- Home
- »
- Pharmaceuticals
- »
-
Gastrointestinal Therapeutics Market, Industry Report, 2033GVR Report cover
![Gastrointestinal Therapeutics Market Size, Share & Trends Report]()
Gastrointestinal Therapeutics Market (2026 - 2033) Size, Share & Trends Analysis Report By Drug Type (Branded, Generics), By Drug Class (Laxatives, Anti-emetics), By Route Of Administration, By Application, By Distribution Channel, By Region, And Segment Forecasts
- Report ID: GVR-2-68038-338-6
- Number of Report Pages: 140
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Gastrointestinal Therapeutics Market Summary
The global gastrointestinal therapeutics market size was estimated at USD 42.75 billion in 2025 and is projected to reach USD 51.45 billion by 2033, growing at a CAGR of 2.2% from 2026 to 2033. The industry is expanding due to the rising incidence of digestive disorders across both developed and emerging regions.
Key Market Trends & Insights
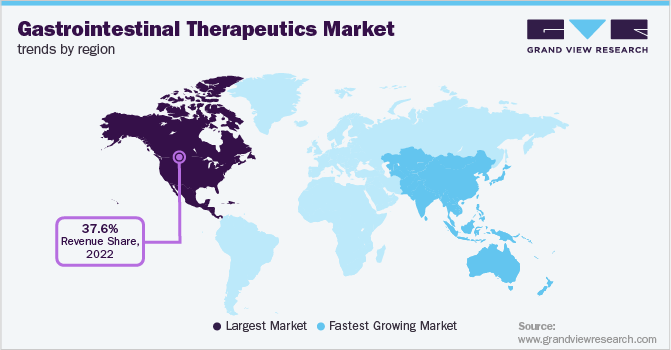
- North America gastrointestinal therapeutics market held the largest share of 37.96% of the global market in 2025.
- The gastrointestinal therapeutics industry in the U.S. is expected to grow significantly over the forecast period.
- By drug type, the branded segment held the highest market share of 66.68% in 2025.
- By drug class, the biologics/biosimilar segment held the highest market share in 2025.
- By route of administration, the injectables segment held the highest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 42.75 Billion
- 2033 Projected Market Size: USD 51.45 Billion
- CAGR (2026-2033): 2.2%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
The increasing prevalence of gastroesophageal reflux disease, irritable bowel syndrome, inflammatory bowel disease, and peptic ulcer disorders is strengthening the demand for long-term pharmacological management. Rapid urbanization, processed food consumption, irregular eating patterns, and elevated stress exposure are contributing to persistent gastrointestinal complications. A larger diagnosed patient population is seeking continuous symptom relief and disease control therapies. Higher consultation rates in primary and specialty care settings are further supporting prescription volumes.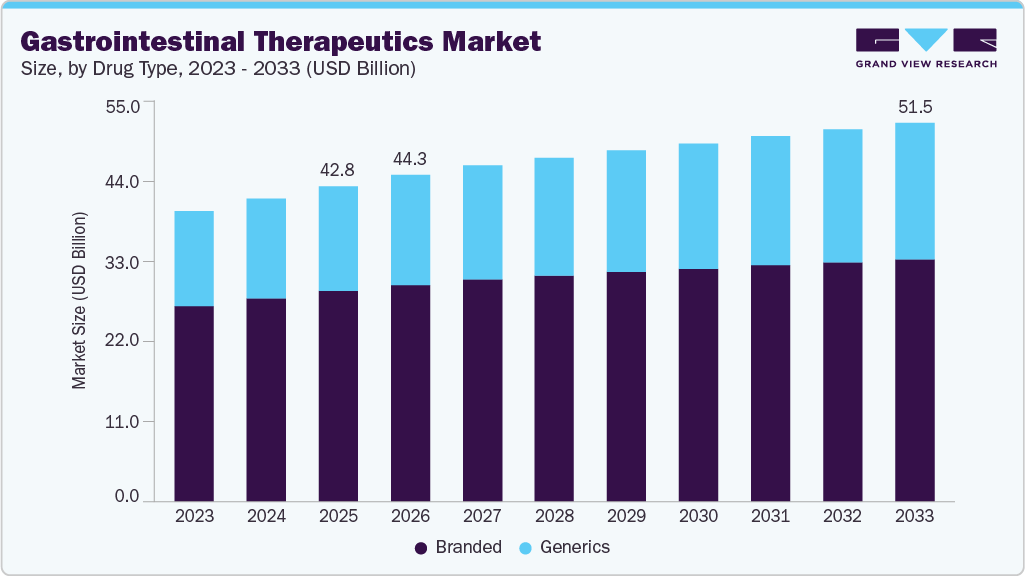
Growing awareness regarding early symptom recognition is encouraging timely treatment initiation. For instance, in January 2026, Current Gastroenterology Reports, a Springer publication, reviewed acute severe ulcerative colitis and reported that intravenous corticosteroid therapy, used for about 70 years remained associated with colectomy rates of 25% to 30%, while severe flares occurred in 25% to 50% of patients during their disease course and 20% presented with a severe flare initially. The analysis further noted cumulative colectomy rates rising from 19.9% after the first admission to 38.6% after four admissions, and first year mortality of 40% to 75% in the pre steroid era, which declined to below 1% in the modern era, yet increased to about 5% in older or refractory cases, underscoring significant unmet clinical need.
