- Home
- »
- Medical Devices
- »
-
ECG Equipment Market Size, Share & Trends Report, 2030GVR Report cover
![ECG Equipment Market Size, Share & Trends Report]()
ECG Equipment Market (2024 - 2030) Size, Share & Trends Analysis Report By Product, By Lead (Single Lead ECG, 3 Lead ECG, 2 Lead ECG, 6 Lead ECG, 12-Lead ECG), By End Use (Hospitals), By Region, And Segment Forecasts
- Report ID: 978-1-68038-573-1
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2022
- Forecast Period: 2024 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
ECG Equipment Market Summary
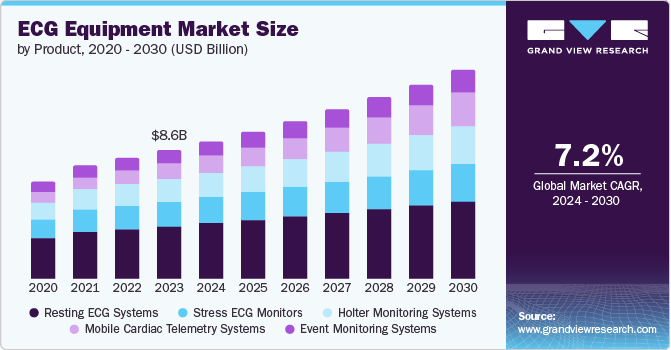
The global ECG equipment market size was estimated at USD 8.62 billion in 2023 and is projected to grow at a CAGR of 7.2% from 2024 to 2030. The increasing prevalence of cardiovascular diseases worldwide, coupled with the growing geriatric population, is one of the primary factors contributing to the market growth.
Key Market Trends & Insights
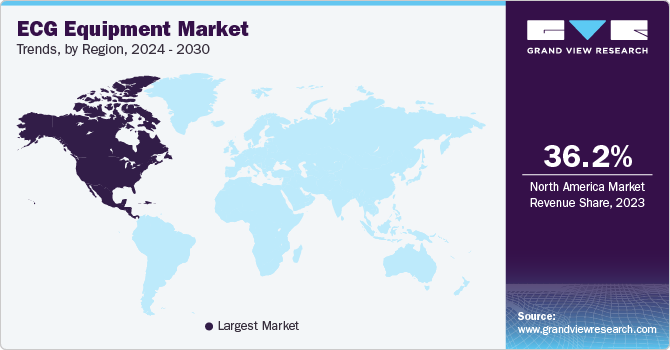
- North America dominated the global ECG equipment market with the largest revenue share in 2023.
- The ECG equipment market in the U.S. led the North American market and held the largest revenue share in 2023.
- By product, the Resting ECG systems segment led the market, holding the largest revenue share of 40.7% in 2023.
- By lead, the 12-lead ECG segment is expected to grow at the fastest CAGR of 8.8% from 2024 to 2030.
- By end use, the hospitals segment held the dominant position in the market and accounted for the leading revenue share in 2023.
Market Size & Forecast
- 2023 Market Size: USD 8.62 Billion
- 2030 Projected Market Size: USD 13.94 Billion
- CAGR (2024-2030): 8.8%
- North America: Largest market in 2023
In addition, technological advancements in ECG devices, such as the development of portable and handheld devices, are further propelling the market expansion. Increased spending on healthcare and a growing awareness of the importance of early detection of heart issues also contribute to this growth.
Emerging countries provide huge growth opportunities for cardiac diagnostics due to rising cases of cardiovascular diseases, rapid growth in the aging population, and ongoing investments in healthcare infrastructure. Moreover, growing awareness regarding preventive care and early diagnosis of CVD in emerging countries enhances the growth potential. Additionally, most of the emerging countries are one of the distinguishing destinations for medical tourism because of the low cost of treatment.
Governments worldwide have taken some initiatives to create awareness about the early treatment of cardiovascular diseases, improving healthcare sectors, which has led to a rise in partnerships & collaborations among manufacturers and private & public hospitals and cardiac centers. These are also essential factors in having significant market growth.
