- Home
- »
- Pharmaceuticals
- »
-
Colorectal Cancer Therapeutics Market Size Report, 2030GVR Report cover
![Colorectal Cancer Therapeutics Market Size, Share & Trends Report]()
Colorectal Cancer Therapeutics Market Size, Share & Trends Analysis Report By Drug Class (Chemotherapy, Immunotherapy), By Region (North America, Asia Pacific, Latin America, Europe), And Segment Forecasts, 2023 - 2030
- Report ID: GVR-2-68038-512-0
- Number of Report Pages: 100
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
Report Overview
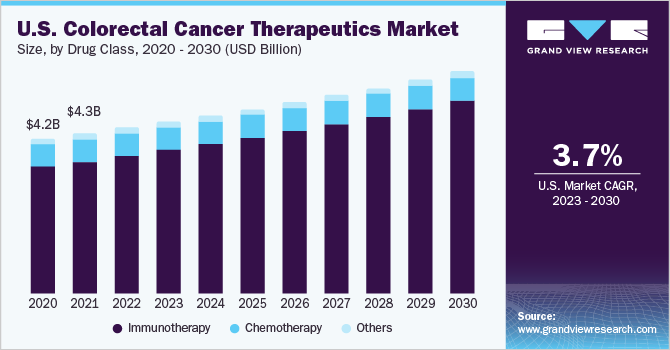
The global colorectal cancer therapeutics market size was estimated at USD 11.5 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4.7% from 2023 to 2030. According to a study published in the National Library of Medicine in October 2021, colorectal cancer (CRC) accounted for approximately 10.0% of the global cancer incidence in 2020, and 9.4% deaths, making it the second deadliest cancer type, following lung cancer, which accounted for 18% cancer deaths worldwide. Causes of colorectal cancer are unspecified. However, several environmental and genetic factors may increase the risk of developing the disease. According to the Colorectal Cancer Facts & figures 2020-2022, published by the American Cancer Society, about 1 in 25 women and 1 in 23 men in the U.S. are estimated to develop CRC during their lifetime.
Whereas, according to the World Health Organization, approximately 2 million people around the world were diagnosed with colorectal cancer in 2020. However, the CRC mortality has been declining over the last few years due to advanced screening and diagnostics as well as improved treatments, which is likely to drive the growth of this market over the forecast period. Moreover, factors such as the emergence of more front-line therapies, upcoming launches of promising pipeline products, and favorable government initiatives are likely to drive market expansion. In June 2023, the Food and Drug Administration department of the U.S. approved a genomic profiling test FoundationOneLiquid CDx, which can be used for the identification of patients with BRAF V600E Alterations in cases of metastatic colorectal cancer.
This product is introduced to assist in identifying the patients who are eligible for treatment with BRAFTOVI and cetuximab, which can further help in providing appropriate treatments. Furthermore, an aging population, rising awareness, and different government initiatives to provide better and more affordable treatments coupled with favorable reimbursement policies are expected to foster the market growth. Increasing acceptance of advanced therapies, such as Avastin (bevacizumab), Erbitux (cetuximab), and Stivarga (regorafenib), is also expected to drive market growth. Several biologics related to this market, such as Vectibix, and Zaktrap, are expected to lose their exclusivity over the forecast period, which can further foster the demand in this market.
Avastin, which is used in the treatment of some advanced levels of cancer, expired in July 2019 in the U.S. and in January 2022 in Europe. The Amgen and Allergan’s Mvasi became first biosimilar to Avastin to be approved in the U.S. and Europe, marking the entry of biosimilars in the field of oncology. Moreover, according to an article published by the AJMC Center For Biosimiliars in February 2020, 20 oncology biologics were expected to expire by 2023. These patent expirations are likely to foster market growth over the forecast period. The industrial pipeline is exploring treatment options in rare types, such as BRAF-mutant or dMMR/MSI-H metastatic CRC. However, neglected areas, such as high-risk resectable CRC present excellent opportunities for drug developers. Easy availability of treatment options and more targeted therapies indicate a promising future for the global market.
Drug Class Insights
On the basis of drug class, the market is segmented into chemotherapy, immunotherapy, and others. The immunotherapy segment dominated the market with a revenue share of 83.0% in 2022 and is expected to grow at the fastest CAGR of 5.3% over the forecast period. Immunotherapeutic class has shown fewer adverse effects compared to chemotherapeutics. Non-specificity, toxic side effects, and development of resistance associated with chemotherapy are likely to encourage adoption of immunotherapeutics in the future.
