financials:
The biopharmaceutical CMO and CRO market was analyzed via companies operating in the sector. Analyzing these companies and cross-referencing them to the demand equation helped us validate our assumptions and conclusions. Key market players analyzed include:
-
Boehringer Ingelheim is involved in the R&D of therapeutics for humans and animals. It operates across various therapeutic areas of respiratory, central nervous system, cardiovascular, metabolic, and virological diseases, and oncology. Boehringer BioXcellence is a subsidiary of the parent group, which operates in the contract manufacturing space and caters to pharmaceutical and biotechnology companies. The company offers a portfolio of technologies, including antibody production, non-antibody scaffolds, recombinant proteins, and pDNA from either microbial fermentation or mammalian cell culture systems. The company has geographical presence in the U.S. and the UK, as well as major countries in Europe & Asia.
-
Lonza is a global supplier of biopharmaceuticals. The company offers a wide product range in bioresearch, pharma & biotech, consumer care, water treatment, industrial solutions, agro ingredients, and wood protection. Its operations can be segmented into pharma & biotech and specialty ingredients. In March 2009, the company joined United Nations Global Compact as a strategic initiative to enhance its business. Lonza has worldwide operations in North America, Europe, and Asia, with a strong presence in France, the UK, & other countries.
-
TOYOBO CO., LTD.’s healthcare business operations are run by Toyobo Biologics Inc. The company is engaged in the manufacturing of biopharmaceuticals through its genetic recombination mammalian cell culture technology. It entered the medical field in 1979 and has geographical presence in the U.S., Mexico, Brazil, Saudi Arabia, El Salvador, Thailand, Germany, India, Spain, Hong Kong, Japan, China, South Korea, Taiwan, Malaysia and Indonesia.
-
Thermo Fisher Scientific, Inc. is a publicly held biotechnology company established due to the partnership of Thermo Electron and Fisher Scientific. In 2013, in a competitive bidding with Hoffman La Roche, it acquired Life Technologies Corporation. Thermo Fisher offers products in life sciences, industrial & applied science, and clinical diagnostics. The company operates in more than 50 countries. It offers products through the following brands: Thermo Scientific, Invitrogen, Fisher Scientific, Applied Biosystems, and Unity Lab Services. In August 2017, the company acquired Patheon.
-
Samsung Biologics provides tools and solutions for the biopharmaceutical industry. It is involved in the development and marketing of bio-healthcare products. The company’s business portfolio includes process development, aseptic fill & finish, cGMP manufacturing, quality & compliance, and facilities including cell culture, purification, MSAT laboratory, quality control, and CCR. Its total capacity of mammalian cell culture bioreactors is 182,000 liters.
-
Binex Co., Ltd., formerly known as SoonCheonDang, provides solutions for healthcare operations in Korea. It offers services for development of immune cell therapies for cancer treatment. In 2009, the company acquired Korea Biotechnology Commercialization Center (KBCC) from Korea's Ministry of Knowledge Economy. KBCC offers outsourcing services for biopharmaceutical firms and industries.
-
WuXi Biologics offers services for discovery and production of biologics. Its service portfolio includes solutions for development, discovery, testing, fill & finish, antibody-drug conjugates (ADC) production, and technologies & platforms for biologics production. It has operations across 3 locations in China: Wuxi, Shanghai, and Suzhou. All these facilities are compliant with cGMP regulations.
-
AbbVie, Inc. is a biopharmaceutical company that specializes in the development of advanced therapies for complex diseases. The company has expertise in biologics and targeted therapies. Its direct & indirect distributors are present in more than 170 countries, including the U.S., Italy, Ireland, Puerto Rico, Germany, and Singapore. Its manufacturing plants are located in Sligo, Ireland; Michigan, Illinois, and Massachusetts, U.S.; Ludwigshafen, Germany; Campoverde, Italy; Cork, Ireland; and Barceloneta, Puerto Rico.
-
Charles River Laboratories International, Inc. offers solutions and services in research, clinical support, drug discovery, safety & efficacy assessment, and process manufacturing services to biotechnology, government, pharmaceutical, and academic organizations around the world. Its therapeutic areas include cardiovascular, inflammation, musculoskeletal, respiratory, and CNS metabolic diseases, as well as oncology & ophthalmology. Its product portfolio includes biomarkers, cellular therapeutics, biosimilar, and vaccines. The company is involved in providing services for agrochemicals, chemicals & veterinary medicine, dialysis, compounding pharmacy, home & beauty, medical devices, nutraceuticals, and dietary supplements. It operates in Australia, Canada, China, Finland, France, Germany, India, Ireland, Italy, Japan, the Netherlands, South Korea, Spain, the UK, and the U.S.
