- Home
- »
- Medical Devices
- »
-
Deep Brain Stimulation Devices Market, Industry Report 2033GVR Report cover
![Deep Brain Stimulation Devices Market Size, Share & Trends Report]()
Deep Brain Stimulation Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Single Channel, Dual Channel), By Application (Pain Management, Epilepsy, Essential Tremor), By End-use (Hospital, Neurology Clinics) By Region, And Segment Forecasts
- Report ID: 978-1-68038-335-5
- Number of Report Pages: 300
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Deep Brain Stimulation Devices Market Summary
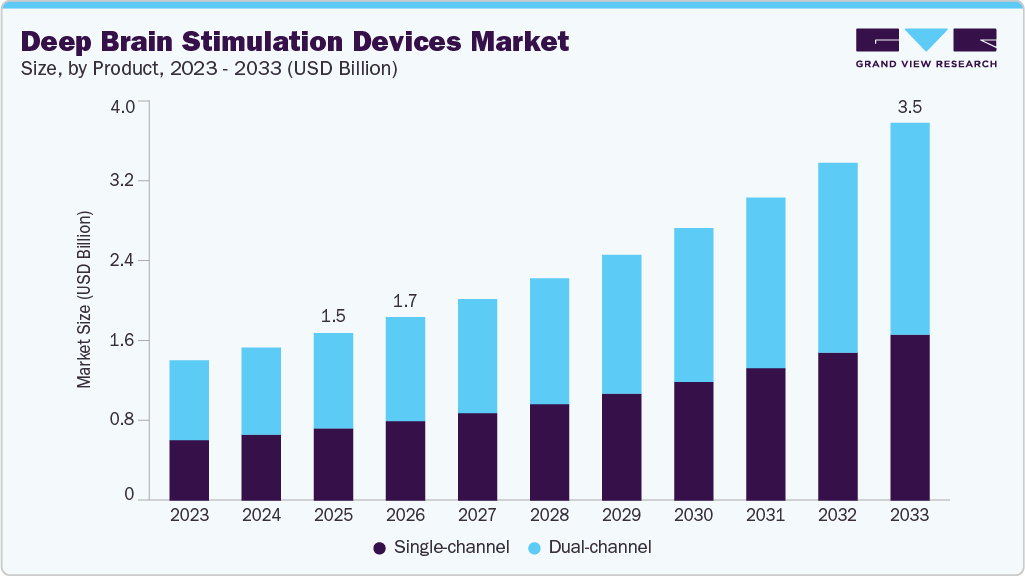
The global deep brain stimulation devices market size was estimated at USD 1.54 billion in 2025 and is projected to reach USD 3.47 billion by 2033, growing at a CAGR of 10.88% from 2026 to 2033. The major factors driving the market growth are rising prevalence of neurological disorders such as Parkinson’s disease and epilepsy, introduction of technologically advanced products, escalating product demand as add-on therapy, and the growing demand for minimally invasive procedures.
Key Market Trends & Insights
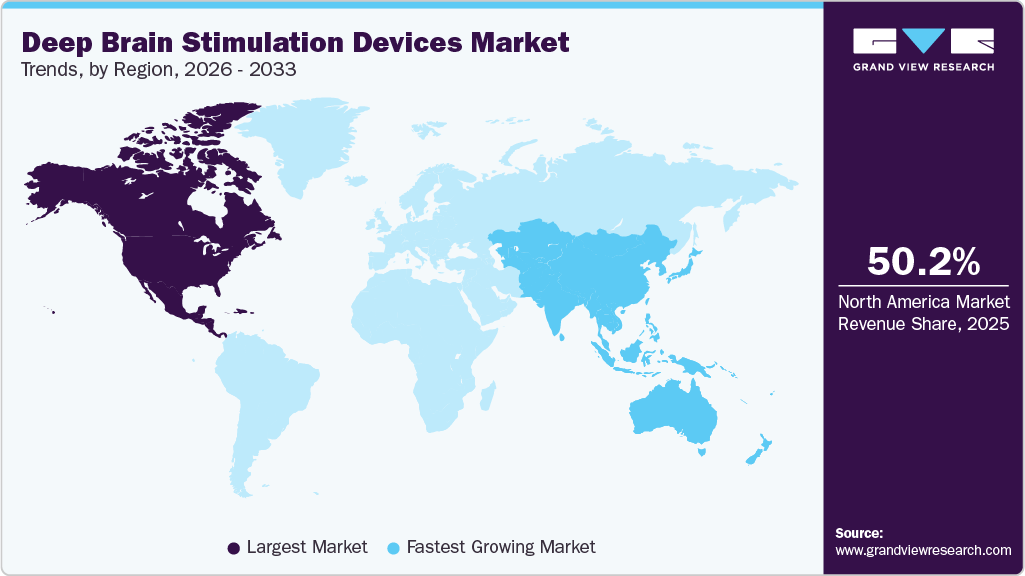
- North America dominated the deep brain stimulation devices market with a share of 50.21% in 2025.
