- Home
- »
- Clinical Diagnostics
- »
-
Point Of Care Diagnostics Market Size, Industry Report, 2033GVR Report cover
![Point Of Care Diagnostics Market Size, Share & Trends Report]()
Point Of Care Diagnostics Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (Infectious Diseases, Glucose Testing, Cardiac Markers), By End Use (Hospitals, Home, Assisted Living Healthcare Facilities, Laboratory), By Region, And Segment Forecasts
- Report ID: 978-1-68038-046-0
- Number of Report Pages: 300
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Point Of Care Diagnostics Market Summary
The global point of care diagnostics market size was estimated at USD 21.79 billion in 2025 and is projected to reach USD 29.94 billion by 2033, growing at a CAGR of 4.07% from 2026 to 2033. The increasing geriatric population and the ability of point of care (POC) diagnostic tests to provide immediate results are expected to fuel market growth.
Key Market Trends & Insights
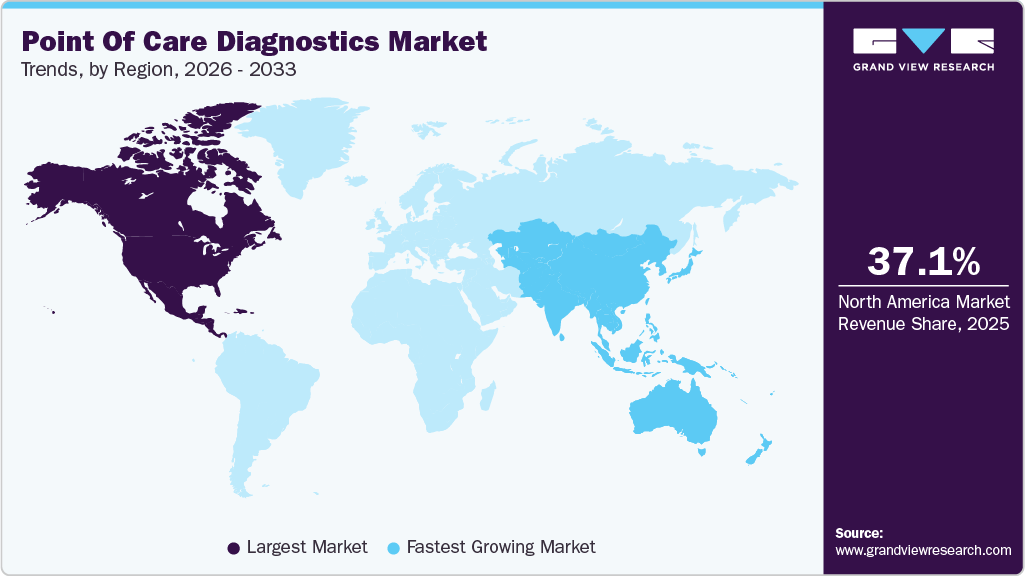
- North America point of care diagnostics industry dominated and accounted for a 44.37% share in 2025.
- Asia Pacific is emerging as the fastest-growing point of care diagnostics industry, with an anticipated CAGR of 6.29% over the forecast period.
- The point of care diagnostics market in Japan is expanding rapidly, holding the second-largest revenue share in 2025.
- Based on product, the infectious disease segment led the market and accounted for 30.53% of global revenue share in 2025.
