- Home
- »
- Pharmaceuticals
- »
-
Glioblastoma Multiforme Treatment Market Size Report, 2033GVR Report cover
![Glioblastoma Multiforme Treatment Market Size, Share & Trends Report]()
Glioblastoma Multiforme Treatment Market (2026 - 2033) Size, Share & Trends Analysis Report By Treatment (Surgery, Radiation Therapy, Chemotherapy), By End Use (Hospitals, Ambulatory Surgery Centers), By Region, And Segment Forecasts
- Report ID: GVR-4-68039-527-3
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
Glioblastoma Multiforme Treatment Market Summary
The global glioblastoma multiforme treatment market size was estimated at USD 4.04 billion in 2025 and is projected to reach USD 7.87 billion by 2033, growing at a CAGR of 8.69% from 2026 to 2033. The rising prevalence of Glioblastoma Multiforme (GBM) and its significant clinical burden are critical drivers in the GBM treatment industry.
Key Market Trends & Insights
- North America glioblastoma multiforme treatment industry held the largest share of 40.01% of the global market in 2025.
- The glioblastoma multiforme treatment industry in the U.S. is expected to grow significantly over the forecast period.
- By treatment, the surgery segment held the largest market share of 32.52% in 2025.
- By end use, the hospitals segment held the largest market share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 4.04 Billion
- 2033 Projected Market Size: USD 7.87 Billion
- CAGR (2026-2033): 8.69%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
The expanding evidence base has reinforced temozolomide’s (TMZ) role as a cornerstone in glioblastoma multiforme (GBM) treatment. Several recent real-world studies and meta-analyses have shown that continuing TMZ therapy beyond the standard six cycles may provide additional survival benefits, particularly in patients with favorable molecular features such as MGMT promoter methylation or low Ki-67 expression. A meta-analysis published in Neuro-Oncology Advances in July 2023 reviewed outcomes from 2,578 patients across 16 nonrandomized studies and 5 randomized controlled trials. The analysis found that extended therapy was linked to a 28% reduction in disease progression and a 29% decrease in mortality risk, though most of these benefits were observed in retrospective cohorts. These findings highlight the need for further randomized investigations to establish the definitive value of prolonged adjuvant use. In addition, ongoing trials are evaluating TMZ in combination with immunotherapies and tumor-treating fields, supported by its tolerability and consistent clinical performance.
Cost-effectiveness and broad availability have also contributed to the widespread uptake of TMZ worldwide. The introduction of generic formulations has reduced treatment expenses, improving access without compromising therapeutic outcomes. Its favorable safety profile and established efficacy make it a preferred choice across diverse healthcare systems. A Phase IIa trial, published in March 2023 in the Journal of Clinical Oncology, assessed the SurVaxM vaccine alongside adjuvant TMZ in newly diagnosed GBM patients. Results demonstrated a median progression-free survival of 11.4 months and an overall survival of 25.9 months, with robust immune responses and no severe vaccine-related side effects, underscoring the potential of combination approaches. As emerging therapies evolve, many continue to build on TMZ as a foundation, reinforcing its critical role in the standard management of GBM.
The global uptake of tumor treating fields (TTF) continues to accelerate, supported by broader access and favorable reimbursement policies. Many healthcare systems have included TTF among reimbursable treatment options, reducing financial hurdles for patients who qualify. By late 2024, Novocure reported more than 4,000 patients worldwide actively using its Optune Gio TTF system, with strong adoption in regions such as the United States, Germany, France, and Japan. This rising user base reflects growing physician confidence and patient acceptance of the therapy across both advanced and emerging healthcare settings. The positive outcomes are also driving innovation, with ongoing efforts focused on technological upgrades and expanding its use in other tumor types. Collectively, these developments are positioning TTF as an integral element in the evolving management of glioblastoma multiforme.