- The deep brain stimulation devices market in U.S. is expected to dominate the market over the forecast period.
- Based on product, the dual-channel segment is dominating the market in 2025.
- Based on end-use, the hospitals segment dominated the overall deep brain stimulation devices market in 2025.
- Basis on application, the Parkinson’s disease segment is dominating the market in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.54 Billion
- 2033 Projected Market Size: USD 3.47 Billion
- CAGR (2026-2033): 10.88%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
The increasing prevalence of neurological disorders is a major driver of the deep brain stimulation devices market. Conditions such as Parkinson’s disease, epilepsy, depression, dystonia, and essential tremor are rising globally, creating a larger patient population requiring advanced treatment options.
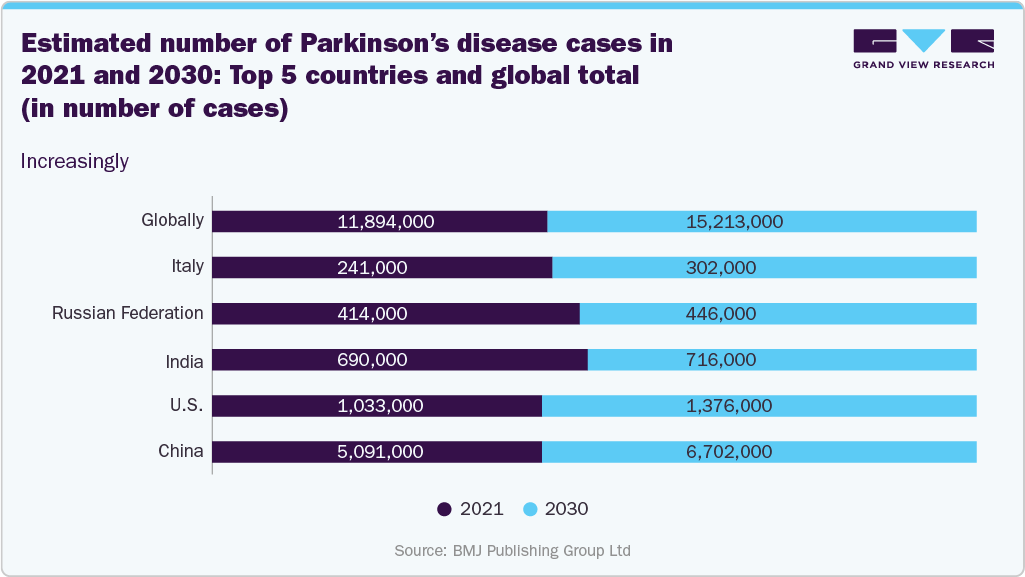
Increasingly
Source: BMJ Publishing Group Ltd
According to the World Health Organization (WHO), in August 2025, an estimated 5.7% of adults globally suffer from depression, further highlighting the growing burden of neurological and neuropsychiatric disorders and the need for advanced neuromodulation therapies.
Global Prevalence and Demographic Distribution of Depression (2024)
Statistic
Value
Global population experiencing depression
~4% of the total population
Adults with depression worldwide
5.7% of adults
Prevalence among men
4.60%
Prevalence among women
6.90%
Adults aged 70 years and older
5.90%
Total number of people with depression globally
~332 million people
Gender comparison
Depression is ~1.5 times more common in women than men
Pregnant and postpartum women affected
Over 10% globally
Source: WHO
The increasing preference for minimally invasive neurosurgical procedures is driving the growth of the deep-brain stimulation devices market. DBS involves targeted electrode implantation in the brain through small incisions, offering advantages such as reduced surgical trauma, faster recovery, and improved treatment precision. These benefits are encouraging greater adoption of DBS among patients and healthcare providers for the management of neurological disorders.
