- Home
- »
- Medical Devices
- »
-
Diaphragm Pacing Therapy System Market Size Report, 2028GVR Report cover
![Diaphragm Pacing Therapy System Market Size, Share & Trends Report]()
Diaphragm Pacing Therapy System Market (2021 - 2028) Size, Share & Trends Analysis Report By Product, By Application (Spinal Cord Injury, Amyotrophic Lateral Sclerosis), By Region, And Segment Forecasts
- Report ID: GVR-4-68039-524-2
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2016 - 2019
- Forecast Period: 2021 - 2028
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Report Overview
The global diaphragm pacing therapy system market size was valued at USD 7.6 million in 2020 and is expected to expand at a compound annual growth rate (CAGR) of 4.6% from 2021 to 2028. The growing prevalence of disorders including sleep apnea, amyotrophic lateral sclerosis, and spinal cord injuries is expected to support the growth of the market. The diaphragm pacemaker is more accurate, comfortable compared to the mechanical ventilators and reduces the risk of upper airway infections such as ventilator-associated pneumonia. In addition, the favorable regulatory policies and approvals are fueling the adoption of the diaphragm pacing therapy systems. The COVID-19 pandemic propelled the demand owing to the upsurge in the ventilation requirements to provide respiratory care for the infected patients.
The diaphragm pacing therapy system reduces the dependence on mechanical ventilators and most patients prefer the diaphragm pacemaker as it facilitates normal breathing and better speech patterns, improved sense of smell, and ease of drinking and eating activities. In addition, the silent operation and functioning of a breathing pacemaker and the size of the device enabling mobility is further expected to boost the adoption of the diaphragm pacing therapy systems.
Product Insights
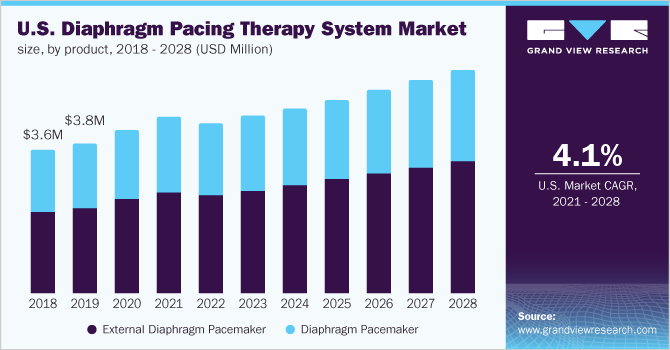
In 2020, the external diaphragm pacemaker segment dominated the market and accounted for the largest revenue share of 57.8%. Based on the product, the market for the diaphragm pacing therapy system is segmented into external diaphragm pacemaker and diaphragm pacemaker.
The external diaphragm pacemaker segment is gaining popularity owing to the rise in treatment of conditions such as cervical spinal cord injury and congenital central hypoventilation syndrome. The diaphragm pacemaker decreases the power outages concern and external power source requirement, and the convenience of movement and silent functioning further supports the market growth.
Application Insights
The spinal cord injury segment dominated the market and accounted for the largest revenue share of 45.5% in 2020 owing to the growing number of spinal cord injuries annually worldwide. According to the National Spinal Cord Injury Statistical Center, around 17,000 new spinal cord injury cases are reported every year in the U.S.
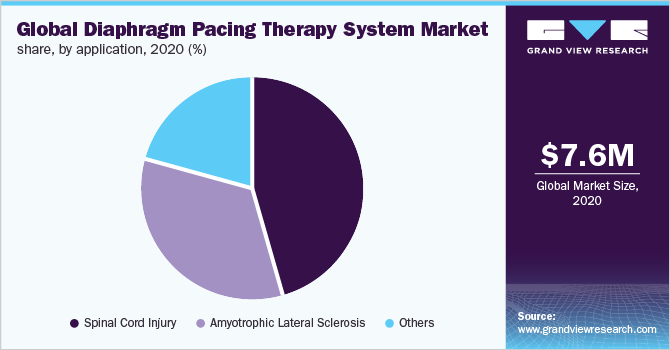
Moreover, the upsurge in accidents and falls, and injury during sports/recreation activities boost the prevalence of spinal cord injury and the number of patients suffering resulting in the demand for diaphragm pacing therapy systems. Furthermore, the growing prevalence of amyotrophic lateral sclerosis is expected to support the market growth of the diaphragm pacing therapy system. Amyotrophic lateral sclerosis is most common in the geriatric population, and it is reported that around 5,000 new cases are diagnosed each year in the U.S.