Advancements in pharmaceutical research and product innovation are significantly contributing to market growth. Development of biologics, targeted small molecules, and improved acid suppression therapies is enhancing clinical outcomes for chronic gastrointestinal conditions. Novel drug delivery systems are improving absorption profiles and reducing adverse effects, strengthening patient adherence. Precision based treatment strategies are supporting individualized therapeutic regimens in inflammatory bowel disease and related disorders.
Several companies are expanding development pipelines to introduce differentiated products with stronger efficacy data. For instance, in May 2024, Eli Lilly and Company reported results from the VIVID 1 Phase 3 study in moderately to severely active Crohn’s disease, where 54.1% of patients treated with mirikizumab achieved clinical remission at one year and 48.4% achieved endoscopic response at 52 weeks. Clinical remission was observed in 56.7% of biologic naïve patients and 51.2% of patients with previous biologic failure, while composite clinical and endoscopic response rates at Week 12 and Week 52 were 39.3% and 36.7% for mirikizumab versus 11.8% and 6.2% for placebo, and composite clinical response and remission by Crohn’s Disease Activity Index reached 47.3% and 43.4% compared with 26.5% and 12.4% for placebo.
Demographic and healthcare access trends are further reinforcing long-term market expansion. The global aging population is more vulnerable to chronic digestive disorders that require sustained treatment plans. Higher screening rates and improved diagnostic technologies are enabling earlier detection of gastrointestinal diseases. Expansion of retail pharmacy chains and e-commerce healthcare platforms is increasing product accessibility in urban and semi-urban regions. Broader availability of over-the-counter gastrointestinal medications is supporting self-management of mild to moderate conditions.
Rising healthcare expenditure in several economies is improving the affordability of advanced therapies. For instance, in January 2025, Reuters reported that the U.S. Food and Drug Administration approved expanded use of Eli Lilly’s Omvoh for adults with moderate to severe Crohn’s disease, in addition to its prior authorization for moderate to severe ulcerative colitis. Crohn’s disease was estimated to affect about 1,000,000 people in the U.S., and clinical data showed 53% remission at one year with Omvoh compared with 36% for placebo, while Eli Lilly had completed a USD 3.2 billion acquisition of Morphic Holding to strengthen its oral treatment pipeline.
Market Concentration & Characteristics
The gastrointestinal therapeutics industry demonstrates a high degree of innovation driven by advances in biologics, targeted small molecules, and microbiome-based therapies. Companies focus on improving treatment efficacy, safety profiles, and long-term disease control for chronic gastrointestinal conditions. Novel drug delivery systems and gut-selective mechanisms are gaining attention in research pipelines. Precision medicine approaches are supporting more individualized treatment strategies. Continuous clinical trials expand therapeutic indications across multiple disease segments. Innovation remains essential for competitive differentiation and lifecycle management.
Barriers to market entry are significant due to high research and development costs and complex clinical trial requirements. The need for extensive safety and efficacy data increases the time to market for new therapies. Established pharmaceutical companies benefit from strong brand recognition and physician trust. Intellectual property protections and patent portfolios create additional competitive challenges for new entrants. Manufacturing complexities associated with biologics require specialized infrastructure and expertise. These factors limit participation primarily to well-funded organizations.