Furthermore, technological advancements in DBS technologies are anticipated to create growth opportunities in this market. These technological improvements include multi-target stimulation, robot-assisted implantation, improved microelectrode designs, rechargeable implantable pulse generators, and personalized directed programming. For instance, in January 2024, Abbott announced that it received approval from the U.S. Food and Drug Administration (FDA) to launch the Liberta RC DBS system. The system is world's smallest rechargeable DBS device with remote programming, to treat people with movement disorders. The Liberta RC DBS system also requires the fewest recharges of any FDA-approved DBS system, needing only 10 recharge sessions a year for most patients.
Market Concentration & Characteristics
The deep brain stimulation (DBS) devices market is characterized by rapid technological advancements, increasing clinical adoption, and a strong focus on treating neurological disorders such as Parkinson’s disease and essential tremor. The market is moderately consolidated, with major medical device companies investing in innovation, clinical research, and expansion of therapeutic indications to strengthen their market presence.
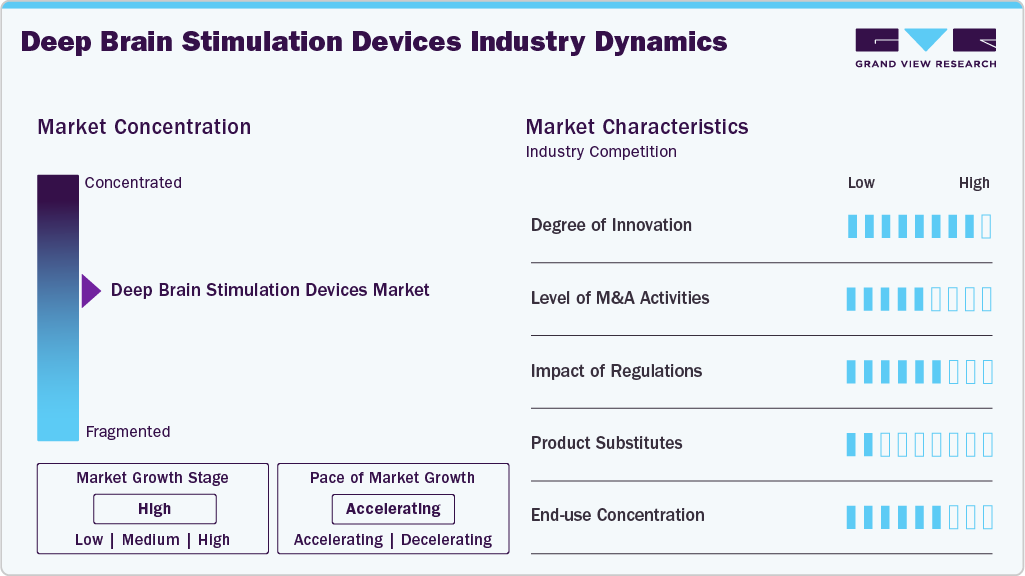
The deep brain stimulation (DBS) devices market demonstrates a high degree of innovation driven by advancements in neuromodulation technologies. Manufacturers are introducing features such as directional leads, rechargeable implantable pulse generators, adaptive stimulation, and wireless programming systems to improve treatment precision and patient outcomes. Continuous R&D and clinical trials are further expanding the therapeutic applications of DBS devices. In March 2025, Newronika announced that it had received CE Mark approval for its AlphaDBS device, a next-generation closed-loop deep brain stimulation (DBS) system that dynamically adjusts stimulation based on real-time brain signals. The approval allows the company to commercialize AlphaDBS in Europe, offering an advanced treatment option for patients with Parkinson’s disease and other neurological disorders.
"The CE Mark approval of AlphaDBS is a defining moment for Newronika and for the field of deep brain stimulation," said Lorenzo Rossi, CTO and Co-Founder of Newronika. "This certification validates our vision of bringing truly adaptive neuromodulation to patients. We are excited to bring this technology to market and to set a new standard in the treatment of Parkinson's disease."
Moreover, in October 2025, Medtronic plc announced that its BrainSense Adaptive Deep Brain Stimulation (aDBS) technology-recognized as the world’s first closed-loop DBS system for people with Parkinson’s disease-was included in TIME magazine’s annual list of Best Inventions, highlighting its breakthrough innovation in personalized neuromodulation therapy.
