- Home
- »
- Medical Devices
- »
-
Clinical Trial Supplies Market Size, Industry Report, 2033GVR Report cover
![Clinical Trial Supplies Market Size, Share & Trends Report]()
Clinical Trial Supplies Market (2026 - 2033) Size, Share & Trends Analysis Report By Phase (Phase I, Phase II, Phase III), By Service (Cold Chain, Non-cold Chain), By Product (Pharmaceutical, Biologics, Medical device), By Therapeutic Area, By Region, And Segment Forecasts
- Report ID: GVR-1-68038-514-4
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
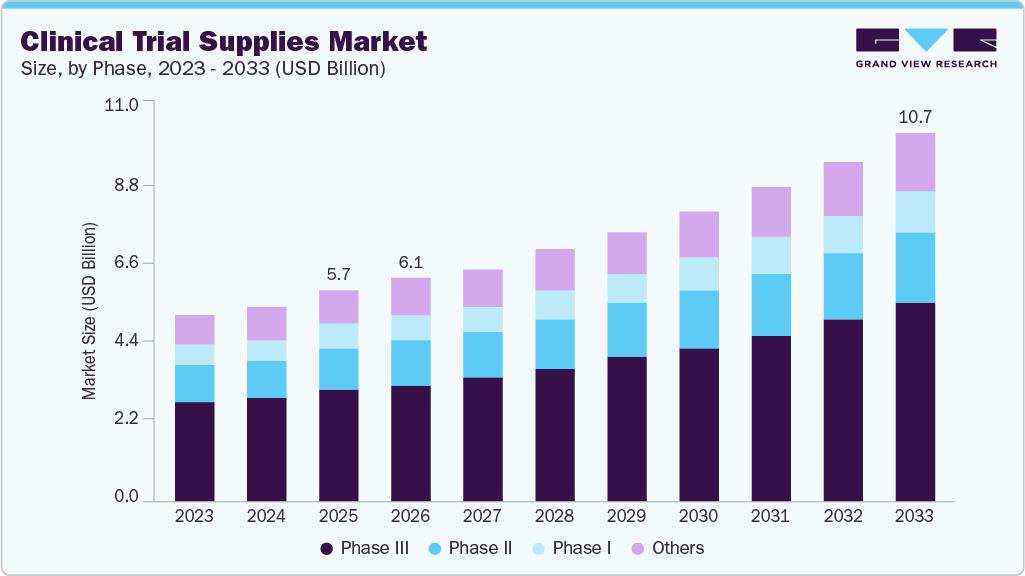
Clinical Trial Supplies Market Summary
The global clinical trial supplies market size was estimated at USD 5.7 billion in 2025 and is projected to reach USD 10.7 billion by 2033, growing at a CAGR of 8.4% from 2026 to 2033. The market growth is driven by increasing R&D investments, the rising prevalence of chronic diseases, and the globalization of clinical trials.
Key Market Trends & Insights
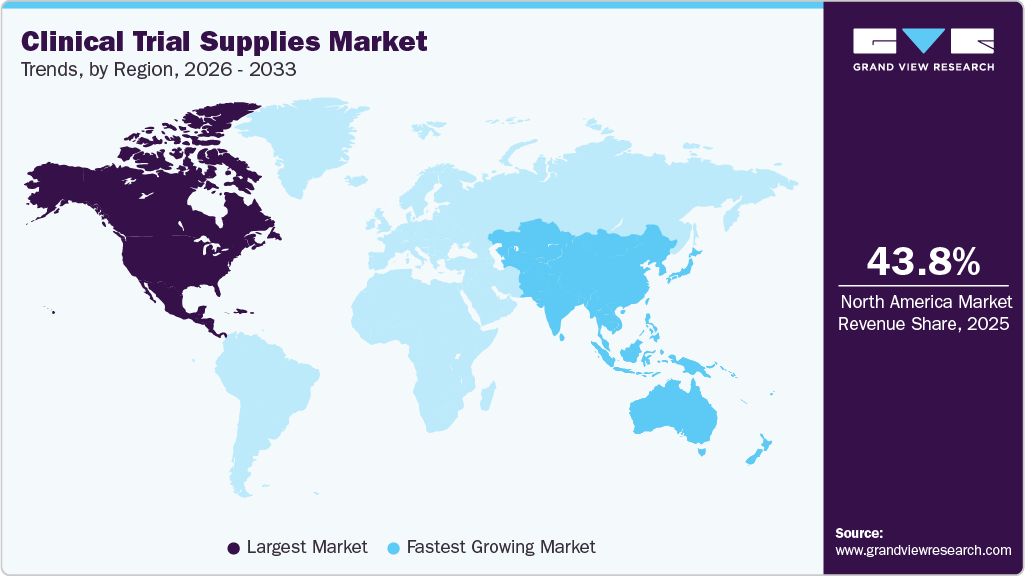
- North America clinical trial supplies market held the largest share of 43.8% of the global market in 2025.
- The clinical trial supplies market in the U.S. is expected to grow significantly over the forecast period.
- Based on the phase, the phase III segment led the market with the largest revenue share of 53.1% in 2025.
- Based on service, the supply chain management segment accounted for the largest market revenue share in 2025.