Regional Insights
North America dominated the diaphragm pacing therapy system market and accounted for the largest revenue share of 59.7% in 2020. The market is expected to grow well in the region over the forecast period owing to the prevalence of breathing disorders including sleep apnea, amyotrophic lateral sclerosis, and spinal cord injuries in the region. Furthermore, the improving healthcare expenditure and the growing disposable incomes of patients are expected to contribute to market growth in the region.
In Asia Pacific, the market is expected to witness the fastest growth rate over the forecast period. This is owing to the rising prevalence of breathing disorders, improving healthcare expenditure, supportive government initiatives, growing focus on R&D activities, and technological development to improve the healthcare infrastructure.
Key Companies & Market Share Insights
Technological developments in the healthcare sector have led to further innovation and development by the market players to meet the growing demand. Moreover, the ongoing COVID-19 pandemic has resulted in the surge of critical patients requiring mechanical ventilation for a prolonged period. This has burdened the healthcare systems and ICU resources with an unprecedented demand for ventilators and the patients face the risk of developing ventilator-induced diaphragm dysfunction. Lungpacer Medical’s diaphragm pacing therapy system received the U.S. FDA Emergency Use Authorization in April 2020 for COVID-19 patients.
In addition, Synapse Biomedical received the Emergency Use Authorization by the U.S. FDA for its TransAeris diaphragm pacing therapy system. The TransAeris system received CE Mark approval in 2018 and is under clinical investigation in the U.S. Some of the prominent players in the diaphragm pacing therapy system market include:
-
Lungpacer Medical Inc.
-
Avery Biomedical Devices, Inc.
-
Synapse Biomedical
-
Atrotech
Diaphragm Pacing Therapy System Market Report Scope
Report Attribute
Details
Market size value in 2021
USD 8.4 million
Revenue forecast in 2028
USD 11.5 million
Growth rate
CAGR of 4.6% from 2021 to 2028
Base year for estimation
2020
Actual estimates/Historic data
2016 - 2019
Forecast period
2021 - 2028
Quantitative units
Revenue in USD Million and CAGR from 2021 to 2028
Report coverage
Revenue forecast, company share, competitive landscape, growth factors & trends
Segments covered
Product, application, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S., Canada, U.K., Germany, France, Italy, Spain, Japan; China, India, Australia, Brazil, Mexico, South Africa
Key Companies profiled
Lungpacer Medical Inc.; Avery Biomedical Devices, Inc.; Synapse Biomedical; Atrotech
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional, and segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Segments Covered in the Report
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2016 to 2028. For this study, Grand View Research has segmented the global diaphragm pacing therapy system market report based on product, application, and region:
-
Product Outlook (Revenue, USD Million, 2016 - 2028)
-
External Diaphragm Pacemaker
-
Diaphragm Pacemaker
-
-
Application Outlook (Revenue, USD Million, 2016 - 2028)
-
Spinal Cord Injury
-
Amyotrophic Lateral Sclerosis
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2016 - 2028)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
U.K.
-
Germany
-
France
-
Italy
-
Spain
-
-
Asia Pacific
-
China
-
India
-
Japan
-
Australia
-
-
Latin America
-
Brazil
-
Mexico
-
-
Middle East & Africa
-
South Africa
-
-
Frequently Asked Questions About This Report
b. The global diaphragm pacing therapy system market size was estimated at USD 7.6 million in 2020 and is expected to reach USD 8.4 million in 2021.
b. The global diaphragm pacing therapy system market is expected to grow at a compound annual growth rate of 4.6% from 2021 to 2028 to reach USD 11.5 million by 2028.
b. North America dominated the diaphragm pacing therapy system market with the highest share of 59.7%in 2020. This is attributable to technologically advanced healthcare infrastructure coupled with high disposable income and the presence of key market players.
b. Some key players operating in the diaphragm pacing therapy system market include Avery Biomedical Devices, Lungpacer Medical, Inc, Synapse Biomedical, and Atrotech.
b. Key factors driving the diaphragm pacing therapy system market growth include rising favorable advantages of DPTS such as reduction of the dependency on mechanical ventilators and improvement of speech and breathing in patients, advanced healthcare infrastructure coupled with high disposable income, and favorable government initiatives.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