"Medtronic BrainSense Adaptive Deep Brain Stimulation marks a new era in neuromodulation and the use of therapeutic brain-computer interface technology to restore human health. We congratulate Medtronic on joining this year's esteemed list of Best Inventions." - TIME Editors, Best Inventions 2025
"We're honored that our BrainSense Adaptive DBS technology for people with Parkinson's is being recognized as one of the year's most important medical innovations," said Paolo Di Vincenzo, president of the Neuromodulation business, which is part of the Neuroscience portfolio at Medtronic. "This groundbreaking technology represents intentional innovation that responds to a patient's changing needs, equips clinicians with unparalleled insights, and sets a new standard for DBS therapy."
The deep brain stimulation (DBS) devices market shows a moderate level of merger and acquisition activity, with established medical device companies acquiring smaller neuromodulation and neurotechnology firms to strengthen their technological capabilities. These strategic deals focus on expanding product pipelines, enhancing research capabilities, and accelerating the development of advanced neuromodulation therapies.
Regulations have a significant impact on the deep brain stimulation (DBS) market, as these implantable neurostimulation systems must meet strict safety, clinical, and quality standards before commercialization. Regulatory approvals from agencies such as the FDA and CE authorities influence product development timelines, clinical trials, and market entry. Moreover, regulatory clearances help improve patient safety and increase physician confidence in adopting DBS technologies. For instance, in February 2025, Newronika announced that it received an Investigational Device Exemption (IDE) from the U.S. Food and Drug Administration (FDA) for its adaptive deep brain stimulation (DBS) system. The approval allows the company to initiate a pivotal clinical trial in the United States to evaluate the safety and effectiveness of the technology for patients with movement disorders, including Parkinson’s disease.
"Receiving an IDE from the FDA is a landmark achievement for Newronika," said Dr. Lorenzo Rossi, Co-Founder and CEO of Newronika. "It affirms the potential of our adaptive DBS system to fill an unmet need in the U.S. market, bringing personalized, real-time brain stimulation to patients who need it most. Our ultimate goal is to change the paradigm for managing neurological conditions worldwide, and this IDE moves us one step closer."
"The IDE approval for Newronika's adaptive DBS system represents an exciting advancement in the field of neuromodulation," said Prof. Dr. Med. Jens Volkmann, Principal Investigator of the study. "This pivotal trial will allow us to rigorously evaluate the benefits of real-time, patient-specific deep brain stimulation and its potential to surpass conventional DBS in symptom management. By leveraging adaptive neurotechnology, we aim to provide more precise, personalized, and effective treatments for Parkinson's patients, ultimately improving their quality of life."
The deep brain stimulation (DBS) devices market exhibits high-end-use concentration, with most procedures performed in specialized hospitals and neurosurgical centers equipped with advanced neuroimaging and surgical capabilities. These facilities have trained neurologists and neurosurgeons required for DBS implantation and post-operative programming, making them the primary end users of DBS systems.
Product Insights
The dual-channel segment dominated in 2025 owing to its higher adoption in surgical procedures. Dual-channel devices represent some of the most secure and efficient tools employed in surgical interventions. Consequently, the surge in disabling neurological conditions' prevalence, the escalating demand for Parkinson’s disease (PD) surgeries, and the rising adoption of dual-channel DBS devices by a growing number of medical facilities have all contributed to the remarkable growth of this segment. Furthermore, the segment's expansion is being fueled by ongoing technological advancements and the introduction of new products.
The single-channel deep brain stimulation device segment is expected to grow at the highest CAGR from 2026 to 2033. The consensus among healthcare experts is that single-channel DBS devices provide an extensive array of programming options for neurologists, consequently boosting their appeal and usage. Supporting this trend, the Parkinson's Foundation highlights the heightened vulnerability of the geriatric population to neurological ailments. As per the WHO report 2022, by 2050, the global population of individuals aged 60 and older will double to 2.1 billion, while those aged 80 and above will triple to reach 426 million by 2050, compared to 2020 figures. Given the increasing geriatric demographic, coupled with the rising patient awareness regarding neurological conditions, and the strong preference exhibited by healthcare professionals, the growth trajectory of this segment is expected to experience a significant upswing.