- Based on therapeutic use, the oncology segment accounted for the largest market revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 5.7 Billion
- 2033 Projected Market Size: USD 10.7 Billion
- CAGR (2026-2033): 8.4%
- North America: Largest market in 2025
- Asia Pacific: Fastest-growing market
Some other factors contributing to the market are the expansion of biologics and personalized medicine, increased demand for complex supply chain solutions, including temperature-sensitive logistics and just-in-time delivery models. Furthermore, growing outsourcing trends among pharmaceutical and biotech companies to specialized supply providers further boost the market expansion.Growing outsourcing trends among pharmaceutical and biotech companies to specialized supply providers drive the market expansion. Besides, increasing focus on biologics and personalized medicine is accelerating the clinical trial supplies industry, requiring specialized supply chain solutions. Biologic drugs, such as monoclonal antibodies, cell therapies, and gene therapies, require highly controlled storage and transportation conditions, including cold chain logistics. As these treatments become more prominent, supply chain providers are strengthening their capabilities to efficiently handle temperature-sensitive materials.
Moreover, the rise of personalized medicine, which customizes treatments based on genetic profiles, has increased the demand for just-in-time (JIT) manufacturing and delivery models. These approaches minimize waste, reduce costs, and ensure that patient-specific drugs reach clinical sites efficiently. Therefore, pharmaceutical companies collaborate with specialized logistics providers to streamline operations and maintain product integrity.
The growing adoption of decentralized clinical trials (DCTs) further enhances the need for flexible supply chain strategies, supporting the broader market expansion by improving accessibility and efficiency in trial execution. Most clinical trials are currently being conducted in developing economies. The increasing cost of clinical trials and complications in the recruitment of patients have encouraged biopharmaceutical companies to outsource clinical trials to regions such as the Asia Pacific, Latin America, Central & Eastern Europe, and the Middle East. Disease variation in developing economies further aids biopharmaceutical companies in performing clinical trials on rare diseases. Asia Pacific also provides more significant economic benefits to biopharmaceutical companies, as governments in Singapore and China allocate funds to promote biomedical research. In Latin America, patient recruitment is easy due to reduced language barriers, which can help obtain informed consent easily, resulting in a faster clinical trial process.
Furthermore, the global clinical trial supplies industry is driven by increasing R&D investments in drug development and the rising prevalence of chronic diseases. Pharmaceutical and biotechnology companies are allocating higher budgets to clinical trials, driven by the growing need for novel therapeutics across oncology, cardiovascular, and rare diseases. This has led to an increase in the number and complexity of clinical trials, requiring efficient supply chain management.
In addition, the rise in chronic diseases among aging populations and lifestyle changes has expanded the demand for clinical trials, further propelling the need for reliable supply solutions. As a result, clinical trial supply providers are investing in advanced logistics, temperature-controlled storage, and real-time monitoring technologies to ensure the integrity of trial materials. The growing regulatory focus on Good Manufacturing Practices (GMP) and Good Distribution Practices (GDP) also imposes robust supply chain solutions to ensure compliance, further fueling the market growth. Thus, the increasing complexity of clinical trials is prompting pharmaceutical and biotechnology companies to outsource their supply chain management to specialized clinical trial supply providers. Outsourcing allows sponsors to utilize the expertise of third-party logistics providers, contract manufacturing organizations (CMOs), and contract research organizations (CROs) that offer specialized solutions for investigational drug distribution, packaging, and regulatory compliance. By partnering with specialized vendors, companies can optimize costs, streamline operations, and mitigate risks associated with supply chain disruptions. Thus, the rising adoption of flexible outsourcing models, including functional service provider (FSP) partnerships and end-to-end supply chain solutions, further accelerates the market growth. As clinical trials expand into emerging markets and decentralized models, there is a rising demand for specialized logistics partners with global reach and regulatory expertise.
Opportunity Analysis
The global clinical trial supplies industry presents new growth opportunities driven by advancements in biologics and personalized medicine, increasing digitalization, outsourcing trends, and expansion into emerging markets. The growing utilization of AI-driven forecasting, blockchain for transparency, and just-in-time logistics to enhance operational efficiency in the supply chain drives overall market growth. As clinical trial models evolve, stakeholders investing in digital innovation, regulatory compliance solutions, and global distribution networks are poised for long-term industry growth.
-
The increasing adoption of biologics, gene therapies, and personalized medicine drives demand for specialized cold chain logistics, just-in-time delivery, and direct-to-patient supply models. Future advancements in cryogenic storage and monitoring technologies will enhance supply chain efficiency.
-
The integration of blockchain, AI-powered forecasting, and IoT tracking is improving supply chain transparency and efficiency. Predictive analytics and smart logistics will further optimize inventory management, reducing trial delays and compliance risks.
-
Pharma companies are increasingly outsourcing clinical trial supply management to specialized logistics providers, optimizing costs and streamlining operations. AI-driven logistics automation and scalable outsourcing solutions will enhance supply chain efficiency and resilience.
-
Expansion into Asia-Pacific, Latin America, and Eastern Europe offers cost advantages and larger patient pools. Future investments in regional cold chain logistics and automated distribution networks will improve trial execution and accessibility.
Impact of U.S. Tariffs on Clinical Trial Supplies Industry
The imposition of U.S. tariffs on the pharmaceutical industry on a broader scope has resulted in a new level of cost pressure in the market. The cost of raw materials, comparator drugs, and packaging materials that are being imported has increased, ultimately causing strain on the overall supply chain. This, in turn, has led to budget constraints along with re-evaluation of the overall strategy sourcing. Furthermore, the tariffs impacted the global supply chain with additional delays and have also forced companies to look for other sources of supplies. Thus, the companies are slowly moving towards local manufacturing to avoid the risks associated with the tariffs. However, there are certain challenges associated with this process, wherein small biopharma companies will face several challenges to relocate their clinical trial assemblies and/or friend-shore. Thus, the tariff imposition has affected the costs as well as the way clinical trials are conducted.