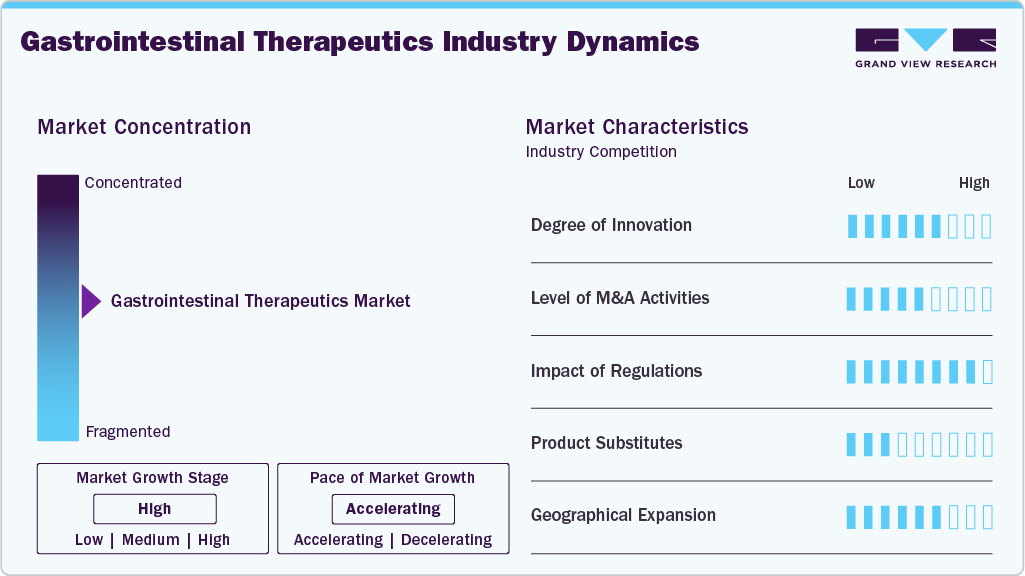
Regulatory frameworks strongly influence product approval timelines, commercialization strategies, and post-market monitoring requirements. Gastrointestinal therapies must meet strict safety and efficacy standards before receiving market authorization. Regulatory agencies require comprehensive clinical data to demonstrate therapeutic benefits across patient populations. Ongoing pharmacovigilance obligations add operational complexity for manufacturers. Compliance with evolving quality standards impacts production and supply chain processes. Regulatory oversight contributes to maintaining treatment safety and market credibility.
Product substitutes in the industry include over-the-counter medications, dietary interventions, and lifestyle modifications aimed at symptom management. Surgical procedures serve as alternatives for severe or treatment-resistant conditions. Nutraceuticals and probiotics are gaining attention among patients seeking complementary approaches. Generic medications provide cost-effective options that compete with branded therapies. Non-pharmacological management strategies such as dietary therapy also influence treatment choices. The availability of substitutes creates moderate competitive pressure within certain segments.
Geographical expansion remains a key characteristic as companies seek growth opportunities in emerging healthcare markets. Rising awareness of gastrointestinal disorders and improving healthcare access support market penetration across Asia Pacific, Latin America, and the Middle East. Partnerships with regional distributors and local manufacturing initiatives enhance product availability. Multinational firms adapt pricing and commercialization strategies to address diverse economic conditions. Expansion of specialty healthcare infrastructure increases diagnosis and treatment rates. Regional diversification helps companies strengthen global revenue streams.
Drug Type Insights
The branded segment dominated the market with the largest revenue share of 66.68% in 2025, driven by strong adoption of patented biologics and specialty therapies for chronic gastrointestinal conditions. Physicians frequently prescribe branded medications due to established clinical efficacy, safety data, and predictable therapeutic outcomes across complex disease profiles. Advanced drug formulations with improved pharmacokinetics and targeted mechanisms contribute to higher treatment success rates. Pharmaceutical companies invest significantly in commercialization strategies, physician education, and patient assistance programs that strengthen brand preference. Limited therapeutic substitutes for severe inflammatory disorders support sustained demand for branded therapies. For instance, in January 2025, Eli Lilly announced that the U.S. FDA approved Omvoh (mirikizumab-mrkz) for adults with moderately to severely active Crohn’s disease, supported by Phase III trials in which 53% of patients achieved clinical remission at one year compared with 36% receiving placebo, reinforcing branded biologic adoption. Higher pricing associated with innovative treatments further concentrates revenue within this segment.