- Based on end use, clinics led the market and accounted for the highest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 21.79 Billion
- 2033 Projected Market Size: USD 29.94 Billion
- CAGR (2026-2033): 4.07%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Furthermore, increasing adaptability of mobile diagnostic devices in middle-income countries is also one of the key factors for the growth of point of care testing. In addition, the rise in funding from government & private institutions is a key trend driving the growth of this market. For instance, in October 2025, the Biomedical Advanced Research and Development Authority (BARDA) awarded funding to Beckman Coulter to validate the effectiveness of the biomarker to detect MIS-C with a large, multi-center clinical trial. In addition, the market expansion is primarily driven by the ongoing introduction of new products, with leading companies consistently introducing advanced coagulation testing devices that cater to the evolving needs of healthcare providers and patients, offering enhanced features like precision and user-friendly interfaces. For instance, in February 2024, Roche introduced three new coagulation tests for oral Factor Xa inhibitors, rivaroxaban, apixaban, and edoxaban, in countries accepting the CE mark. These tests could aid clinical decision-making in patients treated with direct oral anticoagulants for systemic embolism, stroke prevention, and venous thromboembolism prevention & treatment.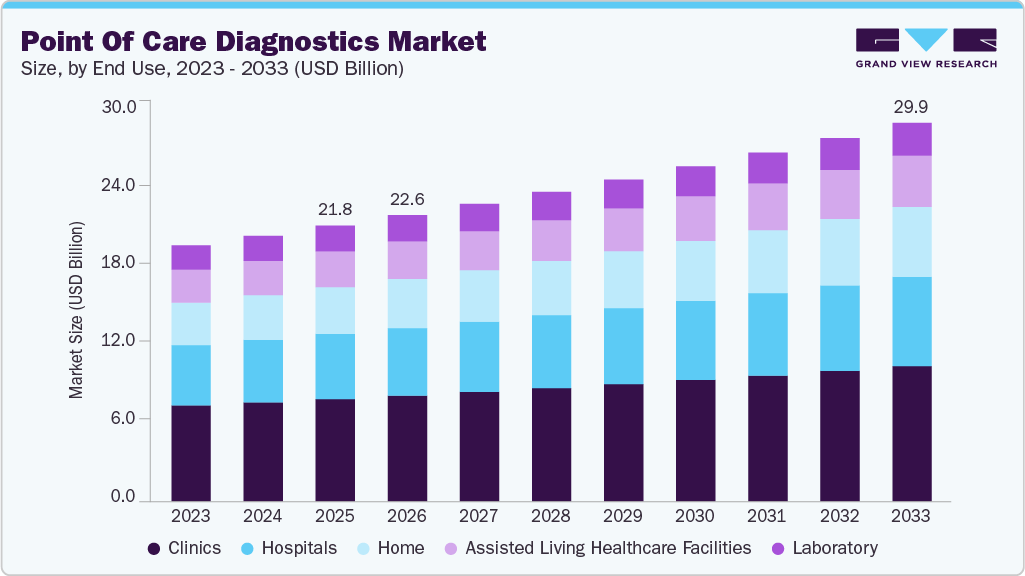
The increasing prevalence of genetic disorders is likely to drive the diagnostics market in the coming years. According to a report published by the CDC, congenital heart defects were the most common birth defects in the U.S., affecting approximately 1% of births every year. Early detection of genetic variations via prenatal testing can help in rapid disease diagnosis, prevention, and selection of suitable treatment. Changing lifestyle and environmental factors are increasing the incidence of genetic diseases. The incidence of breast and ovarian cancers is increasing in many regions. For instance, according to the Breast Cancer Research Foundation, in 2023, approximately 300,590 people are expected to be diagnosed with breast cancer in the U.S. Thus, the growing incidence of various diseases is likely to boost the market over the forecast period.
Moreover, the geriatric population is rapidly increasing across the globe. According to a UN report, in 2020, there were about 727 million people aged 65 & above globally. In addition, the number of individuals aged 80 and above is projected to double by 2050, which is over 1.5 billion. Aging has become a substantial risk factor for numerous diseases, including obesity and diabetes, which, in turn, significantly increases the risk of infectious diseases. The geriatric population is susceptible to numerous diseases such as cancer, cardiovascular diseases, obesity, neurological disorders, and diabetes. Thus, the growing geriatric population globally is anticipated to be a high-impact rendering driver of the point of care diagnostics market.
The table below highlights the prevalence of diabetes in the age group of 20 to 79:
Table 1 Prevalence Of Diabetes In 2021, 2030, And 2045
Region
2021
2030
2045
Percentage Increase
Global
537 million
643 million
783 million
46%
North America and the Caribbean
51 million
57 million
63 million
24%
South and Central America
32 million
40 million
49 million
50%
Africa
24 million
33 million
55 million
134%
Middle East and North Africa
73 million
95 million
136 million
87%
Southeast Asia
90 million
113 million
152 million
68%
Western Pacific
206 million
238 million
260 million
27%
Europe
61 million
67 million
69 million
13%
Source: International Diabetes Federation 2021, Grand View Research
The increasing prevalence of genetic disorders is likely to drive the POC molecular diagnostics market in the coming years. According to a report published by CDC, congenital heart defects were the most common birth defects in the U.S., affecting approximately 1% of births every year. Early detection of genetic variations via prenatal testing can help in rapid disease diagnosis, prevention, and selection of suitable treatment. Changing lifestyle and environmental factors are increasing the incidence of genetic diseases. The incidence of breast and ovarian cancers is increasing in many regions. Thus, the growing incidence of various diseases is likely to boost the market over the forecast period.