Technological Advancements
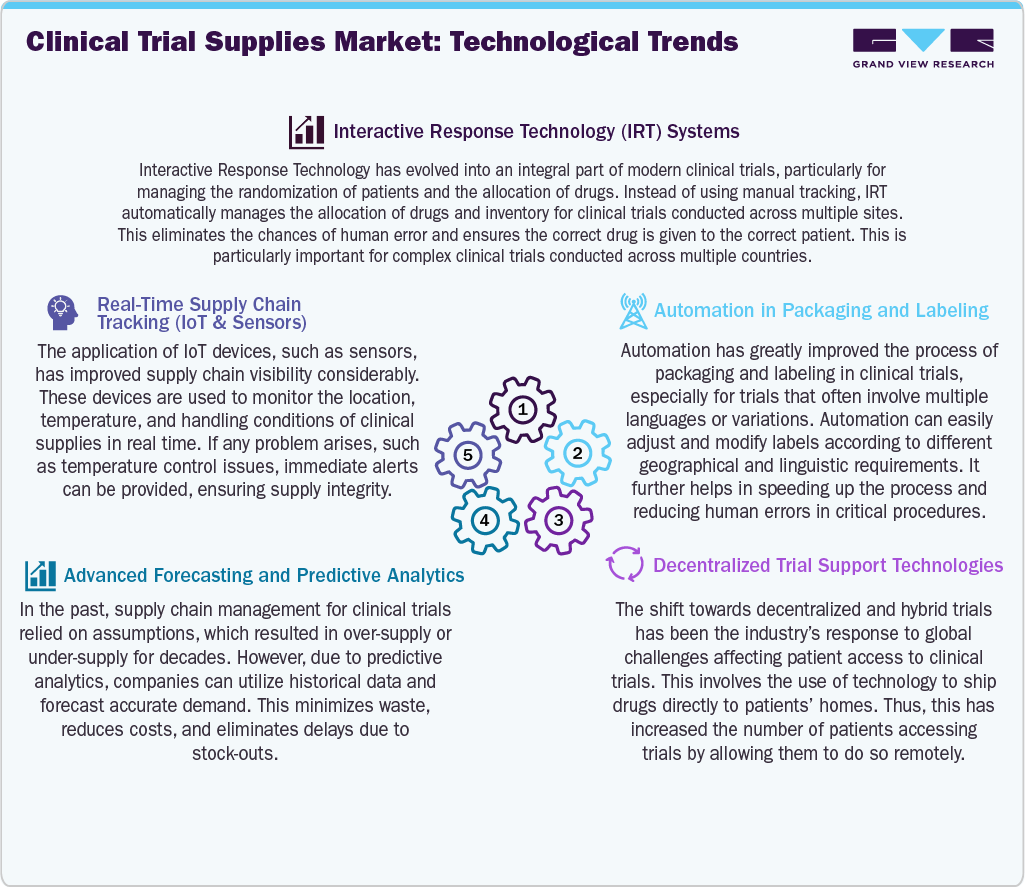
The market is becoming increasingly efficient and streamlined with the help of technological advances. Technologies like interactive response technology have helped make clinical trial operations easier, with the randomization of patients and the allocation of drugs. Besides, the Internet of Things has helped track the conditions of shipments, thus maintaining the quality of the drugs, especially in the case of sensitive drugs. In addition, the use of predictive analysis has helped in the accurate forecast of needs and the avoidance of any imbalances in the supplies. With the onset of decentralized clinical trials, the use of technology has improved the platforms that aid in the direct-to-patient delivery of drugs. Thus, the use of automation in the process of packaging and labeling has further sped up the process of clinical trials.