The generics segment is projected to grow at the fastest CAGR of 3.2% over the forecast period, fueled by patent expirations of major gastrointestinal drugs and increasing demand for affordable treatment options. Generic medications provide cost-efficient solutions for long-term management of chronic digestive conditions across diverse patient populations. Healthcare providers recommend generics for maintenance therapy once disease stabilization is achieved. Expansion of manufacturing capacity among pharmaceutical companies improves supply consistency and global distribution. Competitive pricing encourages adoption across middle-income and price-sensitive markets. Regulatory approvals for bioequivalent formulations enhance physician confidence in generic substitution. Growing pharmacy penetration and large-scale procurement strategies further improve accessibility. Increasing awareness regarding therapeutic equivalence supports patient acceptance, contributing to strong growth potential for the generics segment worldwide.
Drug Class Insights
The biologics and biosimilar segment dominated the market with the largest revenue share of 44.70% in 2025, supported by strong clinical effectiveness in inflammatory bowel diseases and other severe gastrointestinal disorders. These therapies target specific immune pathways that promote sustained remission and limit disease progression in moderate to severe patients. Physicians increasingly prescribe biologics when conventional treatments deliver inadequate response, resulting in longer treatment durations and recurring revenue streams. Biosimilars are improving access by offering comparable efficacy at lower cost levels across key markets. For instance, in February 2026, Johnson and Johnson reported that in the QUASAR long-term extension study of TREMFYA guselkumab, 80.8% achieved clinical remission at Week 140, 78.6% achieved histo endoscopic mucosal improvement, 53.6% reached endoscopic remission, 89% completed treatment, and 87.5% maintained remission through Week 140.
The proton pump inhibitors segment is projected to grow at a CAGR of 2.2% over the forecast period, owing to the widespread prevalence of acid-related disorders such as gastroesophageal reflux disease and peptic ulcers. These medications remain a primary treatment option across hospitals, clinics, and outpatient settings due to established effectiveness in suppressing gastric acid secretion. Over-the-counter availability increases accessibility for patients seeking rapid symptom relief, while high prescription volumes from primary care physicians sustain consistent demand. For instance, in August 2025, Medicina published an evidence-based review reporting that more than 35 million PPI prescriptions were issued in the UK during 2022 to 2023, 8.6% of adults in the USA were using PPIs by 2017 to 2018, and 2.87 billion PPI tablets were sold in Germany in 2020 to 2021. The review further stated that up to 60% of prescriptions lacked a valid indication and one in four patients continued therapy beyond one year without reassessment, highlighting extensive utilization and long-term safety concerns.
Route of Administration Insights
The injectables segment dominated the market with the largest revenue share of 52.43% in 2025, due to increasing utilization of biologic therapies administered through intravenous and subcutaneous routes for severe gastrointestinal diseases. Injectable formulations provide targeted therapeutic action and improved bioavailability in patients with moderate to severe inflammatory conditions. Healthcare professionals often prefer controlled administration in clinical settings to ensure accurate dosing and treatment monitoring. Long-acting injectable drugs reduce dosing frequency, improving patient adherence to therapy schedules. Specialty infusion centers and hospital facilities support the delivery of advanced injectable treatments. Higher pricing associated with biologic injectables contributes significantly to revenue generation. Continuous innovation in self-injection devices further enhances patient convenience. Rising adoption of immunotherapy-based treatments continues to strengthen the dominance of injectable administration within gastrointestinal therapeutics.
The others segment is projected to grow at a CAGR of 2.1% over the forecast period, owing to increasing adoption of oral medications, topical therapies, and emerging drug delivery technologies for gastrointestinal management. Oral administration remains convenient for long-term treatment of chronic conditions, allowing patients to manage therapy independently at home. Advances in formulation science improve drug absorption, stability, and therapeutic effectiveness. Non-invasive delivery options enhance patient comfort and treatment compliance across diverse age groups. Expansion of outpatient care models supports preference for self-administered therapies. Pharmaceutical companies are investing in innovative delivery platforms such as delayed-release capsules and targeted intestinal delivery systems. Increasing patient preference for convenient treatment methods continues to support growth. Broader availability of oral and alternative formulations strengthens the expansion of this segment globally.