Product Insights
Resting ECG systems products dominated the market and accounted for a share of 40.7% in 2023. These devices are widely employed in managing cardiovascular disease due to their cost-effectiveness, space-efficient design, and user-friendly nature. As a result, these devices are consistently in demand and find application across various sectors, including private and public hospitals, dental clinics, and others. These are the driving forces behind the growing resting ECG systems market.
Mobile cardiac telemetry systems are expected to be the fastest-growing segment, with a CAGR of 12.6% from 2024 to 2030. The factors for this growth are its improved technology advancements, which makes it a better option to use for its features such as improved MCT systems, adhesive electrodes, portable recording units that transfer data to remote monitoring centers and healthcare facilities, wearable sensors, and its timely ability to recognize heart conditions time to time.s
Lead Insights
6-Lead ECG accounted for the largest market revenue share of 32.05% in 2023. This growth is driven by its advanced features, including the capability to interpret data on cardiac health issues and its effectiveness in managing and diagnosing heart rhythm abnormalities, particularly in detecting atrial fibrillation and tachycardia. The 6-Lead ECG is also suitable for use in remote areas or personal healthcare due to its ability to consistently and promptly detect cardiac issues.
The 12-lead ECG segment is projected to grow at a CAGR of 8.8% from 2024 to 2030. These devices are mainly employed by paramedics to diagnose Acute Myocardial infarction (AMI) and shorten treatment time. Several studies have examined the potential uses of these devices in diagnosing different arrhythmias and conduction disturbances through noninvasive techniques.
End Use Insights
The hospitals segment dominated the market in 2023. This dominance is largely due to the improved facilities available for patient care. The integration of advanced monitoring software and technological enhancements has significantly contributed to the treatment of patients requiring comprehensive cardiac care, positioning hospitals as the primary end-users of ECG equipment in this segment. The increased availability of ambulatory facilities, the adoption of Holter devices, and the accessibility of portable monitoring systems have also played key roles in propelling the growth of this segment.
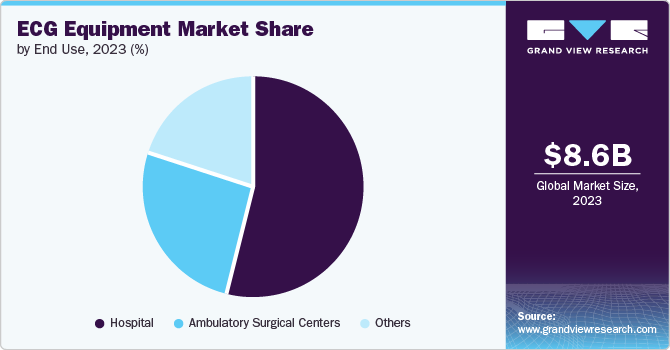
On the other hand, Ambulatory Surgical Centers (ASCs) are anticipated to witness the fastest growth, with a CAGR of 7.9% from 2024 to 2030. The rapid growth of ASCs can be attributed to the increasing preference for outpatient surgeries due to their cost-effectiveness and convenience. The integration of ECG equipment in ASCs aids in efficient patient monitoring before and after surgical procedures, enhancing patient safety and care. The projected high growth rate indicates a shifting trend towards ASCs, making them a vital end use segment in the ECG equipment market.
Regional Insights
North America ECG equipment market dominated the market in 2023. Rising cases of cardiovascular diseases with enhanced healthcare infrastructure and technological advancement are factors behind the growth of the market in the region.
U.S. ECG Equipment Market Trends
The U.S. ECG equipment market dominated the North American market with a revenue share of 82.1% in 2023 due to increasing cardiac disorders caused by unhealthy lifestyles. With technological advancements and experienced doctors, the U.S. has a large number of patients from other countries coming there for the treatment of cardiac disorders.
Europe ECG Equipment Market Trends
Europe was identified as a highly lucrative region in 2023. The market has experienced significant growth, largely attributed to the continuous advancement of technology in the region. This technological advancement has played a crucial role in driving the market's expansion and development.
The UK ECG equipment market is poised for rapid growth in the near future. This can be attributed to the increasing adoption of advanced healthcare delivery tools and technologies aimed at improving patient care and diagnostics within the country's healthcare system.
