- Home
- »
- Pharmaceuticals
- »
-
Myeloproliferative Disorders Drugs Market, Industry Report, 2030GVR Report cover
![Myeloproliferative Disorders Drugs Market Size, Share & Trends Report]()
Myeloproliferative Disorders Drugs Market (2025 - 2030) Size, Share & Trends Analysis Report By Indication (Ph+ Chronic Myelogenous Leukemia (CML)), By Treatment Type, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-2-68038-436-9
- Number of Report Pages: 100
- Format: PDF
- Historical Range: 2018 - 2023
- Forecast Period: 2025 - 2030
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Myeloproliferative Disorders Drugs Market Summary
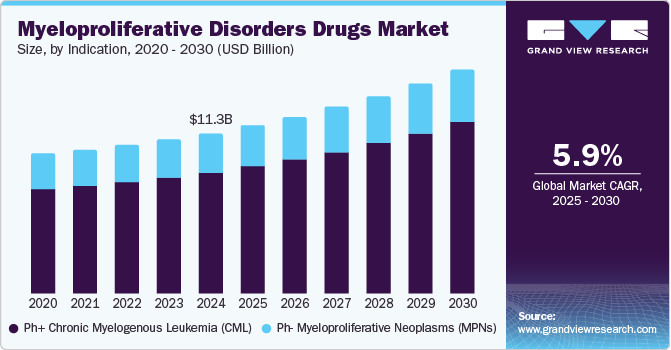
The global myelodysplastic syndrome drugs market size was estimated at USD 11.34 billion in 2024 and is projected to reach USD 15.83 billion by 2030, growing at a CAGR of 5.9% from 2025 to 2030. The increasing prevalence of myeloproliferative disorders, particularly among older adults, is a major contributor to market expansion.
Key Market Trends & Insights
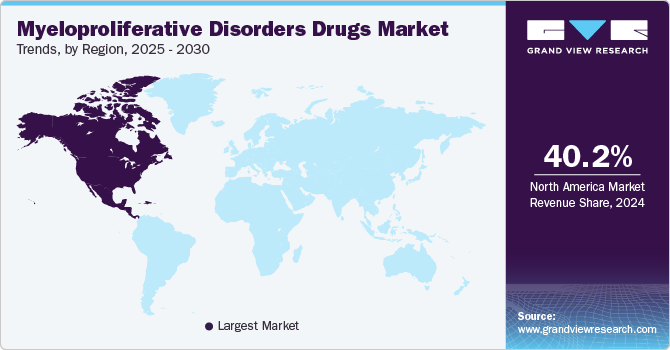
- The North America myeloproliferative disorders drugs market dominated the global market with a revenue share of 40.2% in 2024.
- The Asia Pacific myeloproliferative disorders drugs market is expected to register the highest CAGR of 9.5% over the forecast period.
- Based on indication, the Ph+ chronic myelogenous leukemia (CML) segment dominated the market with a revenue share of 75.5% in 2024.
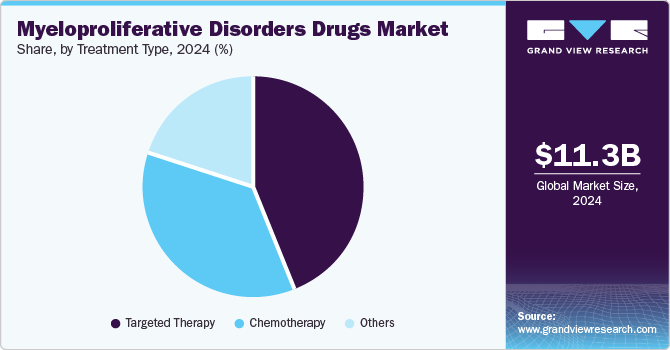
- Based on treatment type, the targeted therapy segment dominated the market with the largest revenue share in 2024.