Table 2 Global Incidence Of Diseases (2022/2023)
Diseases
New Cases (in Million)
Chronic Diseases
Cancer
19.1
Diabetes
537
Heart disease
17.9
Asthma
339
Infectious Disease
HIV
38.4
Hepatitis C
71
Hepatitis B
296
Tuberculosis
10.6
Genetic Disorders
Cystic fibrosis
0.1
Congenital heart disease
11.99
Sickle cell anemia
0.3
Source: WHO, GLOBOCAN, CDC, UNAIDS, Grand View Research
Moreover, increasing penetration of POC diagnostic home-based health services is projected to support the market expansion. As of May 2023, there were 278 molecular diagnostics tests approved for COVID-19 infection detection by the U.S. FDA. Out of the 278 molecular tests available in the market, around 50 tests can be performed using home-collected samples. The market is flooded with new molecular diagnostic tests for the detection of SARS-CoV-2, making it saturated.
PoC testing, as the name suggests, refers to tests designed to be used at the patient site. Some of the major factors propelling demand for PoC healthcare facilities are:
-
Government/political support: To curb healthcare expenditure, U.S. and Canadian governments are focusing on reducing hospital stays and minimizing inpatient treatment costs via outpatient care models, such as clinics and home healthcare.
-
Clinical/economic outcomes: To provide effective and rapid diagnostic results, clinics and other healthcare establishments are increasingly adopting POC tests.
-
Technology: Technological improvements and the consequent introduction of cheap & high-quality medical solutions aimed at achieving lab automation are likely to support POC diagnostics market.
-
End-user awareness: Increasing patient awareness is likely to drive this market, as patients—especially the ones suffering from long-term diseases such as diabetes—are willingly participating in the shift toward implementation of POC diagnostics. Moreover, healthcare practitioners are becoming increasingly dependent on POC medical devices and consumables.
-
Information technology: This factor plays a crucial role in the further development of POC diagnostics. The introduction of healthcare information systems, such as Electronic Medical Records (EMR), has largely increased the market penetration rate of these tests.
-
Implementation of waived tests under Clinical Laboratory Improvement Amendments (CLIA): This amendment refers to POC testing, which is categorized into waived and non-waived tests. Waived tests are those approved by the FDA for home use, employing simple and accurate methodologies to eliminate the likelihood of erroneous results and ensure no reasonable risk or harm can come to patients if performed incorrectly. Non-waived tests are further categorized into:
-
Moderately complex testing: Tests requiring minimal scientific & technical knowledge and training to accurately perform operational steps, which are either automatically executed or easily controlled involving minimal interpretation and judgment.
-
Highly complex testing: Tests requiring specialized scientific & technical knowledge, training, and experience to accurately perform operational steps, with close monitoring or control that involves extensive independent interpretation and judgment (the below figure provides a brief description of CLIA-waived tests).
Market Concentration & Characteristics
Market growth stage is high, and the pace of growth is accelerating. The industry is characterized by a high degree of innovation owing to rapid technological advancements and the launch of novel products by key players in the market. For instance, in February 2023, Huwel Lifesciences designed a portable RT-PCR machine to test types of viruses. The company claimed that the test takes around 30 minutes and can be used to detect respiratory and other infections using blood and gastrointestinal samples. Innovation in HbA1c testing technology has led to the development of breakthrough products that set new standards in diabetes care. For instance, in January 2024, the FDA approved Qvin's Q-Pad A1c Test System for measuring hemoglobin A1c in diabetes patients using menstrual blood. The system includes the Q-Pad A1c Test and Q-Pad Kit, designed for individuals aged 18 and older to collect menstrual blood samples using the Q-Pad, an unscented pad with an embedded blood collection strip.
The point of care diagnostics industry experiences a high level of mergers and acquisitions (M&A), as companies pursue strategic partnerships and acquisitions to expand product portfolios, enter new market segments, and strengthen their global presence. Large players often acquire smaller, innovative firms to integrate advanced technologies, such as molecular diagnostics and digital health solutions, into their offerings. For instance, in March 2023, Werfen acquired Immucor, Inc. to establish a strong presence in Specialized Diagnostics. The deal was valued at USD 2 billion. Similarly, in May 2023, Siemens Healthcare GmbH partnered with Unilabs, with an agreed value exceeding USD 200 million. Within the partnership, Unilabs will purchase 400 laboratory analyzers to improve patient care infrastructure. Operating players in point of care diagnostics market leverage these strategies to increase their product capabilities and promote the reach of their offerings.