The ECG equipment market in Germany is growing significantly due to improved insurance policies for citizens. The statutory health system has made it mandatory to cover treatment, and private insurance assists with other expenses. This situation has made it favorable for patients to opt for treatment, which makes the market grow in the region.
Asia Pacific ECG Equipment Market Trends
The Asia Pacific market is anticipated to grow significantly over the forecast period. The rise in the region's economic prosperity and citizens' incomes has led to improved healthcare facilities and advanced medical technologies. This, in turn, has bolstered patients' access to treatment, consequently driving the demand for ECG equipment, thereby playing a crucial role in the market's expansion.
The ECG equipment market in Japan is experiencing significant growth, driven by major technological companies forming partnerships to develop advanced healthcare solutions. These alliances leverage technological advancements to enhance the country's healthcare systems.
Key ECG Equipment Company Insights
Some of the key companies in the ECG equipment market include Medtronic, GE Healthcare, Schiller AG, and many others. Organizations are focusing on increasing customer base to gain a competitive edge in the industry. Therefore, key players are taking several strategic initiatives, such as mergers and acquisitions, and partnerships with other major companies.
Key ECG Equipment Companies:
The following are the leading companies in the ECG equipment market. These companies collectively hold the largest market share and dictate industry trends.
- Medtronic
- Johnson & Johnson Services, Inc.
- GE HealthCare
- Koninklijke Philips N.V.
- Schiller AG
- Spacelabs Healthcare
- AliveCor, Inc.
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
- ACS Diagnostics.
- Allengers
- Hill-Rom Services, Inc. (Baxter)
- NIHON KOHDEN CORPORATION
Recent Developments
-
In February 2024, Thyracore acquires Think Health a key industry player in home ECG services. As a result, Thyrocare has now positioned itself as one of the leading providers of at-home healthcare services in India.
-
In January 2024, Omron Healthcare launched a portable ECG AliveCor's KardiaMobile in Italy and France to advance fibrillation awareness and stroke prevention in alignment with its ‘Going for zero’ vision.
ECG Equipment Market Report Scope
Report Attribute
Details
Market size value in 2024
USD 9.20 billion
Revenue forecast in 2030
USD 13.94 billion
Growth rate
CAGR of 7.2% from 2024 to 2030
Base year for estimation
2023
Historical data
2018 - 2022
Forecast period
2024 - 2030
Quantitative units
Revenue in USD billion and CAGR from 2024 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, lead, end use, region
Regional scope
North America, Europe, Asia Pacific, Latin America, MEA
Country scope
U.S., Canada, Mexico, Germany, UK, France, Italy, Spain, Denmark, Sweden, Norway, China, Japan, India, South Korea, Australia, Thailand, Brazil, Argentina, Saudi Arabia, UAE, Kuwait, South Africa
Key companies profiled
Medtronic; Johnson & Johnson Services, Inc.; GE HealthCare.; Koninklijke Philips N.V.; Schiller AG; Spacelabs Healthcare; AliveCor, Inc.; Shenzhen Mindray Bio-Medical Electronics Co., Ltd.; ACS Diagnostics.; Allengers; Hill-Rom Services, Inc. (Baxter); NIHON KOHDEN CORPORATION
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global ECG Equipment Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the ECG equipment market report based on product, lead, end use, and region:
-
Product Outlook (Revenue, USD Billion, 2018 - 2030)
-
Resting ECG Systems
-
Holter Monitoring Systems
-
Stress ECG Monitors
-
Event Monitoring Systems
-
Mobile Cardiac Telemetry Systems
-
-
Lead Outlook (Revenue, USD Billion, 2018 - 2030)
-
Single Lead ECG
-
3 Lead ECG
-
2 Lead ECG
-
6 Lead ECG
-
12-Lead ECG
-
-
End Use Outlook (Revenue, USD Billion, 2018 - 2030)
-
Hospital
-
Ambulatory Surgical Centers
-
Others
-
-
Regional Outlook (Revenue, USD Billion, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
UK
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
Saudi Arabia
-
UAE
-
South Africa
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