- Based on end-use, the hospitals segment dominated the market with the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 11.34 Billion
- 2030 Projected Market USD 15.83 Billion
- CAGR (2025-2030): 5.9%
- North America: Largest market in 2024
- Asia Pacific: Fastest growing market
According to the National Institute of Health, chronic myelogenous leukemia has an incidence rate of approximately 2.4 new cases per 100,000 men compared to 1.4 in women as of August 2022. This rising incidence correlates with the aging population, which is more susceptible to such diseases, thereby increasing the demand for effective treatments in the myeloproliferative disorders drugs industry.
Advancements in precision medicine are transforming treatment approaches within the myeloproliferative disorders drugs market. Precision medicine allows tailored therapies based on individual genetic profiles, enhancing treatment effectiveness and reducing side effects. The approval of targeted therapies such as ruxolitinib has marked a significant advancement in treatment options, leading to better patient outcomes and higher adoption rates. As more innovative therapies emerge that address specific genetic mutations associated with these disorders, the myeloproliferative disorders drugs industry is expected to witness robust growth.
In addition, the growing emphasis on research and development for rare diseases fuels innovation within the myeloproliferative disorders drugs market. As pharmaceutical companies invest more resources into understanding and treating rare conditions, the number of novel drug candidates entering clinical trials increases. This focus on developing effective therapies for myeloproliferative disorders expands the available treatment options and enhances competition among manufacturers. Consequently, this trend contributes to strong growth prospects for the myeloproliferative disorders drugs industry as new therapies are introduced to meet patient needs.
Indication Insights
The Ph+ Chronic Myelogenous Leukemia (CML) segment dominated the market with a revenue share of 75.5% in 2024, driven by the increasing incidence of CML, which has led to increased demand for effective treatment options. Introducing targeted therapies, particularly tyrosine kinase inhibitors (TKIs), has revolutionized treatment protocols, improving patient outcomes significantly. Furthermore, ongoing research and development efforts yield novel therapies that enhance efficacy and safety profiles, thus attracting more patients to treatment pathways. The robust pipeline of drugs specifically targeting CML also contributes to the segment growth, as healthcare providers increasingly adopt these innovative solutions to manage the disease effectively.
The Ph- Myeloproliferative Neoplasms (MPNs) segment is projected to grow at a significant CAGR over the forecast period, fueled by the rising awareness and diagnosis rates of MPNs, leading to earlier interventions. In addition, advancements in molecular biology have facilitated the identification of specific genetic mutations associated with MPNs, which has spurred the development of targeted therapies tailored to these abnormalities. The increasing prevalence of MPNs among aging populations further drives myeloproliferative disorders drugs industry demand, as older adults are more susceptible to these disorders. Moreover, expanding clinical trials focusing on innovative treatment approaches enhances the therapeutic landscape for patients suffering from MPNs.
Treatment Type Insights
The targeted therapy segment dominated the market with the largest revenue share in 2024, attributed to the growing preference for precision medicine, which tailors treatments based on individual patient profiles and disease characteristics. Targeted therapies have shown superior efficacy to traditional chemotherapy, leading to better patient outcomes and fewer side effects. In addition, ongoing research into next-generation TKIs is expected to expand treatment options further and improve response rates among patients with resistant forms of myeloproliferative disorders. The increasing availability of these advanced therapies also contributes to their market dominance.
The others segment is projected to grow at the highest CAGR over the forecast period due to the expansion of combination therapies that integrate various treatment modalities, which is becoming increasingly popular among healthcare providers. These approaches aim to enhance therapeutic efficacy while minimizing adverse effects associated with single-agent therapies. Furthermore, innovations in supportive care treatments that address symptoms and improve the quality of life for patients with myeloproliferative disorders are gaining traction. As healthcare professionals continue exploring diverse treatment options, this segment is expected to capture a larger market share in the myeloproliferative disorders drugs industry.