Pricing Model Analysis
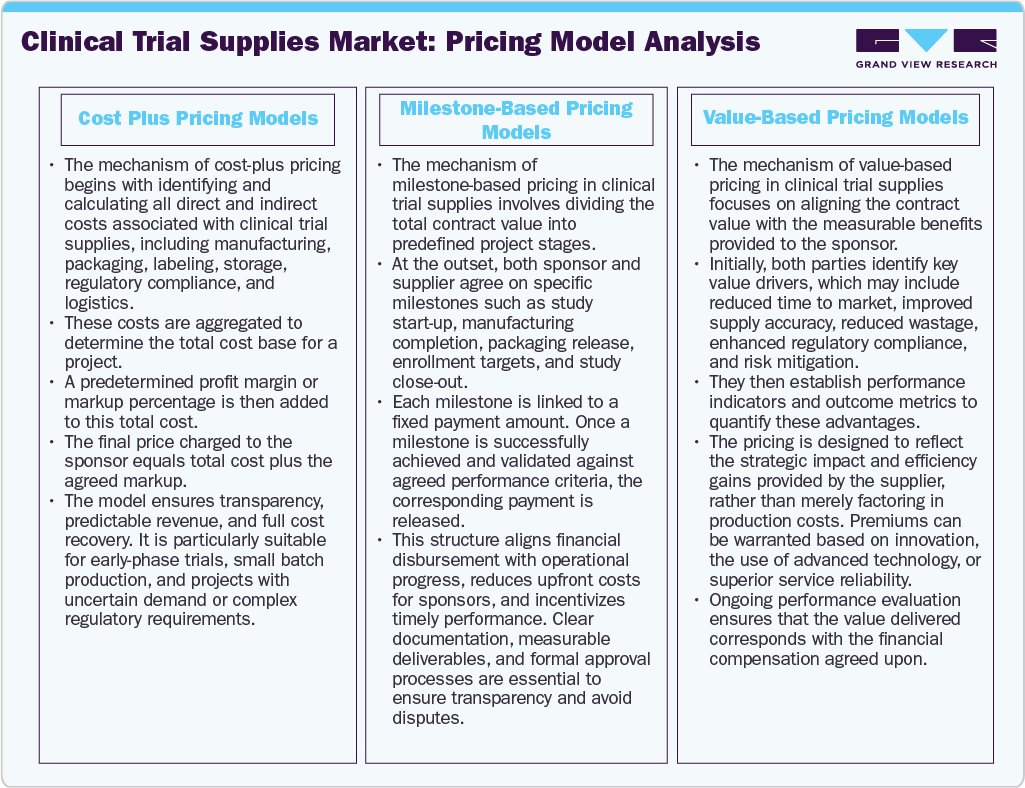
The analysis of pricing models in the market focuses on several factors, including cost structures, demand variability, regulatory complexities, and risk management. Companies often employ a hybrid pricing strategy that combines different models: cost-plus, milestone-based, and value-based pricing. Cost-plus pricing is used to ensure that all expenses related to manufacturing, packaging, labeling, storage, and logistics are recovered. Milestone-based pricing is designed to align payments with specific phases of the trial and enrollment targets, providing a clear structure for financial transactions as the trial progresses. Value-based pricing is referred to for more complex products, like biologics or temperature-sensitive items, as well as for global distribution services. In addition, dynamic pricing strategies are becoming more prevalent to address challenges such as supply chain uncertainties, currency fluctuations, and the need for shipping. This approach further supports maintaining profitability while preserving strong relationships with sponsors.
Market Concentration & Characteristics
The market growth stage is medium, and the pace is accelerating. The clinical trial supplies industry is characterized by a high degree of innovation, a level of M&A activities, the impact of regulations, service expansion, and regional expansion.
In the clinical trial supplies industry, the level of innovation is increasingly revolving around the digitalization and rationalization of supply. With the integration of technology, the standard level of innovation has elevated to the level of automated inventory and real-time tracking. Furthermore, the partnership between clinical trial supply companies and pharmaceutical companies has allowed for the development of flexible clinical trials, including support for decentralized clinical trials. The continued investment in data, packaging, and cold chain solutions has allowed efficiency, reduction of wastage, and timely delivery of clinical trial materials.
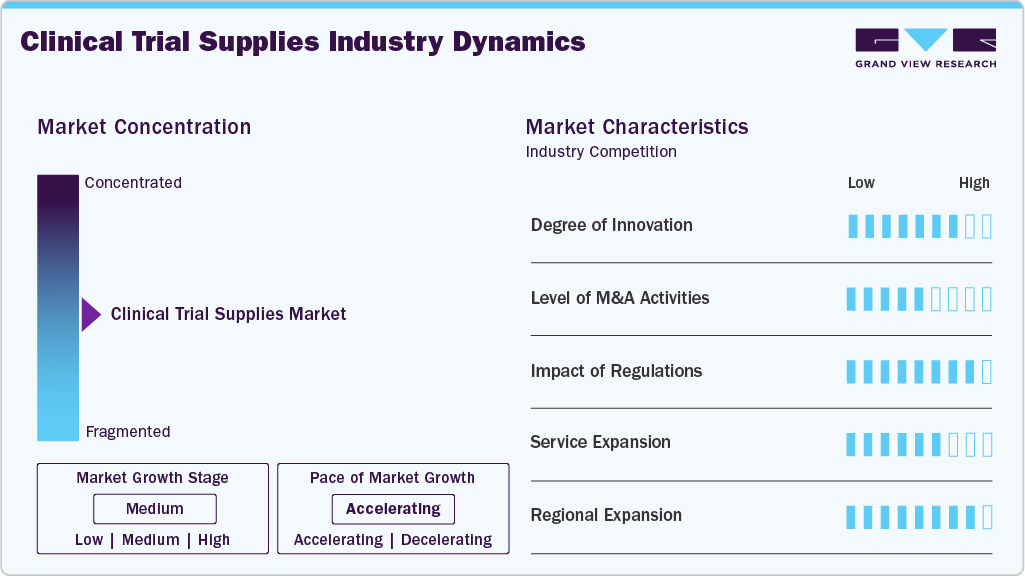
Regulatory requirements play a major role in the clinical trial supply industry. Regulatory agencies such as the U.S. FDA and the EMA have issued guidelines for the need for clinical trial supply firms to ensure strict compliance with regulatory needs. Although these increase the level of operational complexity and costs, they also generate the need for specialized firms with the knowledge and expertise to ensure smooth clinical trial supply.
The level of M&A activities is increasing in the industry, as companies are looking to enhance their capabilities and service offerings. Organizations are acquiring niche companies that specialize in certain areas, like cold chain logistics, packaging, or comparator sourcing. These activities are proving to be a boon for the overall industry, as companies are expanding their footprint and capabilities to manage complex global trials. As the industry is growing, the level of competition is increasing due to M&A activities in the industry.