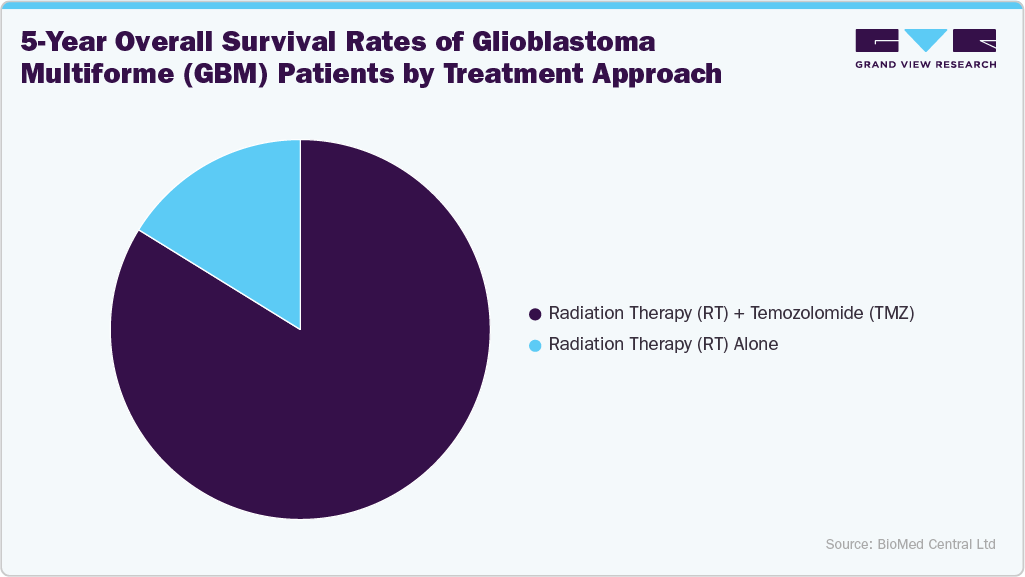
The five-year survival data shows a marked difference between treatment approaches, with radiation therapy (RT) alone resulting in a 16.2% survival rate compared to 83.8% when combined with temozolomide (TMZ). This demonstrates the significant clinical advantage of multimodal therapy, particularly TMZ’s role in enhancing outcomes for GBM patients. Such findings validate the continued reliance on TMZ as the backbone of GBM therapy and reinforce the demand for chemotherapy drugs in combination protocols. The evidence also supports healthcare providers’ preference for integrated treatment approaches, strengthening the adoption of combination therapies in clinical practice.
From a market perspective, this survival benefit will continue to drive investment in combination regimens and fuel demand for supportive therapeutics like TMZ. Pharmaceutical companies are expected to leverage these outcomes by developing next-generation therapies that complement or enhance TMZ’s effects, including immunotherapies and targeted agents. The higher survival outcomes with RT+TMZ will influence treatment guidelines, leading to broader reimbursement coverage and increased uptake across hospitals and cancer centers. This shift will not only sustain the dominance of TMZ in the GBM treatment market but also create opportunities for novel adjunctive therapies positioned alongside it.
Market Concentration & Characteristics
The glioblastoma multiforme treatment industry is characterized by a high degree of innovation due to the aggressive nature of the disease and poor survival outcomes with standard therapies. Research is increasingly focused on targeted therapies, tumor treating fields (TTF), and immuno-oncology approaches to improve efficacy beyond conventional surgery, radiation, and chemotherapy. Drug delivery innovations, such as biodegradable wafers and nanoparticle-based carriers, are also under development. The adoption of personalized medicine and biomarker-driven strategies further shapes innovation. Partnerships between biotech firms and large pharmaceutical players are accelerating novel pipeline progress. As a result, continuous R&D drives competitive differentiation in this market.
Barriers to entry in the GBM treatment market are significant, largely due to high clinical trial costs, stringent regulatory requirements, and the scientific complexity of brain tumor biology. Developing therapies requires advanced technology, significant capital, and long development timelines with uncertain success rates. Entrants face challenges from entrenched players with established drug portfolios, hospital relationships, and distribution networks. Intellectual property protections limit generic competition in certain segments. In addition, the small patient pool makes recouping R&D investments more difficult. Collectively, these barriers make entry into the GBM market highly restrictive for new firms.

Regulations play a pivotal role in shaping the GBM treatment market, given the high unmet need but also safety concerns around brain tumor therapies. Agencies like the FDA and EMA often provide accelerated approval pathways, orphan drug designations, and breakthrough therapy status to encourage innovation. However, strict safety and efficacy requirements can delay launches and increase trial costs. Post-marketing surveillance and pharmacovigilance obligations are particularly critical in oncology. Regulatory support for novel approaches, such as TTF therapy and targeted biologics is fostering growth. Thus, while regulations increase development costs, they also create opportunities for faster access in critical cases.