Application Insights
The Crohn’s disease segment dominated the market with the largest revenue share of 33.54% in 2025, supported by rising global prevalence and strong demand for advanced biologic therapies required for long-term disease control. Chronic progression requires continuous medication to prevent complications and maintain remission, while frequent flare episodes drive repeated therapeutic intervention and higher healthcare utilization. For instance, in December 2025, StatPearls Publishing reported that ileal, ileocolonic, and colonic involvement occurred in roughly equal proportions, with disease migration in 6% to 14% of patients, 19% presenting with stricturing or fistulizing disease within 90 days, and about 50% developing complications within 20 years. Noncaseating granulomas were observed in up to 33% of patients; the incidence ranged from 5 to over 12 per 100,000 person-years. 46.6% required surgery at 10 years, and 90% experienced postsurgical endoscopic recurrence, reinforcing sustained demand for advanced therapies.
The IBS segment is projected to grow at a CAGR of 3.7% over the forecast period, owing to increasing diagnosis of functional gastrointestinal disorders and rising patient awareness regarding symptom management. Lifestyle changes, dietary habits, and stress-related factors contribute to expanding patient populations experiencing IBS symptoms. Patients often seek pharmacological treatment to improve the quality of life and reduce symptom recurrence. Availability of multiple therapeutic options across prescription and over-the-counter categories supports adoption. Growing emphasis on personalized treatment approaches enhances clinical management strategies. Expansion of healthcare access enables more patients to receive medical consultation for functional disorders. Increasing demand for safe and effective therapies continues to drive product development. Continuous research into gut-brain interaction mechanisms supports the long-term growth potential of this application segment.
Distribution Channel Insights
The retail pharmacies segment dominated the market with the largest revenue share of 48.76% in 2025, due to extensive accessibility and convenience for patients requiring gastrointestinal medications. Retail outlets provide immediate availability of prescription drugs, over-the-counter treatments, and maintenance therapies for chronic digestive conditions. Pharmacist guidance supports appropriate medication selection and improves adherence to treatment regimens. Established supply chains ensure consistent product availability across urban and rural locations. Patients with long-term conditions frequently rely on retail pharmacies for recurring prescriptions and refills. Strong consumer trust in pharmacy services supports sustained utilization. Expansion of pharmacy chains enhances geographic coverage and product reach. Integration of digital prescription management within retail settings further improves patient convenience and contributes to segment dominance globally.
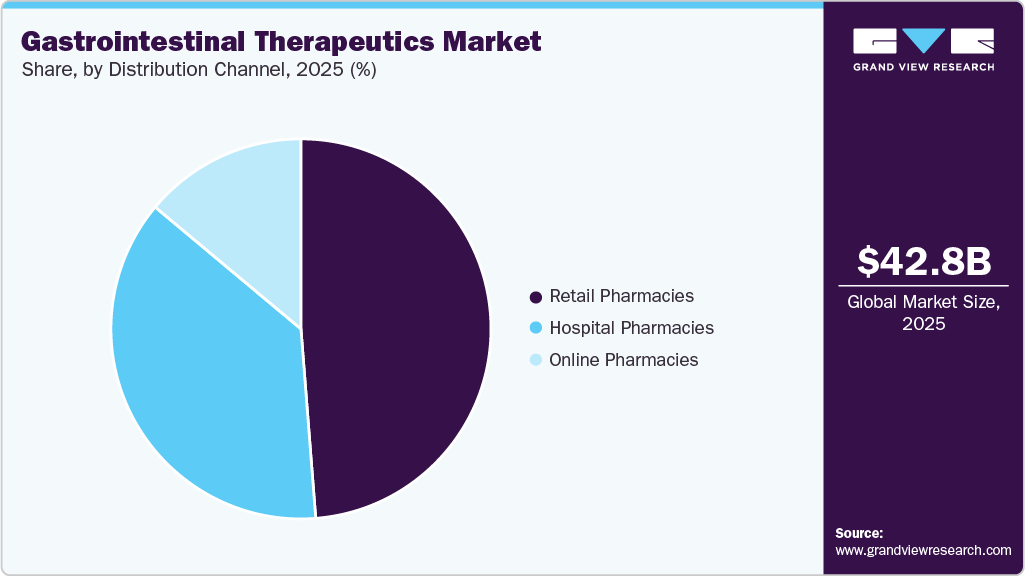
The online pharmacies segment is projected to grow at the fastest CAGR of 2.9% over the forecast period due to increasing digital adoption and preference for home delivery of medications. E-commerce platforms enable convenient ordering, price comparison, and automated refill services for chronic disease patients. Individuals managing gastrointestinal disorders benefit from discreet purchasing and doorstep delivery options. Integration of telehealth consultations supports online prescription fulfillment and treatment continuity. Competitive pricing and promotional offers attract cost-conscious consumers across multiple regions. Growing smartphone penetration and internet accessibility support the adoption of digital pharmacy services. Subscription-based medication delivery programs improve adherence and convenience. Expansion of digital healthcare ecosystems continues to accelerate growth within the online pharmacy distribution channel worldwide.