The service providers in the industry are expanding their services to include more than just supply and distribution activities. Organizations are expanding to include services like forecasting, packaging, storage, and returns management, among others. This is proving to be a boon for the overall industry, as sponsors can look to a single service provider for multiple services, which can help streamline the overall process, saving time and reducing the overall complexities involved.
Regional expansion supports the companies in expanding their presence in significant geographical regions. North America and Europe are the dominant markets for clinical trials due to high clinical research activities in these regions. In addition to this, the Asia-Pacific region is also gaining importance for clinical trials. Countries in this region are gaining more clinical trials due to cost advantages and a growing patient population. This has helped companies to set up regional hubs to serve clinical trials in the region.
Phase Insights
Based on the clinical phase segment, the phase III segment dominated the market in 2025, accounting for a revenue share of 53.1%. The growth is driven by large patient enrollment, longer study timelines, and highly complex logistical requirements. In addition, rising R&D spending, increasing biologics development, and strict regulatory standards continue to fuel demand for clinical trial supplies. Furthermore, enhanced cold chain infrastructure, just-in-time inventory systems, and widespread global site distribution are strengthening the need for efficient and well-coordinated clinical trial supply management solutions.
The phase I clinical trials segment is expected to grow significantly during the forecast period, attributed to the rising outsourcing of early-phase studies. In addition, smaller patient populations and significant capital requirements are key factors encouraging sponsors to outsource a large share of these trials. It has been identified that about 54.0% of phase I clinical trials are outsourced, and the number is expected to increase owing to the globalization of clinical trials.
Service Insights
The supply chain management segment accounted for the largest revenue share in 2025. The segment growth is driven by the growing complexity of multinational clinical trials and the need for streamlined logistics operations. The segment encompasses inventory control, packaging, labeling, storage, and distribution services. In addition, integration of advanced technologies such as AI-based demand forecasting, blockchain-enabled traceability, and IoT-supported tracking systems improves operational agility. As decentralized trials and biologics expand, optimized supply chain solutions are vital for timely and compliant trial execution.
The storage and distribution is projected to be the fastest-growing segment, driven by the globalization of clinical trials. The segment ensures that investigational drugs reach trial sites safely, on time, and in compliance with regulatory standards. Besides, proper storage further supports drug stability, potency, and safety, requiring controlled environments such as temperature- and humidity-regulated facilities, including cold chain solutions for biologics & temperature-sensitive compounds. Serialization, tamper-evident packaging, and real-time monitoring systems are employed to prevent counterfeiting, track inventory, and ensure product integrity throughout the supply chain.
Product Insights
Pharmaceutical accounted for the largest revenue share in 2025, owing to rising R&D investments, expansion of biologics and personalized medicine pipelines, and increasing outsourcing of clinical supply operations. In addition, a growing number of global and decentralized trials are expected to drive the demand for integrated, regulatory-compliant supply chain solutions that ensure the timely delivery of investigational products across multiple geographies.
The biologics segment is projected to record the fastest growth in the market, driven by the rising development of monoclonal antibodies, cell and gene therapies, and personalized medicines. In addition, increasing early-phase biologics pipelines, higher outsourcing of sterile fill/finish, and the need for temperature-controlled logistics are accelerating demand for specialized manufacturing, packaging, and distribution solutions across global clinical trials.
In addition, expansion of sterile fill/finish capacity is a major driver supporting biologics trials. For instance, in February 2025, Selkirk Pharma introduced ClinFAST to expedite small-batch injectable production, addressing capacity bottlenecks that often delay biologics studies. Besides this, rapid turnaround services, GMP-compliant aseptic processing, and prioritization of early-stage biotech programs are critical to meeting tight clinical milestones and reducing time-to-trial initiation.
Therapeutic Area Insights
Based on therapeutic area, the oncology segment dominated the market with the largest revenue share in 2025. The segment’s growth is due to rising cancer prevalence, expanding oncology trial activity, and increasing demand for biologics and immunotherapies. In addition, growing oncology pipelines are a major factor contributing to the market growth. Furthermore, the shift toward biomarker-driven therapies, personalized medicine, and decentralized oncology trials increases supply chain complexity.