Application Insights
Parkinson’s disease dominated the deep brain stimulation devices market in 2025, primarily due to the high global prevalence of the disorder and the increasing use of DBS therapy for patients with advanced symptoms. DBS is widely adopted for Parkinson’s disease as it helps reduce motor complications and improves quality of life when medications become less effective. In addition, growing clinical evidence and physician preference for DBS in managing Parkinson’s symptoms have further strengthened the market. According to the Parkinson’s Foundation, the number of people living with Parkinson’s disease is projected to increase to around 1.2 million by 2030. For instance, in March 2023, researchers at Michigan Technological University embarked on a groundbreaking initiative, leveraging neuromorphic computing to enhance both the efficacy and energy efficiency of deep brain stimulation systems utilized in the treatment of Parkinson's disease. This innovative approach is poised to significantly propel the growth of this segment during the forecast period.
Epilepsy segment is expected to grow at the highest CAGR from 2026 to 2033. The increasing awareness of advanced epilepsy treatment options, combined with the ongoing development of healthcare infrastructure is expected to increase the growth of the segment in near future. According to the World Health Organization (WHO), in February 2024, around 50 million people worldwide are living with epilepsy, making it one of the most common neurological disorders globally. The organization also reports that nearly 80% of people with epilepsy reside in low- and middle-income countries, highlighting a significant global disease burden.
End-use Insights
Hospitals dominated the deep brain stimulation devices market in 2025. The substantial growth witnessed in this segment can predominantly be attributed to the escalating number of DBS surgeries performed within hospital settings and the concurrent rise in the prevalence of conditions such as Parkinson's disease and essential tremor. In addition, the availability of technologically advanced DBS devices, coupled with favorable reimbursement policies, is poised to further propel the hospitals segment during the forecast period. A notable example of this is Medtronic, which offers comprehensive services aimed at securing and maintaining coverage, as well as facilitating payment for a range of DBS devices.
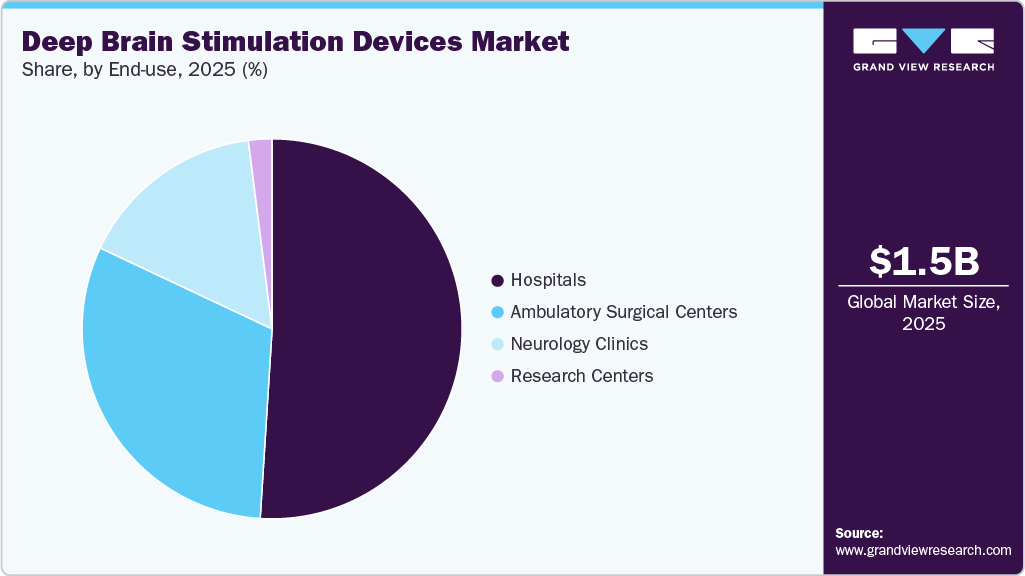
Ambulatory Surgery Centers (ASCs) segment expected to grow at a highest CAGR from 2026 to 2033. This is due to lower cost of procedures as compared to hospitals, convenient access for patient care, reduced waiting time, and low infection rate as compared to neurology clinics and hospitals. Furthermore, according to a study conducted by Advancing Surgical Care, 92% of patients were satisfied with the medical care and service provided in the ASCs, thus boosting the segment growth. In addition, most of the neurosurgeries can now be performed at ASCs, as surgical procedures undertaken here are advanced and less invasive. Thus, growth of the segment can majorly be attributed to shorter procedure time and ongoing advancements in minimally invasive surgical techniques.