Regional Insights
North America gastrointestinal therapeutics market held the largest share of 37.96% in 2025, due to the high prevalence of chronic digestive disorders and strong adoption of advanced biologic therapies. The region benefits from a well-established healthcare infrastructure and early access to innovative treatment options. High diagnosis rates and patient awareness contribute to sustained demand for prescription therapies. Pharmaceutical companies maintain extensive commercialization networks and active clinical research programs across the region. Specialty care centers support long-term disease management for complex gastrointestinal conditions. Favorable reimbursement coverage further strengthens treatment accessibility.
U.S. Gastrointestinal Therapeutics Market Trends
The gastrointestinal therapeutics market in the U.S. dominates North America due to significant spending on specialty pharmaceuticals and advanced treatment modalities. A large patient population affected by inflammatory bowel disease, gastroesophageal reflux disease, and liver disorders supports market demand. Strong presence of leading pharmaceutical manufacturers accelerates product availability and innovation. Physicians widely adopt biologics and targeted therapies for chronic disease management. Ongoing clinical trials and rapid regulatory approvals contribute to market expansion. High patient awareness supports consistent therapy utilization.
Europe Gastrointestinal Therapeutics Market Trends
The gastrointestinal therapeutics market in Europe holds a substantial position in the gastrointestinal therapeutics market, supported by increasing disease burden and strong pharmaceutical research capabilities. The region demonstrates growing adoption of biologics and biosimilars across inflammatory bowel disease treatment. Healthcare systems emphasize early diagnosis and long-term disease monitoring, improving treatment uptake. Collaboration between research institutions and pharmaceutical companies enhances innovation pipelines. Expanding awareness of digestive health supports demand for prescription therapies. Market growth is further influenced by the presence of major pharmaceutical companies.
The UK gastrointestinal therapeutics market is expanding due to the rising incidence of chronic digestive conditions and increasing treatment awareness. Healthcare providers focus on early intervention and disease management strategies that improve patient outcomes. Adoption of biologics and advanced therapies continues to expand across specialty care centers. Research collaborations support the development of innovative treatment approaches for gastrointestinal disorders. Access to specialized gastroenterology services enhances diagnosis and treatment rates. The presence of clinical research initiatives contributes to market progression.
The gastrointestinal therapeutics market in Germany represents a key market in Europe due to advanced healthcare facilities and strong pharmaceutical manufacturing capabilities. The country demonstrates high adoption of innovative therapies for inflammatory bowel disease and related conditions. Physicians emphasize evidence-based treatment protocols that improve therapeutic outcomes. Increasing aging population contributes to a higher prevalence of gastrointestinal disorders. Well-established distribution channels support medication accessibility across healthcare settings. Continuous clinical research activities strengthen the national market landscape.
France gastrointestinal therapeutics market is supported by increasing diagnoses of digestive diseases and access to specialty care. The country shows growing adoption of biologic therapies for chronic inflammatory conditions. Healthcare providers prioritize long-term management strategies that enhance treatment adherence. Pharmaceutical companies expand their presence through product launches and collaborations with research institutions. Rising patient awareness of gastrointestinal health contributes to therapy demand. Strong clinical expertise supports the adoption of innovative treatment options.