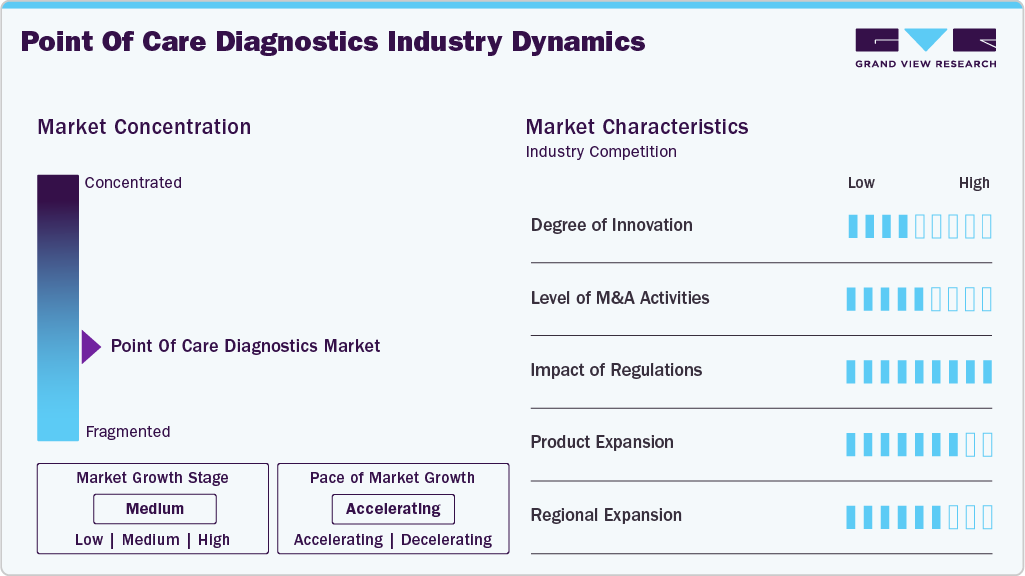
Regulatory frameworks play a crucial role in shaping the point-of-care (POC) diagnostics market, influencing product approvals, market entry strategies, and overall industry growth. Stringent guidelines from agencies like the U.S. FDA, European Medicines Agency (EMA), and China's NMPA ensure the safety, accuracy, and reliability of POC diagnostic devices. However, regulatory complexities can also delay product launches and add to compliance costs for manufacturers. Recent trends indicate a shift towards accelerated approval pathways, especially for infectious disease diagnostics, enabling faster commercialization. Additionally, initiatives promoting decentralized testing, such as the CLIA waiver program in the U.S., are expanding POC adoption in non-traditional healthcare settings, including pharmacies and home testing.
Product expansion in the point of care diagnostics industry is at a high level, driven by the growing demand for comprehensive diagnostic solutions across multiple disease areas. For instance, in July 2023, Brii Biosciences Limited, a biotechnology company, expanded its hepatitis B virus portfolio in partnership with VBI Vaccines Inc., extending its exclusive license for BRII-179 to global markets and gaining exclusive rights to develop & market PreHevbri in Greater China and the Asia Pacific region. PreHevbri is a three-antigen adult HBV vaccine approved in the European Union/European Economic Area, the U.S., the UK, Canada, and Israel.
The industry is witnessing a medium to high level of regional expansion as companies aim to tap into growth opportunities in emerging markets while solidifying their presence in established regions. Asia-Pacific, Latin America, and the Middle East are becoming key targets due to rising healthcare investments, increasing awareness of diagnostic testing, and expanding healthcare infrastructures. Market leaders are setting up local manufacturing units, establishing partnerships with regional distributors, and customizing product offerings to meet specific market requirements. However, regional expansion efforts are often challenged by varying regulatory landscapes and market access barriers, requiring tailored strategies for each geography.
Hematology POC devices
Hematology testing equipment, which may be used at the POC, has improved diagnostic and patient outcomes. Furthermore, these devices are simple to use and can be used outside hospital settings. This is expected to improve the acceptance and accessibility of hematological testing, resulting in market growth. Furthermore, the growing partnership among key players for the distribution of these products is further driving market growth. For instance, in September 2022, PixCell Medical entered into a partnership with Insight Medical for the distribution of the company’s POC hematology analyzer.