Regional Insights
North America dominated the deep brain stimulation devices market with a share of 50.21% in 2025 owing to the increase in government funding and initiatives for raising awareness about PD is expected to drive the demand for deep brain stimulators. Furthermore, presence of major competitors, availability of sophisticated healthcare infrastructure, and supportive government initiatives are also responsible for the market growth in this region. For instance, the National Institute of Neurological Disorder and Stroke (NINDS) supports research activities related to DBS for determining its safety and effectiveness as the treatment of PD.
U.S. Deep Brain Stimulation Devices Market Trends
The deep brain stimulation devices market in U.S. is expected to dominate the market over the forecast period driven by the increasing prevalence of neurological disorders such as Parkinson's disease, dystonia, and essential tremors and growing number of clinical trials for treatment of psychiatric disorders. Advancements in DBS technology, such as enhancements in lead design, implantable pulse generators, and remote monitoring capabilities, are improving the effectiveness and safety of the DBS therapy in the U.S. According to 2022 Parkinson’s Foundation study, around one million people in the U.S. are living with Parkinson’s disease which is expected to reach nearly 1.2 million by 2030. Furthermore, high cost of DBS therapy and complexity and potential risks associated with DBS implantation is expected to hinder the growth of DBS devices market in the U.S.
Europe Deep Brain Stimulation Devices Market Trends
The deep brain stimulation devices market in Europe is expected to grow significantly over the forecast period. This growth can be attributed to the rising geriatric population and growing advancements in the deep brain stimulation devices market. Moreover, the region's advanced healthcare infrastructure, coupled with a focus on minimally invasive medical interventions, fosters the adoption of Deep Brain Stimulation Devices.
The deep brain stimulation devices market in the UK is expected to grow moderately over the forecast period.The increasing government spending on mental health services with about 14% of local NHS funding allocations is one of the primary driving factors for DBS devices market growth in UK. The NHS spent over USD 11.0 Billion in 2022/23 in England according to Mental Health Statistics report.
France deep brain stimulation devices market is expected to grow over the forecast period owing to rising prevalence of essential tremor and Parkinson’s disease and growing government support for the clinical research on neurological disorders.
Deep brain stimulation devices market in Germany is witnessing a steady growth owing to increasing aging population and growing prevalence of neurological diseases. Germany is a significant market for DBS devices, with a large and growing population of patients suffering from neurological disorders. The country has a well-established healthcare system and a high demand for innovative and effective treatments.Further,increasing awareness of DBS therapy among patients and healthcare providers is driving the growth of the DBS devices market in Germany. DBS therapy is becoming popular as a treatment option for various neurological disorders, due to its high effectiveness. In addition, growing initiatives taken by German to promote the use of minimally invasive procedures, is also further boosting the demand for DBS devices.
Asia Pacific Deep Brain Stimulation Devices Market Trends
Asia Pacific is expected to be the fastest growing region over the forecast period. This is attributed to the rising prevalence of neurodegenerative disorders coupled with unmet demand for effective and long-term solutions. Rising awareness about neurological disease treatment options and improvements in clinical development framework of emerging economies are expected to drive the market growth in this region. Moreover, presence of high growth opportunities in developing countries such as Japan, China, and India are likely to contribute to market growth. Furthermore, establishment of organizations such as the Asia Pacific Centre for Neuromodulation (APCN), which is founded for conducting research and promoting awareness about associated benefits of deep brain stimulation, is anticipated to boost regional growth.
The deep brain stimulation devices market in India is propelled by factors such as the country’s growing healthcare sector, increasing awareness about neurological disorders, and rising demand for minimally invasive treatments have created a significant opportunity for the DBS devices market in India. The low cost of DBS therapy in India as compared to western countries such as U.S. and UK are also fostering the growth of DBS devices market in India. The total price of DBS therapy is 60-90% cheaper than in western countries.