End-use Insights
The hospitals segment dominated the market with the largest revenue share in 2024, driven by the comprehensive care provided within hospital settings, where multidisciplinary teams can offer integrated treatment plans that encompass diagnosis, therapy administration, and ongoing patient management. Hospitals are equipped with advanced technologies and resources that facilitate timely interventions and monitoring of complex cases associated with myeloproliferative disorders. In addition, increased hospital admissions for severe manifestations of these diseases contribute to higher drug utilization rates within these hospitals.
The specialty clinics segment is expected to grow at a significant CAGR over the forecast period, which can be attributed to the increased focus on specialized care for patients with myeloproliferative disorders. These clinics often provide tailored treatment plans that cater specifically to the unique needs of patients suffering from blood cancers. The rise in patient referrals from primary care providers seeking specialized expertise further supports this trend. In addition, as patients become more informed about their treatment options, there is a growing preference for receiving care in specialized settings that offer comprehensive services and support tailored to their conditions.
Regional Insights
North America myeloproliferative disorders drugs market dominated the global market with a revenue share of 40.2% in 2024, driven by high prevalence rates of myeloproliferative disorders coupled with advanced healthcare infrastructure that supports innovative treatments. The region benefits from substantial investments in research and development to discover new therapeutic agents and improve existing ones. Furthermore, regulatory approvals for novel drugs enhance market accessibility and adoption among healthcare providers, contributing significantly to revenue growth within North America.
U.S. Myeloproliferative Disorders Drugs Market Trends
The U.S. myeloproliferative disorders drugs market dominates North America with a significant revenue share in 2024 due to its robust pharmaceutical ecosystem characterized by extensive research activities and numerous clinical trials focused on blood cancers. The presence of leading biopharmaceutical companies promotes innovation and accelerates drug development processes, ensuring that new therapies reach patients swiftly. In addition, favorable reimbursement policies encourage healthcare providers to utilize advanced treatments, further driving the U.S. myeloproliferative disorders drugs market growth.
Europe Myeloproliferative Disorders Drugs Market Trends
Europe myeloproliferative disorders drugs market held a substantial market share in 2024, driven by increasing awareness and early diagnosis initiatives across member states. Enhanced collaboration between healthcare providers and researchers has led to significant advancements in treatment methodologies tailored for the European population. Furthermore, ongoing clinical trials exploring novel agents are expected to support market growth as new therapies receive regulatory approval and become available for patient use.
Asia Pacific Myeloproliferative Disorders Drugs Market Trends
The Asia Pacific myeloproliferative disorders drugs market is expected to register the highest CAGR of 9.5% over the forecast period. This can be attributed to the increasing population combined with rising healthcare expenditures that enhance access to medical services and treatments for myeloproliferative disorders. In addition, growing awareness about these diseases among healthcare professionals leads to better diagnosis rates and subsequent treatment initiation. The expansion of pharmaceutical companies into emerging markets within Asia Pacific also contributes positively by introducing novel therapies tailored to the local population.
The China myeloproliferative disorders drugs market dominates the Asia Pacific with a significant revenue share in 2024 due to its large patient population and increasing investments in healthcare infrastructure to improve disease management strategies. The Chinese government initiatives focusing on enhancing cancer care services have led to greater availability of treatments for myeloproliferative disorders. Moreover, local pharmaceutical companies are actively developing innovative therapies specifically targeting these conditions, further solidifying China's position as a leader within the myeloproliferative disorders drugs market.
Key Myeloproliferative Disorders Drugs Company Insights
Some key companies operating in the market include Novartis AG, Bristol-Myers Squibb Company, Pfizer Inc., Takeda Pharmaceutical Company Limited, and Incyte. Companies are undertaking strategic initiatives such as mergers, acquisitions, and product launches to expand their market presence and address the evolving healthcare demands through the myeloproliferative disorders drugs market.
-
Novartis AG provides diverse, innovative products and solutions for the myeloproliferative disorders drugs market, focusing on addressing critical medical needs. Their notable offerings include Scemblix (asciminib), a groundbreaking treatment for Chronic Myeloid Leukemia (CML) that offers improved efficacy for patients who have not responded well to previous therapies. Jakavi (ruxolitinib) is another key product approved for polycythemia vera and myelofibrosis, which helps manage symptoms and improves patients quality of life.