The availability of technologically advanced and portable hematology analyzers, such as Sight Diagnostics’ Sight OLO Analyzer devices, and PixCell Medical’s HemoScreen analyzer, has made blood testing feasible for a broad range of use cases. Furthermore, POC tests can deliver quality results in a short turnaround time and enable prompt & effective management of treatments even for life-threatening conditions. These factors are expected to drive the segment and boost the demand for hematology POC tests. A broad range of these tests is commercially available, key examples of which, along with their usage settings, have been listed below:
Table 3 Hematology POC Devices And Test Settings
Test
POC test devices
POC settings
Full blood count
Pentra 60 (Horiba), XN-L450, XN-L 550 (Sysmex)
Emergency rooms, hematology clinics, and general practice
Prothrombin time/International normalized ratio
CoaguChek XS series
i-Stat (Abbott)
Emergency rooms, coronary care units, anticoagulant clinics, operation theatres, and ICU
Activated partial thromboplastin time
CoaguChek Pro II (Roche Diagnostics Ltd)
Acute and intensive care settings
Hemoglobin
HemoCue
Operation theatres, clinics, general practice, and obstetrics
D-dimer
Alere (Abbott)
Clearview (Abbott)
Emergency rooms, primary care clinics, and medical assessment units
Viscoelastic assays
Rotational thromboelastometry (ROTEM),
Thromboelastography (TEG)
Operating theatres and emergency rooms
Activated clotting time
Sonoclot system (Sienco)
Cardiothoracic theatre
Malaria antigen
Optimal (BioRad),
Clearview (Abbott)
Malaria screening and identification laboratories in rural settings
Source: Company Websites (Sysmex Corporation, Abbott, F. Hoffmann-La Roche Ltd, Bio-Rad Laboratories, Inc., etc.), Grand View Research
Product Insights
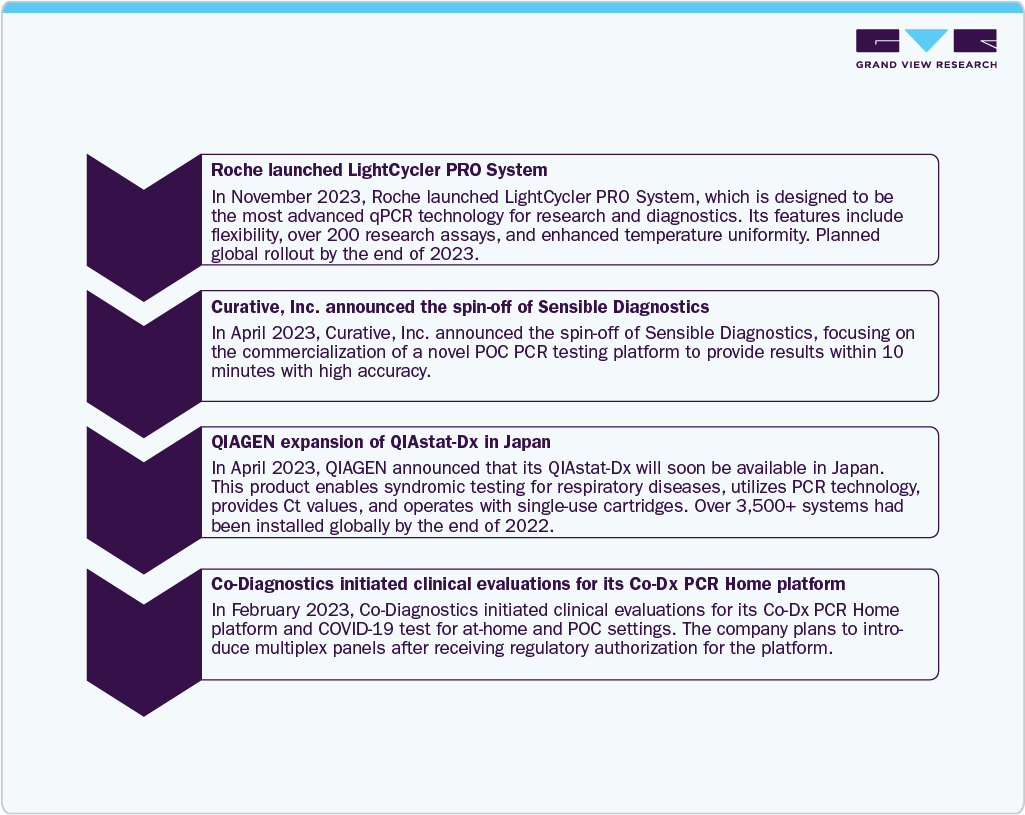
Infectious diseases led the market and accounted for 30.53% of global revenue share in 2025. Growth of the segment is attributed to increasing demand for rapid tests, which has encouraged industry players to deliver point of care solutions to decentralized regions and launch innovative solutions. For instance, in May 2023, Sensible Diagnostics announced plans to launch a POC PCR instrument that can perform PCR in 10 minutes by 2024, with initial focus on infectious diseases. Moreover, Abbott has announced the launch of ID NOW, the world's fastest molecular POC test, which offers COVID-19 results in 13 minutes and can be used in a range of dispersed healthcare settings, such as doctor offices & urgent care clinics. Moreover, the implementation of government initiatives aimed at curbing healthcare expenditure by restricting the growth of the incidence of infectious diseases will serve this industry as a driver.