Japan deep brain stimulation devices market in is witnessing a significant growth, driven by the increasing demand for innovative treatments for neurological disorders. In Japan, DBS is approved for the treatment of Parkinson's disease, and the market has been growing steadily over the past few years. The country's aging population, combined with the increasing incidence of neurological disorders, is driving the demand for DBS devices. According to the WHO data published in 2020, deaths due to Parkinson’s disease in Japan reached about 14,007 or 1.27% of total deaths. The Japanese Government is also implementing several initiatives to support the development of DBS devices in Japan which is expected to fuel the growth of DBS devices market during the forecast period.
Latin America Deep Brain Stimulation Devices Market Trends
The DBS devices market in Latin America is witnessing a significant growth, driven by increasing neurological disorders prevalence, technological advancements, and rising awareness of advanced therapeutic options. The Latin American healthcare landscape is undergoing transformation, marked by the expansion of healthcare infrastructure and improvements in medical technology. Countries such as Brazil and Argentina are at the forefront of this expansion, witnessing enhanced access to advanced neurological treatments, including DBS. The increasing prevalence of neurological disorders in these nations, exacerbated by urbanization and aging populations, propels the demand for effective treatment options. According to various health reports, disorders such as Parkinson's disease are anticipated to rise due to demographic shifts, thereby boosting the DBS market.
Middle East and Africa Deep Brain Stimulation Devices Market Trends
The DBS devices market in the Middle East and Africa is primarily driven by the increasing demand for innovative treatments for neurological disorders and high incidence of neurological disorders. The region is also home to several emerging economies, which are investing heavily in healthcare infrastructure which is creating a growth opportunity for DBS devices, as well as other medical devices and treatments in the region.
Saudi Arabia is a major hub for healthcare in the Middle East, with a growing population of nearly 36.9 million people in 2023 according to Worldometer data. The country has a well-established healthcare system, with a number of modern hospitals and medical facilities. The increasing prevalence of parkinson's disease, is driving demand for DBS devices. The Saudi Arabian government is implementing initiatives to promote the development of medical devices in the country. For example, the Saudi Arabian government has established a number of initiatives to support the development of medical devices in the country, including the "National Program for Medical Devices" and the "Saudi Arabian Medical Device Regulatory Authority".The Saudi Arabia market for medical equipment has an estimated value of over $2 billion and is growing annually at about 10%.Saudi Arabia is also pursuing privatization initiatives, Currently Saudi Arabian government spending accounts for over 60% of the country’s healthcare expenditure.
Key Deep Brain Stimulation Devices Company Insights
The deep brain stimulation devices market is dominated by a few established medical device companies, including Medtronic, Abbott, and Boston Scientific, which maintain strong positions through advanced neuromodulation technologies and broad product portfolios. Market competition is driven by continuous product innovation, clinical research, and the expansion of therapeutic applications for neurological disorders.
Key Deep Brain Stimulation Devices Companies:
The following key companies have been profiled for this study on the deep brain stimulation devices market
- Medtronic
- Boston Scientific Corporation
- Abbott
- Aleva Neurotherapeutics
- Newronika S.p.A.
- Beijing PINS Medical Co., Ltd.
- SceneRay Corporation Ltd
Recent Developments
-
In February 2025, Medtronic plc announced that the U.S. Food and Drug Administration (FDA) approved its BrainSense Adaptive deep brain stimulation (aDBS) and BrainSense Electrode Identifier (EI) technologies. These innovations are designed to support personalized DBS therapy for the approximately one million people diagnosed with Parkinson’s disease in the United States.
-
In March 2025, Newronika, a company specializing in neuromodulation and adaptive deep brain stimulation technologies, announced that it received CE Mark approval for its AlphaDBS device. The next-generation closed-loop DBS system dynamically adjusts stimulation based on real-time brain signals, enabling the company to commercialize AlphaDBS in Europe and provide an advanced treatment option for patients with Parkinson’s disease and other neurological disorders.
-
In September 2024, Abbott announced that it has launched a pivotal clinical trial named TRANSCEND study. This trial aims to assess the efficacy of the company’s DBS system in treating treatment-resistant depression (TRD), a challenging type of major depressive disorder.
-
In September 2024, Medtronic published the trial design and preliminary findings from the ADAPT-PD trial, which focuses on the adaptive DBS algorithm for personalized therapy in Parkinson’s disease.