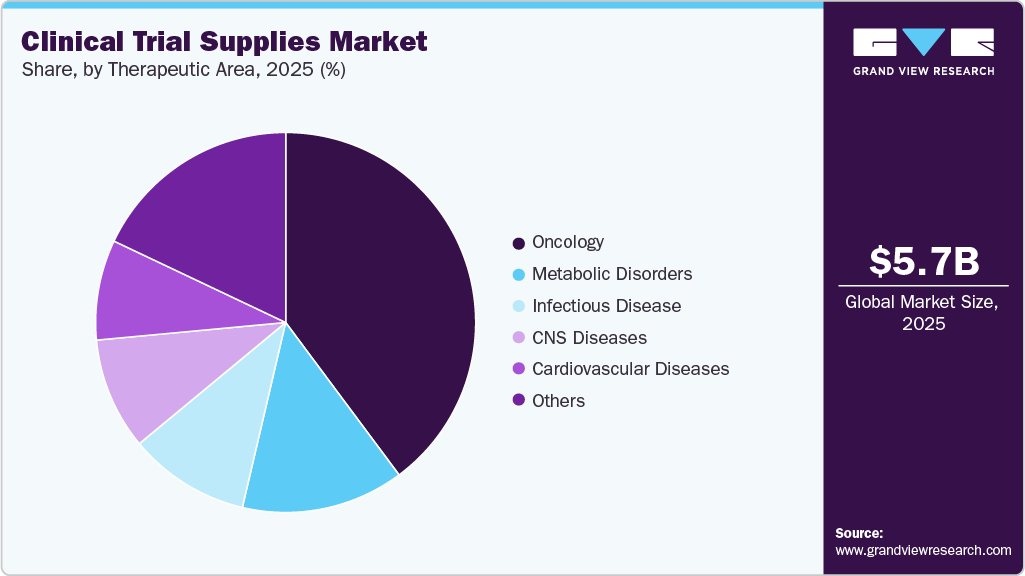
The cardiovascular diseases segment is expected to grow at the fastest CAGR during the forecast period. The segment is driven by increasing R&D investments in innovative therapeutics and rising sedentary lifestyles among individuals. According to the World Health Organization (WHO) report published in July 2025, an estimated 19.8 million deaths around the globe were attributed to cardiovascular diseases (CVDs) in 2022, accounting for nearly 32% of total global mortality. Besides this, the growing development of biologics, gene therapies, and personalized cardiovascular medicines is expanding trial complexity, thereby increasing demand for advanced supply chain, storage, and distribution services across the global study network.
Regional Insights
North America dominated the global clinical trial supplies market in 2025, holding a revenue share of 43.8%. The growth of this region is due to expanding biologics pipelines, decentralized trial adoption, global supply chain expansion, and rising demand for temperature-controlled logistics. In addition, increasing outsourcing and infrastructure investments are strengthening distribution efficiency, improving trial continuity, and accelerating market growth across complex multi-regional studies.
The expanding biologics and personalized medicine pipelines are increasing demand for specialized storage, packaging, and distribution capabilities. For instance, in 2025, the Center for Drug Evaluation and Research (CDER) authorized 46 new drugs, including new molecular entities (NMEs) through New Drug Applications (NDAs) and new therapeutic biologics via Biologics License Applications (BLAs). Besides this, growth in temperature-sensitive investigational products requires advanced cold chain systems, forecasting tools, and integrated supply planning, driving investment in infrastructure, logistics partnerships, and adaptive supply strategies across North America’s clinical research ecosystem. Such factors are expected to drive the market over the forecast period.
U.S. Clinical Trial Supplies Market Trends
The clinical trial supplies market in the U.S. accounted for the highest share in North America, owing to high drug development activity, strong R&D investment, favorable FDA support, and AI-powered logistics adoption. In addition, rising clinical trial activities, advanced cold chain infrastructure, and expanding biologics and gene therapy trials across nationwide research networks. Besides, rising automation and interoperability, accelerating supply efficiency are further contributing to market growth. For instance, in June 2025, Oracle enhanced Randomization and Trial Supply Management to enable drug pooling across studies, real-time inventory visibility, and multi-vendor coordination, reducing waste, optimizing allocation, and speeding U.S. trial execution timelines. This strengthens cross-trial flexibility for sponsors and CROs nationwide while improving planning accuracy significantly.
Canada clinical trial supplies market is advancing with a significant CAGR over the forecast period, through expanding precision medicine initiatives, rising genomics-driven research, growing global supply chain integration, and increasing infrastructure investment supporting storage, packaging, and distribution for complex clinical studies nationwide. Canada’s precision health expansion is accelerating demand for specialized clinical trial supplies. For instance, in March 2025, Genome Canada launched the Canadian Precision Health Initiative to build large-scale genomic datasets, strengthening personalized therapy development. This initiative drives increased clinical trial activity, requiring advanced supply planning, temperature-controlled logistics, and reliable distribution networks supporting biologics and gene-based therapies across Canada.
Europe Clinical Trial Supplies Market Trends
The clinical trial supplies market in Europe is driven by rising regulatory harmonization, expanding cold-chain infrastructure, cross-border trial coordination, and advanced logistics capabilities. In addition, increasing biologics development also strengthens the demand for specialized packaging, storage, and distribution solutions across the region. Besides this, regulatory alignment led by the European Medicines Agency (EMA) streamlines multi-country trials, reducing duplication in approvals and improving supply planning. The harmonized requirements enable standardized labeling, packaging, and distribution processes. This environment attracts global sponsors, accelerates trial initiation timelines, and drives consistent demand for compliant clinical supply storage, comparator sourcing, and temperature-controlled logistics services across European markets.
Germany clinical trial supplies market is expanding due to its strong pharmaceutical manufacturing base, advanced research infrastructure, and high clinical trial density. In addition, government R&D incentives, robust cold-chain logistics, and strategic investments in clinical supply capacity strengthen Germany’s leadership in Europe and support efficient investigational product packaging, storage, and distribution. Some of the strategic investments and collaborations are strengthening supply capabilities and accelerating trial execution. For instance, in October 2024, the collaboration between Biond Biologics and Merck KGaA, Darmstadt, Germany, for a Phase 1 oncology study strengthens Germany’s role in innovative biologics research.
The clinical trial supplies market in the UK is rising due to strong government support, an expanding biotech ecosystem, and post-Brexit investment in domestic supply-chain infrastructure. In addition, strategic collaborations between academia, industry, and healthcare institutions enhance trial execution efficiency, while regulatory agility supports faster study initiation and reliable investigational product distribution across the country.