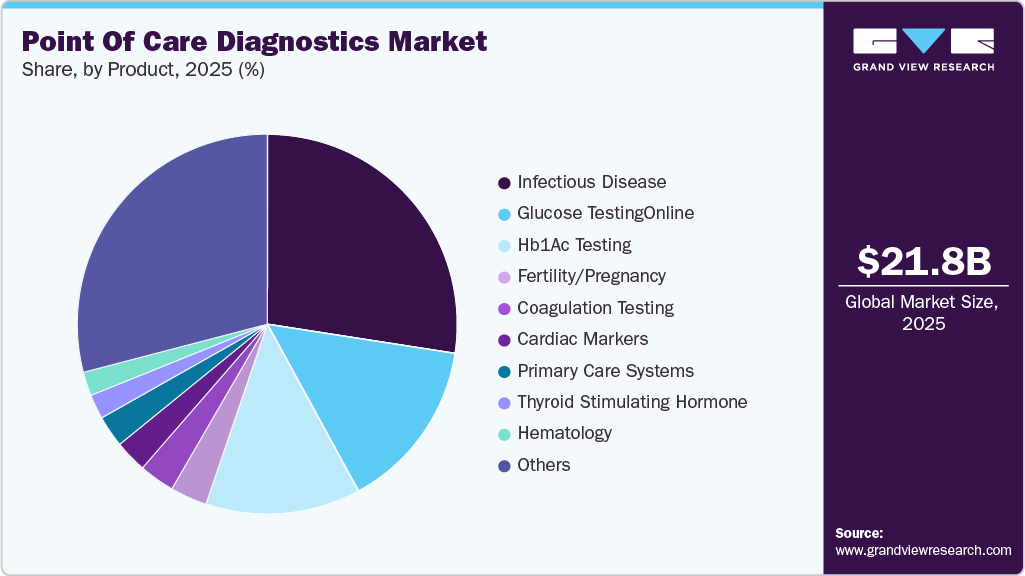
The cancer segment is expected to register the fastest CAGR during the forecast period. Growing use of markers in cancer diagnostics is the primary factor likely to boost the cancer markers segment during the forecast period. The rising global prevalence of cancer is driving the need for early disease detection, which is also expected to propel the market. For instance, according to Cancer.org, approximately 1.9 million new cases of cancer were diagnosed in the U.S. in 2021, with 608,570 cancer deaths. Moreover, key players in the market are introducing novel products to meet untapped opportunities in the market. For instance, in October 2022, F. Hoffman-La Roche Ltd., approval for the first companion diagnostic by the U.S. FDA to detect patients with HER2 low metastatic breast cancer eligible for ENHERTU.
End Use Insights
Clinics led the market and accounted for the highest revenue share in 2025. Point of care diagnostics in primary care settings range from simple glucose testing to complex coagulation testing. In several clinics, professionals are switching to point of care diagnostics from conventional lab testing, and this helps shorten the time taken to decide whether further tests are required by avoiding delays during specimen preparation and transport. Other advantages of point of care diagnostics are rapid availability of results, lower costs, and better outcomes. The market has grown rapidly in the past few years, especially in the U.S. and European countries, owing to increased awareness regarding various issues such as medical & organizational concerns and economic advantages of point of care diagnostics.
The home sector is projected to witness the highest growth rate over the forecast period, owing to the comfort level and cost-effectiveness of point of care diagnostics provided to patients at home. POC in the home healthcare sector also allows patients to address healthcare challenges at home and make decisions instantly. Moreover, with the rise in the aging population, which is more susceptible to chronic diseases, the demand for healthcare services is likely to show steady growth in the coming years. This will boost the home healthcare sector, which can improve the overall access and reach to medical care while helping avoid unnecessary visits, hospital admissions, and readmissions, as well as time & costs associated with traveling to meet healthcare providers.
Regional Insights
North America dominated the point of care diagnostics market and accounted for 37.13% share in 2025. The growing geriatric population and presence of higher healthcare expenditure are some key trends contributing to its largest share. Furthermore, technological advancements such as the launch of miniaturized diagnostic equipment offering accurate and rapid results and increasing market penetration of Picture Archiving and Communication Systems (PACS) and Electronic Medical Records (EMRs) are anticipated to propel market growth during the forecast period. Moreover, the presence of key players such as Abbott, BIOMERIEUX, BD, Siemens Healthineers AG, QIAGEN, Quidel Corporation, and Quest Diagnostics is positively influencing market growth. For instance, in April 2023, Abbott announced that the U.S. FDA cleared a reader for FreeStyle Libre 3 integrated continuous glucose monitoring system.