Asia Pacific Gastrointestinal Therapeutics Market Trends
The gastrointestinal therapeutics market in Asia Pacific is expected to register the fastest CAGR of 3.9% over the forecast period due to large patient populations and improving healthcare access. Rapid urbanization and changing dietary patterns contribute to the increasing prevalence of digestive disorders. Expanding pharmaceutical manufacturing capabilities enhances product availability across the region. Healthcare providers are adopting advanced therapies at a growing pace. Rising awareness of gastrointestinal health supports earlier diagnosis and treatment initiation. Market participants are focusing on regional expansion to capture emerging opportunities.
Japan gastrointestinal therapeutics market represents an important market within Asia Pacific driven by advanced healthcare technology and aging demographics. The country shows strong demand for therapies targeting chronic gastrointestinal diseases and functional disorders. Physicians emphasize early diagnosis supported by advanced diagnostic tools. Pharmaceutical companies actively invest in research and development for innovative treatment approaches. High healthcare standards support the adoption of novel therapies. Continuous clinical studies contribute to therapeutic advancements in the region.
The gastrointestinal therapeutics market in China demonstrates significant growth potential due to increasing disease prevalence and expanding healthcare infrastructure. Rising urban populations and lifestyle changes contribute to digestive health concerns. Domestic pharmaceutical companies are strengthening production capabilities and expanding treatment portfolios. Healthcare providers are improving diagnostic capacity across urban and semi-urban areas. Growing patient awareness supports treatment adoption. Market expansion is further supported by increasing investment in clinical research activities.
Latin America Gastrointestinal Therapeutics Market Trends
The gastrointestinal therapeutics market in Latin America shows moderate growth, driven by improving healthcare access and rising awareness of digestive disorders. The increasing prevalence of gastrointestinal diseases supports demand for pharmaceutical treatments. Regional pharmaceutical companies are expanding distribution networks to improve medication availability. Healthcare providers are focusing on early detection and chronic disease management strategies. Adoption of generic medications supports cost-effective treatment access. Market growth is influenced by gradual improvements in healthcare infrastructure.
Brazil gastrointestinal therapeutics marketrepresents the largest market in Latin America due to its large population and increasing burden of gastrointestinal conditions. Growing awareness of digestive health supports diagnosis and treatment adoption. Pharmaceutical companies are expanding their presence through partnerships and product launches. Healthcare providers emphasize the management of chronic diseases such as inflammatory bowel disease and liver disorders. Expanding private healthcare services improves access to advanced therapies. Demand for affordable treatment options continues to shape the market landscape.
Middle East & Africa Gastrointestinal Therapeutics Market Trends
The gastrointestinal therapeutics market in the Middle East and Africa is supported by improving healthcare services and rising disease awareness. The increasing prevalence of digestive disorders contributes to treatment demand across urban populations. Pharmaceutical companies are expanding distribution partnerships to strengthen regional presence. Healthcare providers are enhancing diagnostic capabilities and specialty care services. Adoption of branded generics supports broader patient access to therapies. Market expansion remains influenced by ongoing healthcare development initiatives.
The Saudi Arabia gastrointestinal therapeutics marketis driven by the increasing prevalence of lifestyle-related gastrointestinal disorders and expanding healthcare facilities. Rising awareness of digestive health encourages patients to seek medical treatment. Healthcare providers are adopting advanced diagnostic technologies to improve disease detection. Pharmaceutical companies are strengthening market presence through product availability and partnerships. Growth in specialty care services supports long-term disease management. Demand for innovative therapies continues to increase across the country.
Key Gastrointestinal Therapeutics Companies Insights
Key players operating in the gastrointestinal therapeutics market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Gastrointestinal Therapeutics Companies:
The following key companies have been profiled for this study on the gastrointestinal therapeutics market.
- AbbVie Inc.
- AstraZeneca
- Salix Pharmaceuticals
- Takeda Pharmaceutical Company Limited
- Pfizer Inc.
- Bayer AG
- Abbott
- Janssen Pharmaceuticals NV
- Sun Pharmaceutical Industries Ltd.
- Cipla Inc.
- Gilead Sciences, Inc.
Recent Developments
-
In February 2026, Takeda reported Phase 3 results from the KEPLER study evaluating ENTYVIO vedolizumab in 120 pediatric patients aged 2 to 17 years with moderately to severely active ulcerative colitis. After a 14-week induction period, 93 responders entered maintenance treatment. Clinical remission was achieved in 34.7% at Week 14 and 47.3% at Week 54, while 29% maintained remission at both timepoints. Adverse events occurring in at least 10% included upper respiratory infection at 30%, worsening ulcerative colitis at 17.5%, and pyrexia at 12.5%.