The availability of product substitutes in the GBM market is limited, as standard-of-care therapies like surgery, radiation, and chemotherapy remain the primary backbone of treatment. Targeted therapies and TTF serve as add-ons rather than full substitutes, given the aggressive tumor progression and resistance patterns. Palliative care and supportive therapies are alternative options but do not replace disease-modifying treatments. Experimental substitutes, such as immunotherapies, gene therapies, and cell-based treatments, are still under clinical evaluation. The lack of highly effective substitutes underscores the continued reliance on multimodal treatment approaches. This limited substitution heightens the value of novel breakthroughs.
Geographical expansion in the GBM treatment market is driven by growing healthcare infrastructure, rising cancer prevalence, and increasing access to oncology treatments in emerging markets. North America and Europe dominate due to strong regulatory frameworks, advanced research facilities, and reimbursement support. However, pharmaceutical companies are increasingly targeting Asia-Pacific and Latin America, where demand is rising alongside healthcare investments. Expanding clinical trial networks in these regions also supports earlier adoption of innovative therapies. Strategic partnerships with local distributors and hospitals are enhancing global reach. Consequently, geographical diversification is becoming a key strategy for sustained market growth.
Treatment Insights
The surgery segment dominated the market with the largest revenue share of 32.52% in 2025, as surgery remains the primary treatment option for GBM. Surgical resection helps in reducing tumor mass, which is critical for improving survival outcomes. Advancements in image-guided and minimally invasive procedures have enhanced the precision of tumor removal. The integration of intraoperative MRI and fluorescence-guided surgery has improved effectiveness in achieving maximum safe resection. A study published in Insights into Imaging in February 2025 found that tumor surface regularity, assessed via preoperative MRI, can predict overall survival and the benefit derived from gross total resection (GTR) in IDH-wildtype GBM patients. Patient preference for surgical removal of tumors at initial diagnosis further supports the segment’s strong position. The availability of skilled neurosurgeons in leading cancer care centers strengthens adoption rates. Growing investment in surgical infrastructure in specialized hospitals has reinforced the dominance of this segment.
The tumor treating field (TTF) therapy segment is projected to grow at the fastest CAGR of 11.59% over the forecast period, due to its non-invasive mechanism of action. TTF therapy disrupts cancer cell division using low-intensity electric fields, which improves progression-free survival. Increasing clinical evidence supporting improved outcomes in combination with chemotherapy is fueling adoption. According to a multicenter analysis published in August 2025, TTF in combination with temozolomide resulted in a median overall survival of 20.9 months, compared to 16.0 months in patients receiving temozolomide alone, while progression-free survival improved from 4.0 to 7.1 months in the TTF group. Rising awareness among patients about novel treatment options is creating strong growth potential. The compact and wearable design of TTF devices enhances patient compliance and quality of life. Favorable clinical trial results and rapid inclusion in treatment guidelines are expanding its market penetration. The shift toward innovative targeted therapies is accelerating demand for TTF therapy in GBM management.
End Use Insights
The hospitals segment dominated the market with the largest revenue share of 76.86% in 2025, which can be attributed to the concentration of advanced treatment infrastructure. Hospitals provide access to surgical facilities, radiation therapy units, and integrated oncology care under one roof. Multidisciplinary teams of neurosurgeons, oncologists, and radiologists deliver coordinated patient management. Availability of intensive care units supports complex surgical interventions and post-treatment monitoring. According to a study published in June 2025, fluorescence-guided surgery has resulted in higher gross total resection rates, minimizing the risk of residual tumor and improving post-surgical recovery. Hospitals also serve as primary centers for clinical trials of novel therapies, attracting a large patient pool. The presence of advanced diagnostic imaging tools enhances treatment planning and monitoring. This comprehensive ecosystem makes hospitals the preferred setting for GBM treatment.

The clinics/outpatient centers segment is projected to grow at the fastest CAGR of 10.04% over the forecast period, as treatment approaches shift toward patient convenience and cost efficiency. These centers are increasingly offering radiation therapy, chemotherapy, and TTF device management in outpatient settings. Rising demand for shorter hospital stays is driving patients toward ambulatory care options. The adoption of advanced imaging and diagnostic tools in clinics has improved precision in treatment monitoring. A study published in September 2024 in the European Journal of Cancer emphasizes that precision oncology can be delivered in a cost-effective manner, even for financially challenged health systems, through the integration of genomic profiling into clinical practice for more targeted and efficient use of healthcare resources. Outpatient centers reduce the economic burden on patients through lower operational costs compared to hospitals. The growing number of private specialty oncology clinics is expanding accessibility for GBM patients. Enhanced patient-centric services in outpatient care are fueling strong market growth for this segment.