-
Bristol-Myers Squibb offers a variety of innovative products for the myeloproliferative disorders drugs market, focusing on enhancing patient care. Their primary product, Inrebic (fedratinib), is a once-daily oral therapy approved for myelofibrosis, effectively reducing spleen size and alleviating related symptoms in patients who are new to JAK inhibitors or have previously been treated with ruxolitinib. In addition, they provide Reblozyl (luspatercept), which treats anemia in patients with myelodysplastic syndromes (MDS) by improving red blood cell production and decreasing the need for transfusions.
Key Myeloproliferative Disorders Drugs Companies:
The following are the leading companies in the myeloproliferative disorders drugs market. These companies collectively hold the largest market share and dictate industry trends.
- Novartis AG
- Bristol-Myers Squibb Company
- Pfizer Inc.
- Takeda Pharmaceutical Company Limited.
- Incyte.
- Teva Pharmaceutical Industries Ltd.
- AbbVie Inc.
- Eli Lilly and Company.
- GSK plc.
- GL Pharma
Recent Developments
-
In October 2024, Novartis announced that the U.S. Food and Drug Administration (FDA) granted accelerated approval for Scemblix (asciminib) as a first-line treatment for adults with newly diagnosed Philadelphia chromosome-positive chronic myeloid leukemia in the chronic phase. This decision was based on the ASC4FIRST Phase III trial results, which showed that Scemblix achieved superior major molecular response rates compared to standard tyrosine kinase inhibitors.
-
In March 2024, Bristol-Myers Squibb announced that the European Commission approved Abecma (idecabtagene vicleucel), making it the first CAR T-cell therapy authorized in the European Union for earlier lines of treatment in patients with recurrent and treatment-resistant multiple myeloma.
Myeloproliferative Disorders Drugs Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 11.87 billion
Revenue forecast in 2030
USD 15.83 billion
Growth rate
CAGR of 5.9% from 2025 to 2030
Base year for estimation
2024
Historical data
2018 - 2023
Forecast period
2025 - 2030
Report updated
December 2024
Quantitative units
Revenue in USD billion and CAGR from 2025 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, trends
Segments covered
Indication,treatment type, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East & Africa
Country scope
U.S., Canada, Mexico, UK, Germany, France, Italy, Spain, Norway, Denmark, Sweden,Japan, China, India, South Korea, Australia, Thailand, Brazil, Argentina, South Africa, Saudi Arabia, UAE, Kuwait
Key companies profiled
Novartis AG; Bristol-Myers Squibb Company; Pfizer Inc.; Takeda Pharmaceutical Company Limited.; Incyte.; Teva Pharmaceutical Industries Ltd.; AbbVie Inc.; Eli Lilly and Company.; GSK plc.; GL Pharma
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Myeloproliferative Disorders Drugs Market Report Segmentation
This report forecasts global, regional, and country revenue growth and analyzes the latest industry trends in each sub-segment from 2018 to 2030. For this study, Grand View Research has segmented the global myeloproliferative disorders drugs market report based on indication, treatment type, end use, and region:
-
Indication Outlook (Revenue, USD Billion, 2018 - 2030)
-
Ph+ Chronic Myelogenous Leukemia (CML)
-
Ph- Myeloproliferative Neoplasms (MPNs)
-
Myelofibrosis (MF)
-
Polycythemia Vera (PV)
-
Essential Thrombocythemia (ET)
-
-
-
Treatment Type Outlook (Revenue, USD Billion, 2018 - 2030)
-
Chemotherapy
-
Targeted Therapy
-
Others
-
-
End Use Outlook (Revenue, USD Billion, 2018 - 2030)
-
Hospitals
-
Specialty Clinics
-
Others
-
-
Regional Outlook (Revenue, USD Billion, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Norway
-
Denmark
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