-
In November 2025, AstraZeneca announced that the U.S. Food and Drug Administration approved IMFINZI durvalumab with FLOT chemotherapy for resectable Stages II, III, and IVA gastric and gastroesophageal junction cancers. The Phase III MATTERHORN trial showed a 29% reduction in risk of progression, recurrence, or death with a hazard ratio of 0.71 and a 95% confidence interval of 0.58 to 0.86. Event-free survival was 78.2% versus 74.0% at one year and 67.4% versus 58.5% at 24 months.
-
In June 2024, AbbVie announced that the U.S. Food and Drug Administration approved SKYRIZI risankizumab rzaa for adults with moderately to severely active ulcerative colitis, marking the first IL 23 specific inhibitor approved for both ulcerative colitis and Crohn’s disease. The decision was supported by two Phase 3 trials, including a 12-week induction study and a 52-week maintenance study. Patients received three 1200 mg induction doses every four weeks, followed by 180 mg or 360 mg maintenance doses every eight weeks.
Gastrointestinal Therapeutics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 44.31 billion
Revenue forecast in 2033
USD 51.45 billion
Growth rate
CAGR of 2.2% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion, and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Drug type, drug class, route of administration, application, distribution channel, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
AbbVie Inc.; AstraZeneca; Salix Pharmaceuticals; Takeda Pharmaceutical Company Limited; Pfizer Inc.; Bayer AG; Abbott; Janssen Pharmaceuticals NV; Sun Pharmaceutical Industries Ltd.; Cipla Inc.; Gilead Sciences, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Gastrointestinal Therapeutics Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global gastrointestinal therapeutics market report based on drug type, drug class, route of administration, application, distribution channel, and region:
-
Drug Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Branded
-
Generics
-
-
Drug Class Outlook (Revenue, USD Million, 2021 - 2033)
-
Aminosalicylates
-
Digestive Enzymes
-
Proton Pump Inhibitors
-
Laxatives
-
Anti-emetics
-
H2 Antagonists
-
Anti-diarrheals
-
Biologics/ Biosimilar
-
Others
-
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Oral
-
Injectables
-
Others
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Crohn's disease
-
Ulcerative colitis
-
GERD
-
IBS
-
Others
-
-
Distribution Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global gastrointestinal therapeutics market size was estimated at USD 42.75 billion in 2025 and is projected to reach USD 44.31 billion in 2026, supported by increasing diagnosis and treatment of chronic digestive disorders.
b. The global gastrointestinal therapeutics market is expected to grow at a compound annual growth rate (CAGR) of 2.2% from 2026 to 2033, reaching approximately USD 51.45 billion by 2033 due to rising demand for advanced therapies and long term disease management solutions.
b. Based on drug type, the branded segment dominated the gastrointestinal therapeutics market with the largest revenue share of 66.68% in 2025, driven by strong adoption of patented biologics and specialty therapies used for chronic gastrointestinal disorders such as Crohn’s disease and ulcerative colitis. Physicians often prefer branded therapies due to proven clinical efficacy, advanced formulations, and established safety profiles.
b. Key players operating in the market include AbbVie Inc., AstraZeneca, Salix Pharmaceuticals, Takeda Pharmaceutical Company Limited, Pfizer Inc., Bayer AG, Abbott, Janssen Pharmaceuticals NV, Sun Pharmaceutical Industries Ltd., Cipla Inc., and Gilead Sciences, Inc. These companies focus on biologics innovation, product portfolio expansion, and clinical research to strengthen their market presence.
b. Major factors driving market growth include the rising prevalence of gastrointestinal disorders such as GERD, inflammatory bowel disease, irritable bowel syndrome, and peptic ulcer disease. Increasing demand for biologics and targeted therapies, improved diagnostic rates, expanding healthcare access, and advancements in pharmaceutical research are further supporting market expansion.
b. North America held the largest share of the gastrointestinal therapeutics market in 2025, accounting for approximately 37.96% of the global market, supported by high prevalence of digestive disorders, advanced healthcare infrastructure, strong adoption of biologic therapies, and significant pharmaceutical research activities across the region.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