Regional Insights
North America Glioblastoma Multiforme Treatment Market Trends
North America glioblastoma multiforme treatment industry held the largest share of the global market in 2025, accounting for 40.01% of revenue, due to advanced healthcare infrastructure and strong adoption of innovative oncology therapies. High prevalence of brain tumors and the availability of specialized neuro-oncology centers contribute to sustained demand. Leading pharmaceutical companies have established extensive distribution networks that ensure wider access to targeted and chemotherapy drugs. Strong clinical research activities in the region enhance the introduction of novel therapies, especially in immuno-oncology and tumor treating fields. Increasing awareness of early diagnosis and patient preference for advanced treatments further support market leadership. The region’s mature reimbursement systems also improve patient access to high-cost therapies, strengthening its position.

U.S. Glioblastoma Multiforme Treatment Market Trends
The U.S. glioblastoma multiforme treatment industry holds the largest share in the North American market owing to its concentration of leading cancer research institutes and a high number of clinical trials. Rising incidence of glioblastoma cases coupled with growing demand for precision medicine drives treatment uptake. The country hosts major pharmaceutical companies that invest heavily in new drug development and commercialization. High adoption of tumor treating field devices and targeted therapy reflects the country’s advanced clinical practices. A strong ecosystem of hospitals and outpatient centers ensures wide treatment accessibility. Increasing patient awareness of disease progression and available therapies accelerates demand in this market.
Europe Glioblastoma Multiforme Treatment Market Trends
Europe glioblastoma multiforme treatment industry holds a strong position in the global market with widespread adoption of chemotherapy, radiation, and surgery supported by strong clinical expertise. The region benefits from a high density of specialized oncology centers that provide integrated care. Pharmaceutical companies actively collaborate with European research institutes to accelerate therapeutic innovation. Patients in the region have increasing access to novel targeted therapies and tumor treating field technologies. Rising prevalence of neurological disorders and cancer supports continued demand for effective treatment. Continuous investments in cancer-focused clinical trials strengthen Europe’s role as a key regional hub.
The UK glioblastoma multiforme treatment industry is experiencing rising demand due to the presence of advanced medical facilities and skilled healthcare professionals. Increasing cases of glioblastoma create a sustained need for surgery, radiation therapy, and chemotherapy. The region has seen a growing uptake of targeted therapies such as bevacizumab, particularly in specialized cancer hospitals. Collaboration between local institutions and global pharmaceutical companies enhances access to innovative therapies. Patient awareness campaigns emphasize the importance of early detection and advanced treatment. Expansion of outpatient oncology centers supports a broader base of treated patients.
Germany glioblastoma multiforme treatment industry is a key contributor to the European market supported by its advanced healthcare infrastructure and strong pharmaceutical industry presence. The country has a high number of specialized oncology hospitals that provide multidisciplinary care. German research institutes are at the forefront of clinical studies in tumor treating field devices and novel biologics. Rising incidence of glioblastoma is driving higher demand for both standard and innovative therapies. The market is also influenced by a growing focus on personalized treatment approaches. Strong clinical expertise makes Germany a central hub for the development and adoption of advanced therapies.
France glioblastoma multiforme treatment industry contributes significantly to the European market due to the growing patient pool and strong presence of oncology-focused hospitals. The adoption of tumor treating fields and chemotherapy combinations is gaining momentum in the country. Local pharmaceutical collaborations with global players encourage the development of new therapeutic options. Patient awareness of advanced treatment approaches has increased, resulting in greater acceptance of targeted therapies. High availability of diagnostic services supports early identification of glioblastoma cases. The country’s skilled medical workforce enhances the quality of neuro-oncology care, supporting market growth.
Asia-Pacific Glioblastoma Multiforme Treatment Market Trends
Asia Pacific glioblastoma multiforme treatment industry is expected to register the fastest CAGR of 9.94% over the forecast period, supported by expanding healthcare infrastructure and a large patient base. Rising incidence of brain tumors, particularly among aging populations, fuels demand for advanced therapies. Increasing adoption of chemotherapy and radiation therapy across hospitals drives initial treatment uptake. Regional pharmaceutical companies are investing in generic and branded oncology drugs, boosting accessibility. The expansion of clinical trial networks is accelerating the introduction of novel treatments. Rising awareness of cancer treatment options is further driving strong growth momentum in this region.