U.S. Point Of Care Diagnostics Market Trends
The U.S. leads the PoC diagnostics market; the presence of key players, such as Abbott, bioMérieux, BD, Siemens Healthineers AG, QIAGEN, Quidel Corporation, and Quest Diagnostics, is positively influencing the U.S. market. For instance, in November 2020, QIAGEN announced offering a portable digital test allowing labs to detect SARS-CoV-2 antigens in individuals with illnesses in 2 to 15 minutes in the U.S. In February 2019, HemoCue, a POC-focused subsidiary of Danaher, announced the launch of its latest hemoglobin test system, Hb 801, in the U.S. This system was designed to rapidly detect anemia and assess hemoglobin levels in one clinical visit. This launch was expected to strengthen the position of Danaher in the clinical applications market space.
Europe Point Of Care Diagnostics Market Trends
The point of care diagnostics market in Europe is rapidly evolving, driven by the increasing prevalence of chronic diseases and an increasing demand for rapid diagnostics in the region are key factors driving the market growth. According to the European Commission, as of January 1, 2023, the population of the EU was approximately 448.8 million individuals, with over one-fifth (21.3%) of the population aged 65 years and older. Furthermore, government initiatives favoring the development of POC diagnostics equipment are expected to fuel market growth. For example, the European Space Agency funded Radisens Diagnostics to develop an innovative blood testing device to be used by astronauts during a mission. Global players in the point of care diagnostics market are launching new tests and devices in the European market to capture a greater market share, which is expected to foster market growth over the forecast period. For instance, in June 2023, Sysmex Corporation launched a POCT system to rapidly detect antimicrobial susceptibility in Europe. This system helps identify infections and assess which antibiotics are effective against them, specifically targeting urinary tract infections.
The UK point of care diagnostics market is experiencing significant growth, driven by the demand for innovative diagnostic and treatment products in the UK, which is further expected to be higher owing to the rising prevalence of chronic diseases, growing geriatric population, and rising awareness among consumers. Although molecular diagnostic services are currently unavailable in most parts of the country, the National Health Service has undertaken several initiatives in training healthcare practitioners.
The point of care diagnostics market in Germany is growing rapidly. The country has a strong healthcare infrastructure in terms of facilities and trained healthcare professionals. Healthcare spending in Germany is comparatively higher than in other European countries. Furthermore, increasing strategic initiatives such as collaboration, new product launches, and partnerships are expected to boost the market. For instance, in July 2023, LumiraDx signed a commercial distribution agreement with Axon Lab AG. The agreement aimed to expand the reach of LumiraDx's POC diagnostic platform and tests in Germany. This partnership leveraged Axon Lab's established network to bring LumiraDx's rapid testing solutions to a wider range of healthcare providers and patients in Germany.
Asia Pacific Point Of Care Diagnostics Market Trends
The Asia Pacific region is emerging as the fastest-growing market for point of care diagnostics, with a CAGR of 6.29% over the forecast period. Development of healthcare infrastructure coupled with a higher prevalence of chronic and targeted diseases, such as diabetes and cancer along with infectious conditions including HIV, syphilis, and RSV, is expected to drive the market in Asia Pacific countries. Adoption of POC diagnostics in operating rooms, emergency rooms, intensive care units, path labs, and hospitals is expected to rise owing to early and efficient results. Moreover, the presence of a strong product pipeline pertaining to the infectious and cardiac markers segment is estimated to enhance the market through to 2033.
Furthermore, a rise in the adoption of miniaturized models and measures adopted for reducing stays in hospitals and clinics, especially in India, is expected to fuel the demand for the India point of care diagnostic industry. Moreover, rapid technological advancements, high prevalence of chronic & infectious diseases, and continuous efforts by local companies as well as organizations are driving the point of care diagnostics market in India. For instance, in January 2023, Cipla Limited, based in India, launched Cippoint, a POC testing device that offers a wide array of testing parameters, such as diabetes, cardiac markers, fertility, infectious diseases, inflammation, thyroid function, coagulation markers, and metabolic markers.
China point of care diagnostics market is expanding rapidly; China is projected to face the world’s largest diabetes epidemic with a steady rise in incidence. Moreover, infectious diseases like malaria, schistosomiasis, tetanus, leishmaniasis, diarrhea, schistosomiasis, diphtheria, measles, tuberculosis, STDs excluding HIV, hepatitis C, hepatitis A, and COVID-19 are prevalent in the country. This rise in the prevalence of infectious diseases and lifestyle disorders, including cardiac diseases, is expected to drive the adoption of at-home POCT, positively impacting growth.