-
In January 2024, Abbott received approval from the U.S. FDA to launch the Liberta RC DBS system which is world's smallest rechargeable DBS device with remote programming, to treat people living with movement disorders.
-
In January 2024, Medtronic received U.S. FDA approval of its Percept RC Deep Brain Stimulation (DBS) system. It is a rechargeable neurostimulator and is the latest innovation in the Medtronic Percept family, which includes the Percept PC neurostimulator, SenSight directional leads, and BrainSense technology.
Deep Brain Stimulation Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.68 billion
Revenue forecast in 2033
USD 3.47 billion
Growth rate
CAGR of 10.88% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends, and volume analysis
Segments covered
Product, application, end-use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; Italy; France; Spain; Russia; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
Medtronic; Boston Scientific Corporation; Abbott; Aleva Neurotherapeutics; Newronika S.p.A.; Beijing PINS Medical Co., Ltd.; SceneRay Corporation Ltd
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Deep Brain Stimulation Devices Market Report Segmentation
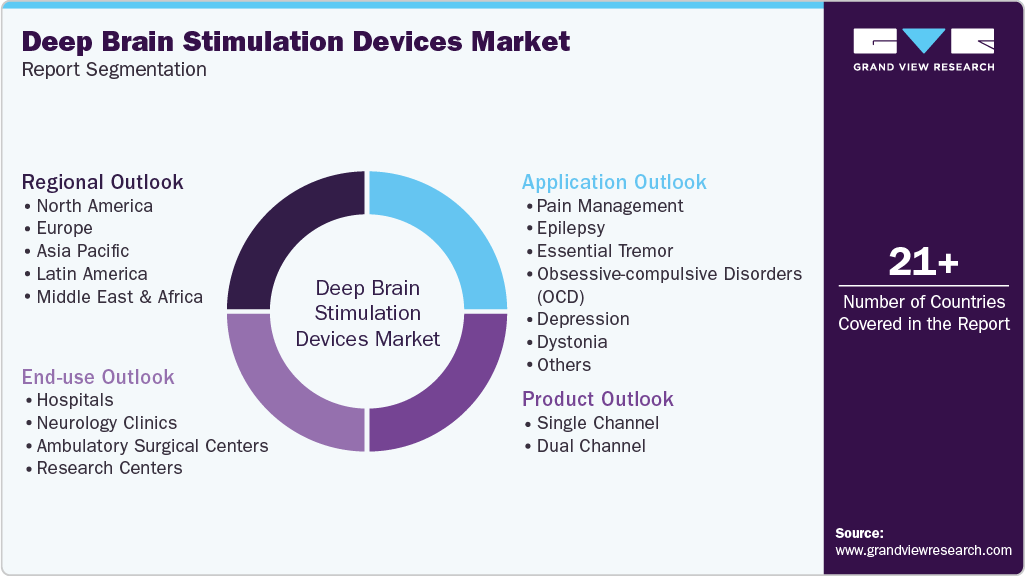
This report forecasts revenue growth at global, regional, and country levels and provides an analysis on the latest industry trends in each of the sub-segments from 2021 to 2033. For the purpose of this study, Grand View Research has segmented the global deep brain stimulation devices market report on the basis of product, application, end-use and region:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Single Channel
-
Dual Channel
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Pain Management
-
Epilepsy
-
Essential Tremor
-
Obsessive-compulsive Disorders (OCD)
-
Depression
-
Dystonia
-
Parkinson’s Disease
-
Others
-
-
End-use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Neurology Clinics
-
Ambulatory Surgical Centers
-
Research Centers
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some of the key players operating in the deep brain stimulation devices market include Medtronic, Boston Scientific Corporation, Abbott, and Aleva Neurotherapeutics
b. Key factors that are driving the deep brain stimulation devices market growth include the increasing incidence of neurological diseases, preference for a targeted approach, and technological advancements.
b. The global deep brain stimulation devices market size was estimated at USD 1.54 billion in 2025 and is expected to reach USD 1.68 billion in 2026.
b. The global deep brain stimulation devices market is expected to grow at a compound annual growth rate of 10.88% from 2026 to 2033 to reach USD 3.47 billion by 2033.
b. North America dominated the deep brain stimulation devices market in 2025. This is attributable to the rising prevalence of movement and psychiatric disorders, coupled with the increasing geriatric population in the region.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