Japan glioblastoma multiforme treatment industry has emerged as a key market within Asia Pacific due to its strong oncology infrastructure and high treatment adoption rates. The aging population is a major factor driving the increasing prevalence of glioblastoma in the country. Japanese pharmaceutical companies actively invest in oncology research, contributing to innovation in chemotherapy and targeted therapy. The market benefits from advanced diagnostic technologies that enable early disease identification. Hospitals and cancer centers in Japan are equipped with modern radiation and surgical facilities. Patient awareness of advanced treatments has led to increased uptake of tumor treating field therapy.
China glioblastoma multiforme treatment industry is experiencing rapid growth, fueled by rising cancer prevalence and increasing demand for advanced oncology care. Expanding hospital infrastructure supports higher adoption of chemotherapy, surgery, and radiation therapies. Local and multinational pharmaceutical companies are launching a wide range of oncology products, improving treatment accessibility. Large-scale clinical research initiatives are enhancing the availability of innovative therapies in the market. Urban centers in China are seeing strong demand for targeted therapy and tumor treating field devices. Growing awareness of brain tumor symptoms is boosting early diagnosis and treatment rates.
Latin America Glioblastoma Multiforme Treatment Market Trends
Latin America glioblastoma multiforme treatment industry is showing steady growth, due to increasing cancer incidence and improving access to oncology facilities. Brazil and Mexico are at the forefront of treatment adoption owing to expanding hospital networks. The region’s pharmaceutical sector is witnessing rising availability of chemotherapy drugs and generics. Patient preference for cost-effective therapies is shaping demand dynamics across different segments. Targeted therapy adoption is gradually increasing in specialized hospitals. Growing clinical research participation is further strengthening Latin America’s role in the global GBM treatment landscape.
Brazil glioblastoma multiforme treatment industry plays a major role in the Latin American market with its growing healthcare capacity and large patient pool. Rising cancer prevalence is creating greater demand for surgery, chemotherapy, and radiation therapy. Multinational pharmaceutical companies have established a strong presence in the country to improve product availability. Adoption of advanced oncology techniques such as targeted therapies is gradually increasing in urban hospitals. Expanding access to specialized oncology centers is enhancing treatment opportunities for patients. Growing participation in clinical trials supports the introduction of novel therapies in Brazil.
Middle East & Africa Glioblastoma Multiforme Treatment Market Trends
The MEA glioblastoma multiforme treatment industry is witnessing gradual progress, driven by rising cancer prevalence and an expanding healthcare sector. Urban centers in the Middle East, in particular, are investing in advanced surgical and radiation facilities. Increasing awareness of oncology care has led to higher treatment uptake in specialized hospitals. Pharmaceutical companies are expanding their presence to meet growing demand for chemotherapy and supportive care drugs. The region is also seeing a steady increase in adoption of targeted therapies in private hospitals. Overall, the MEA market presents emerging opportunities despite existing challenges in healthcare access.
Saudi Arabia glioblastoma multiforme treatment is emerging as a key market within MEA owing to its expanding oncology infrastructure. The country is witnessing a rising number of brain tumor cases that require advanced therapeutic intervention. Hospitals are increasingly equipped with modern radiation therapy and neurosurgery facilities. The market is experiencing higher uptake of chemotherapy and targeted therapy across major treatment centers. Collaborations between local healthcare providers and global pharmaceutical companies are improving drug availability. Growing awareness of cancer treatment options is encouraging patients to seek specialized neuro-oncology care in Saudi Arabia.
Key Glioblastoma Multiforme Treatment Company Insights
Merck & Co., Inc. and Amgen, Inc. are shaping the glioblastoma multiforme treatment industry with strong oncology portfolios and advancements in targeted therapies that aim to enhance treatment outcomes. F. Hoffmann-La Roche Ltd. and Pfizer Inc. continue to expand their influence through precision medicine platforms, clinical trials, and novel immuno-oncology strategies. Teva Pharmaceutical Industries Ltd. and Sun Pharmaceutical Industries Ltd. strengthen market accessibility with extensive generic oncology offerings and supportive care drugs. Arbor Pharmaceuticals, LLC and Amneal Pharmaceuticals are contributing through specialized product lines and distribution expansion across hospitals and outpatient centers. Karyopharm Therapeutics, Inc. is advancing its position with innovative small-molecule therapeutics addressing treatment resistance. Sumitomo Dainippon Pharma Oncology, Inc. (Boston Biomedical, Inc.) is driving clinical development with a focus on next-generation oncology research, further intensifying competition in this high-need market.