Regulatory policies, such as post-Brexit policies, are accelerating localized infrastructure development to maintain uninterrupted clinical supply flows. Besides this, investments in domestic packaging, storage, and distribution capabilities reduce dependency on EU hubs and improve resilience. These developments enhance logistics reliability, strengthen temperature-controlled handling capacity, and increase demand for integrated supply services supporting complex biologics and advanced therapies across UK research networks.
Asia Pacific Clinical Trial Supplies Market Trends
The clinical trial supplies market in the Asia Pacific is expected to grow at a fastest CAGR over the forecast period. The regional growth is driven by cost-effective trial operations, diverse patient recruitment, improved regulatory frameworks, expanding biologics pipelines, and rapid cold chain logistics advancement. Besides this, increasing regional trial activity and infrastructure investment further strengthen the Asia Pacific’s role as a global clinical development hub. The rising clinical trial activity across APAC is significantly transforming global supply chains. According to a report published in December 2025, the clinical trial activity is growing two to three times faster than in Europe, increasing demand for regional packaging, storage, and distribution capabilities. Besides this, infrastructure expansion by providers is supporting complex biologics and cell therapy trials across emerging research markets. For instance, in November 2025, Almac expanded its Singapore facility with a multi-million-pound investment to increase warehouse capacity, cold chain packaging, and regional supply expertise. The expansion strengthens APAC clinical trial logistics, enhances temperature-controlled distribution capabilities, and supports rising demand for complex global clinical supply operations. Such initiatives boost market growth.
China clinical trial supplies market is driven by strong government support, a rapidly expanding pharmaceutical sector, and regulatory reforms by the National Medical Products Administration (NMPA). The country’s large patient pool attracts global trials, whereas investments in cold chain logistics and AI-driven supply management enhance efficiency. Besides, increasing biologics development further accelerates demand for specialized clinical trial supply services and infrastructure expansion.
The clinical trial supplies market in Japanis expected to grow at the fastest CAGR over the forecast period. The growth is driven by strong pharmaceutical innovation, regulatory efficiency under the PMDA, high biologics investment, advanced healthcare infrastructure, and rising adoption of decentralized trials and digital supply chain solutions. In addition, Japan's leadership in biologics and precision medicine is accelerating demand for specialized clinical supply services, including temperature-controlled storage, advanced packaging, and adaptive distribution models. For instance, in June 2022, Catalent expanded its primary packaging capabilities at its Shiga clinical supply facility, strengthening regional infrastructure, improving packaging flexibility, logistics efficiency, and global trial integration, thereby enhancing Japan’s attractiveness for multinational clinical programs.
India clinical trial supplies market is expected to grow at the fastest CAGR over the forecast period. The country’s growth is driven by cost-effective trial operations, a large patient pool, favorable regulatory reforms, rising pharmaceutical R&D investments, and improving cold chain logistics and digital supply tracking capabilities across the country. Such aforementioned factors are expected to drive the market.
Latin America Clinical Trial Supplies Market Trends
The clinical trial supplies market in Latin America is expected to significantly grow over the estimated time period. The regional growth is driven by lower operational costs, diverse patient populations, expanding pharmaceutical investments, and strengthening logistics infrastructure supporting regional and global clinical research programs. In addition, rising collaboration between sponsors and supply partners is accelerating market development. For instance, in September 2025, Bayer highlighted early engagement with suppliers, transparency, and integrated planning to improve trial continuity and supply reliability. Such partnership-driven models enhance forecasting accuracy, reduce delays, and strengthen long-term supply networks across Latin America, supporting complex multinational clinical trials and improving overall operational efficiency.
Brazil clinical trial supplies market is expanding due to strong government initiatives, a large and genetically diverse patient pool, and growing pharmaceutical R&D investment. In addition, regulatory streamlining by ANVISA and improved cold chain logistics are enhancing trial feasibility, positioning Brazil as a key regional hub for clinical research supply operations. Furthermore, rising pharmaceutical investment and trial diversification further accelerate supply and demand. For instance, in August 2025, Sanofi doubled clinical research investment in Brazil, expanding studies into underserved regions to improve participant diversity. The broader geographic trial networks increase logistical complexity and supply volumes, strengthening demand for temperature-controlled distribution, regional depots, and integrated supply chain solutions supporting biologics and precision therapies.
Middle East & Africa Clinical Trial Supplies Market Trends
The clinical trial supplies market in the MEA is expected to experience steady growth over the forecast period. The market is driven by rising healthcare investments, regulatory advancements, and increasing pharmaceutical collaborations. Besides this, increasing healthcare investments across MEA are improving hospital infrastructure, research funding, and specialized clinical facilities, directly strengthening trial supply demand. In addition, governments are prioritizing healthcare modernization and local manufacturing capacity, enabling more clinical studies. These investments enhance storage, packaging, and distribution networks for investigational products, supporting efficient supply management and positioning the region as an emerging clinical research destination globally.
South Africa clinical trial supplies market is driven by expanding temperature-controlled logistics capacity, rising clinical research infrastructure utilization, and increasing inclusion in global trials across diverse therapeutic areas. In addition, strategic logistics investments and broader clinical trial representation across the continent position the country as a regional hub for supply distribution, storage, and specialized handling of investigational products. Furthermore, growing recognition of Africa’s unrecognized clinical trial potential supports market expansion. South Africa hosts a significant share of the continent’s trials, supported by established research sites, improving regulatory capacity, and international collaborations. In addition, increasing global sponsor interest in diverse populations and expanding disease-area research elevate demand for sourcing, packaging, and distribution solutions, strengthening the country’s competitive clinical trial supply ecosystem.