-
Syneos Health Inc. provides phase I and phase II clinical development service. The company offers its services to medical devices and biopharmaceutical companies. It has two functional segments, namely Phase I services and clinical development services. The Phase I service segment includes support for Phase I studies for established compounds, exploratory medicine, and first-in-human studies. The clinical development segment offers global studies, clinical monitoring, patient recruitment, quality assurance reviews, consulting services, investigator recruitment, and study reports to facilitate drug development.
Value chain-based sizing & forecasting
Supply Side Estimates
-
Company revenue estimation via referring to annual reports, investor presentations, and Hoover’s.
-
Segment revenue determination via variable analysis and penetration modeling.
-
Competitive benchmarking to identify market leaders and their collective revenue shares.
-
Forecasting via analyzing commercialization rates, pipelines, market initiatives, distribution networks, etc.
Demand side estimates
-
Identifying parent markets and ancillary markets
-
Segment penetration analysis to obtain pertinent
-
revenue/volume
-
Heuristic forecasting with the help of subject matter experts
-
Forecasting via variable analysis
Biopharmaceutical CMO And CRO Market Report Objectives:
-
Understanding market dynamics (in terms of drivers, restraints, & opportunities) in the countries.
-
Understanding trends & variables in the individual countries & their impact on growth and using analytical tools to provide high-level insights into the market dynamics and the associated growth pattern.
-
Understanding market estimates and forecasts (with the base year as 2022, historic information from 2018 to 2021, and forecast from 2023 to 2030). Regional estimates & forecasts for each category are available and are summed up to form the global market estimates.
Biopharmaceutical CMO And CRO Market Report Assumptions:
-
The report provides market value for the base year 2022 and a yearly forecast till 2030 in terms of revenue/volume or both. The market for each of the segment outlooks has been provided on region & country basis for the above-mentioned forecast period.
-
The key industry dynamics, major technological trends, and application markets are evaluated to understand their impact on the demand for the forecast period. The growth rates were estimated using correlation, regression, and time-series analysis.
-
We have used the bottom-up approach for market sizing, analyzing key regional markets, dynamics, & trends for various products and end-users. The total market has been estimated by integrating the country markets.
-
All market estimates and forecasts have been validated through primary interviews with the key industry participants.
-
Inflation has not been accounted for to estimate and forecast the market.
-
Numbers may not add up due to rounding off.
-
Europe consists of EU-8, Central & Eastern Europe, along with the Commonwealth of Independent States (CIS).
-
Asia Pacific includes South Asia, East Asia, Southeast Asia, and Oceania (Australia & New Zealand).
-
Latin America includes Central American countries and the South American continent
-
Middle East includes Western Asia (as assigned by the UN Statistics Division) and the African continent.
Primary Research
GVR strives to procure the latest and unique information for reports directly from industry experts, which gives it a competitive edge. Quality is of utmost importance to us, therefore every year we focus on increasing our experts’ panel. Primary interviews are one of the critical steps in identifying recent market trends and scenarios. This process enables us to justify and validate our market estimates and forecasts to our clients. With more than 8,000 reports in our database, we have connected with some key opinion leaders across various domains, including healthcare, technology, consumer goods, and the chemical sector. Our process starts with identifying the right platform for a particular type of report, i.e., emails, LinkedIn, seminars, or telephonic conversation, as every report is unique and requires a differentiated approach.
We send out questionnaires to different experts from various regions/ countries, which is dependent on the following factors:
-
Report/Market scope: If the market study is global, we send questionnaires to industry experts across various regions, including North America, Europe, Asia Pacific, Latin America, and MEA.
-
Market Penetration: If the market is driven by technological advancements, population density, disease prevalence, or other factors, we identify experts and send out questionnaires based on region or country dominance.
The time to start receiving responses from industry experts varies based on how niche or well-penetrated the market is. Our reports include a detailed chapter on the KoL opinion section, which helps our clients understand the perspective of experts already in the market space.
What questions do you have? Get quick response from our industry experts. Request a Free ConsultationShare this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