Key Glioblastoma Multiforme Treatment Companies:
The following are the leading companies in the glioblastoma multiforme treatment market. These companies collectively hold the largest market share and dictate industry trends.
- Merck & Co., Inc.
- Amgen, Inc.
- F. Hoffmann-La Roche Ltd.
- Pfizer Inc.
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Arbor Pharmaceuticals, LLC
- Amneal Pharmaceuticals
- Karyopharm Therapeutics, Inc.
- Sumitomo Dainippon Pharma Oncology, Inc. (Boston Biomedical, Inc.)
Recent Developments
-
In May 2025, Sumitomo Dainippon Pharma is progressing with Phase 1/2 clinical trials for DSP-0390, an emopamil-binding protein inhibitor, for the treatment of recurrent GBM. The drug targets key proteins involved in tumor cell survival and proliferation. The trials aim to assess DSP-0390's safety, tolerability, and efficacy in inhibiting GBM tumor growth, potentially providing a new therapeutic option for patients with recurrent high-grade gliomas.
-
In January 2025, Merck is conducting Phase II clinical trials for pembrolizumab (Keytruda) in patients with recurrent GBM. The trials are investigating the drug both as a monotherapy and in combination with the DNX-2401 oncolytic virus. This clinical program aims to assess the potential of pembrolizumab to enhance the immune response against GBM tumors, a challenging cancer type with limited treatment options, particularly for recurrent cases.
-
In January 2025, Pfizer’s Glasdegib, a Hedgehog pathway inhibitor, is in Phase II clinical trials for GBM. The drug targets the Hedgehog signaling pathway, which plays a crucial role in tumorigenesis and resistance to conventional therapies. The ongoing trials aim to evaluate the effectiveness of Glasdegib in GBM treatment and its potential to overcome treatment resistance and improve patient outcomes in recurrent cases of the disease.
Glioblastoma Multiforme Treatment Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 4.39 billion
Revenue forecast in 2033
USD 7.87 billion
Growth rate
CAGR of 8.69% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Treatment, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; Japan; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key company profiled
Merck & Co., Inc.; Amgen, Inc.; F. Hoffmann-La Roche Ltd.; Pfizer Inc.; Teva Pharmaceutical Industries Ltd.; Sun Pharmaceutical Industries Ltd.; Arbor Pharmaceuticals, LLC; Amneal Pharmaceuticals; Karyopharm Therapeutics, Inc.; Sumitomo Dainippon Pharma Oncology, Inc. (Boston Biomedical, Inc.).
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Glioblastoma Multiforme Treatment Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global glioblastoma multiforme treatment market report based on treatment, end use, and region:

-
Treatment Outlook (Revenue, USD Million, 2021 - 2033)
-
Surgery
-
Radiation Therapy
-
Chemotherapy
-
Temozolomide
-
Lomustine
-
Carmustine Wafers
-
Other
-
-
Targeted Therapy (Bevacizumab)
-
Tumor Treating Field (TTF) Therapy
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Hospitals
-
Clinics / Outpatient Centers
-
Ambulatory Surgical Centers
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global glioblastoma multiforme treatment market size was estimated at USD 4.04 billion in 2025 and is expected to reach USD 4.39 billion in 2026.
b. The global glioblastoma multiforme treatment market is expected to grow at a compound annual growth rate of 8.69% from 2026 to 2033 to reach USD 7.87 billion by 2033.
b. Surgery dominated the GBM treatment market with a share of 32.52% in 2025. The market share can be attributed to high penetration and increased adoption rate of the therapy owing to the rising prevalence of the disease in the market.
b. Some key players operating in the glioblastoma multiforme treatment market include Merck & Co., Inc., Amgen, Inc., and F. Hoffmann-La Roche Ltd.
b. Key factors that are driving the GBM treatment market growth include the growing prevalence of glioblastoma multiforme, increasing R&D, favorable regulatory scenario, and the presence of a strong pipeline.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