Moreover, a study published in the National Library of Medicine in June 2020 has shown that application of immunotherapy for the treatment of colorectal cancer has exhibited remarkable outcomes for patients having microsatellite instability or mismatch-repair-deficient mutations in their tumors. Such positive research and results are likely to boost the growth of this segment in the future.
Regional Insights
North America accounted for the largest revenue share of 42.6% in 2022. The market is projected to expand steadily, propelled by high incidence of CRC in the country, increase in treatment rates, inflated medicine prices compared to other major regions, and changing lifestyle choices, such as increasing alcohol consumption. For instance, according to The International Agency for Research on Cancer (IARC), consumption of alcohol can be related to CRC and around 60% of the adult population in the U.S. consumed alcohol in 2021.
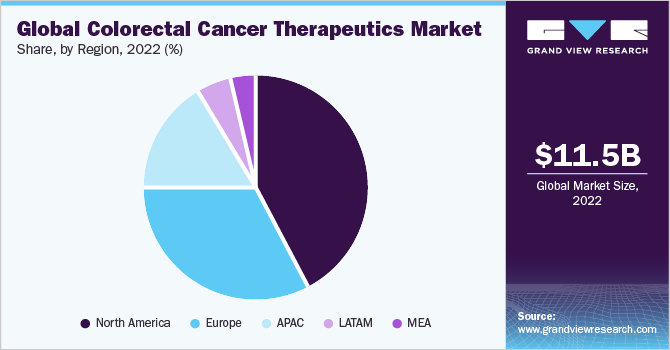
Asia Pacific is expected to grow at the highest CAGR of 6.7% during the forecast period due to the rising geriatric population and increasing participation of public and private establishments to organize awareness programs on colorectal cancer. For instance, according to a study published in the National Library of Medicine in April 2022, several pilot population-based CRC screening programs have been implemented in some administrative divisions of China, which can help in the early diagnosis and treatment of these diseases.
Key Companies & Market Share Insights
The key players in the market are focusing on product approvals & launches, strategic partnerships, mergers & acquisitions, and global expansions to strengthen their industry position. For instance, in January 2023, Akeda of Japan entered into a partnership with Hong Kong-based Hutchmed, to get commercial rights for colorectal cancer drug fruquintinib outside of China. Furthermore, Roche is evaluating the combination of targeted therapy (Cotellic) with immuno-oncologic agents for the treatment of CRC. These developments are likely to foster new developments and growth in the market. Some of the major participants of in the global colorectal cancer therapeutics market include:
-
Amgen, Inc.
-
Bayer AG
-
Bristol-Myers Squibb Company
-
Lilly
-
Merck & co., Inc.
-
F. Hoffmann-La Roche Ltd.
-
Sanofi
Colorectal Cancer Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 12.1 billion
Revenue forecast in 2030
USD 16.7 billion
Growth rate
CAGR of 4.7% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Report updated
September 2023
Quantitative units
Revenue in USD million and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Drug class and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Amgen Inc; Bayer AG; Bristol-Myers Squibb Company; Lilly; Merck & co., Inc.; F. Hoffmann-La Roche Ltd; Sanofi
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Colorectal Cancer Therapeutics Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the global colorectal cancer therapeutics market report on the basis of drug class and region:
-
Drug Class Outlook (Revenue in USD Million, 2018 - 2030)
-
Chemotherapy
-
Immunotherapy
-
Others
-
-
Regional Outlook (Revenue in USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global colorectal cancer therapeutics market size was estimated at USD 11.5 billion in 2022 and is expected to reach USD 12.1 billion in 2023.
b. The global colorectal cancer therapeutics market is expected to grow at a compound annual growth rate of 4.70% from 2023 to 2030 to reach USD 16.7 billion in 2030.
b. Immunotherapy led the colorectal cancer therapeutics market by drug class, with a share of close to 83.0% in 2022.
b. Some of the key players are Amgen, Bayer, Bristol-Myers Squibb, Eli Lilly, Merck, Roche, and Sanofi.
b. Factors such as emergence of more front-line therapies, upcoming launches of promising pipeline candidates, and favorable government initiatives are likely to fuel market expansion.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