The clinical trial supplies market in the UAEis expanding due to advanced healthcare infrastructure, regulatory efficiency, precision medicine initiatives, and strong global pharmaceutical partnerships. Besides this, rapid ethics approvals, AI-enabled research systems, and diverse patient demographics support complex trials, increasing demand for clinical trials across the country’s evolving clinical research ecosystem. In addition, infrastructure modernization and genomics-driven research are growth factors. The national strategies, such as UAE Vision 2031 and programs such as the Emirati Genome Program, enhance patient stratification and trial efficiency. Moreover, AI-enabled platforms, electronic medical records, and precision medicine capabilities improve recruitment accuracy, reduce screening failures, and increase demand for specialized supply chain services supporting biomarker-driven and rare disease trials.
Key Clinical Trial Supplies Company Insights
The key players operating across the market are adopting strategic initiatives such as service and product launches, mergers & acquisitions, partnerships & agreements, and expansions to gain a competitive edge in the market. For instance, in January 2023, Catalent launched a supply case management service for cell and gene therapy supply chains, providing end-to-end coordination, traceability, and logistics oversight. The initiative improves delivery reliability, reduces disruption risks, and enhances operational control for advanced therapy clinical trial distribution globally.
Key Clinical Trial Supplies Companies:
The following key companies have been profiled for this study on the clinical trial supplies market.
- Almac Group
- Biocair
- Catalent Inc.
- KLIFO
- Movianto
- PCI Pharma Services
- Sharp Services, LLC
- Thermo Fischer Scientific Inc.
- United Parcel Service of America, Inc (UPS Healthcare)
- Parexel International Corporation
Recent Developments
-
In November 2025, Movianto partnered with Rangel to deliver integrated pharmaceutical logistics across Portugal and Europe. The collaboration expands GDP-compliant warehousing and cold chain distribution, improving cross-border clinical trial supply access, accelerating market entry, and strengthening pan-European trial logistics capabilities for sponsors.
-
In November 2025, Almac expanded its Singapore facility with a multi-million-pound investment to increase warehouse capacity, cold chain packaging, and regional supply expertise. The expansion strengthens APAC clinical trial logistics, enhances temperature-controlled distribution capabilities, and supports rising demand for complex global clinical supply operations.
-
In August 2025, Biocair expanded APAC operations with a new Shanghai office and warehouse near Pudong International Airport, strengthening regional logistics capacity. The expansion enhances temperature-controlled distribution, improves operational coordination, and supports growing clinical trial supply demand across Asia-Pacific markets.
Clinical Trial Supplies Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 6.1 billion
Revenue forecast in 2033
USD 10.7 billion
Growth rate
CAGR of 8.4% from 2026 to 2033
Historical year
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD billion/million and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Phase, service, product, therapeutic area, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Thailand; South Korea; Australia; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait; Oman; Qatar
Key companies profiled
Almac Group; Biocair; Catalent Inc.; KLIFO; Movianto; PCI Pharma Services; Sharp Services, LLC; Thermo Fischer Scientific Inc.; United Parcel Service of America, Inc. (UPS Healthcare); Parexel International Corporation
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Clinical Trial Supplies Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global clinical trial supplies market based on product, service, phase, therapeutic area, and region:
-
Phase Outlook (Revenue, USD Million, 2021 - 2033)
-
Phase I
-
Phase II
-
Phase III
-
Others
-
-
Service Outlook (Revenue, USD Million, 2021 - 2033)
-
Manufacturing
-
Storage & Distribution
-
Cold Chain
-
Non-cold Chain
-
-
Supply Chain Management
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical
-
Biologics
-
Medical device
-
Others
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Oncology
-
CNS Diseases
-
Cardiovascular Diseases
-
Infectious Diseases
-
Metabolic Disorders
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
UAE
-
Saudi Arabia
-
Kuwait
-
Qatar
-
Oman
-
-
Frequently Asked Questions About This Report
b. The global clinical trial supplies market size was estimated at USD 5.7 billion in 2025 and is expected to reach USD 6.1 billion in 2026.
b. The global clinical trial supplies market is expected to grow at a compound annual growth rate of 8.44% from 2026 to 2033 to reach USD 10.7 billion by 2033.
b. North America dominated the clinical trial supplies market with a share of 43.84% in 2025. The regional growth is attributed to expanding biologics pipelines, decentralized trial adoption, global supply chain expansion, and rising demand for temperature-controlled logistics. In addition, increasing outsourcing and infrastructure investments are strengthening market growth across complex multi-regional studies.
b. Some key players operating in the clinical trial supplies market include Almac Group, Biocair, Catalent Inc., KLIFO, Movianto, PCI Pharma Services, Sharp Services, LLC, Thermo Fischer Scientific Inc., United Parcel Service of America, Inc (UPS Healthcare), and Parexel International Corporation, among others.
b. Key factors that are driving the clinical trial supplies market growth include increasing R&D investments, the rising prevalence of chronic diseases, and the globalization of clinical trials. Some other factors contributing to the market are the expansion of biologics and personalized medicine, increased demand for complex supply chain solutions, including temperature-sensitive logistics and just-in-time delivery models.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