The presence of several companies in China is expected to fuel revenue generation in this segment in the coming years, owing to constant research endeavors taken by the company, mergers & acquisitions, and product development in the POC diagnostics market. For instance, Guangzhou Wondfo Biotech Co Ltd. is one of the major POC testing leaders that developed three new diagnostic tests for COVID-19 diagnosis. Among these tests, the Wondfo SARS-CoV-2 Antibody Test is a POC test based on the principle of the lateral flow method, and it received approval from the National Medical Products Administration (NMPA). This POC test detects IgG and IgM antibodies within 15 minutes in human samples and is efficiently used for on-site monitoring and screening for patients.
Japan point of care diagnostics market is expanding rapidly. Japan held the second-largest revenue share of the market in 2023. The country is reported to have the largest geriatric population in the world due to the increased life expectancy due to medical advancements and reduced fertility rates. This increase in the geriatric population is expected to rise even further over the coming years. According to data from the Ministry of Internal Affairs and Communications, as of September 15, 2023, the population of Japanese aged 65 years and over was 36.2 million. The ratio of elders within the total population increased by 0.1 percentage points to 29.1%, the highest on record. Such a large geriatric population is expected to provide lucrative opportunities to develop POC diagnostics and testing solutions.
Latin America Point Of Care Diagnostics Market Trends
The point of care diagnostics market in Latin America is growing due to the rising focus of multinational key players in this region, and an increase in patient awareness is expected to drive market growth over the forecast period. The region is witnessing a trend toward decentralized IVD testing and increasing demand for affordable POC tests, especially in the area of infectious diseases. Growth in economic & political stability has led to positive economic growth in Latin America, affecting the market growth of POC diagnostics.
Brazil point of care diagnostics market is witnessing substantial growth, fueled by the rising incidence of disease tests related to antenatal syphilis screening, HIV screening, coronavirus, and Zika virus tests are anticipated to register lucrative growth over the forecast period. Healthcare reforms, such as the Unified Health System (SUS) in Brazil, are estimated to boost the usage rates of POC devices.
Middle East & Africa Point Of Care Diagnostics Market Trends
The point of care diagnostics market in the Middle East and Africa (MEA) is poised for growth. Governments and private sectors in MEA are investing in healthcare infrastructure, including the expansion of clinics and hospitals. This development supports the integration of advanced technologies, such as POC diagnostic tools. Several countries in the Middle East, such as UAE, Kuwait, and Egypt, are experiencing economic growth, leading to increased healthcare spending by governments and individuals. This financial capability enables the adoption of advanced healthcare technologies, including POC diagnostics. Furthermore, the growing prevalence of STDs due to lower awareness, increasing purchasing ability, and the awareness of advanced diagnosis methods are the vital factors expected to drive market growth.
Key Point Of Care Diagnostics Company Insights
Some of the key players operating in market include Siemens Healthcare GmbH, Abbott; Danaher Corporation, and F. Hoffmann-La Roche Ltd., Inc. Established players focus majorly on innovation & technology advancements to develop cutting-edge diagnostic solutions and partnering with emerging players to leverage their technology. Mature players also have a strong global presence with a diverse portfolio of PoC products and a well-established brand reputation which gives them a competitive edge. Emerging players however focus launching products in limited countries and then expanding regionally. Some operating strategies also include strategic partnerships, acquisitions, or collaborations to enhance their capabilities and market presence. Additionally, these players may be more flexible and agile than established players in terms of responding and changing to market needs and demand, allowing them to quickly adapt and develop new technologies.
Increasing demand for patient-centric healthcare services and growing adoption of technologically advanced testing products at POC facilities, such as clinics & retail pharmacies, are projected to drive the market expansion over the forecast period. Moreover, market players are continuously involved in the development of novel POC testing products to capitalize on market opportunities.
Key Point Of Care Diagnostics Companies:
The following are the leading companies in the point of care diagnostics market. These companies collectively hold the largest market share and dictate industry trends.
- F. Hoffmann-La Roche Ltd.
- Qiagen
- Danaher Corporation
- Becton Dickinson (BD)
- bioMérieux
- Abbott
- Siemens Healthcare GmbH
- Werfen
- Nova Biomedical
- Trividia Health, Inc.
- QuidelOrtho Corporation
- Trinity Biotech
- Sekisui Diagnostics
- Orasure Technologies, Inc.
- Spectral Medical, Inc.
- EKF Diagnostics Holdings plc.
- Anbio Biotechnology Co., Ltd.
- AccuBioTech Co., Ltd
- ALPHA LABORATORIES.
Recent Developments
-
In November 2025, Sciverse Solutions and Bhat Biotech in India announced a strategic collaboration to co-develop AI-enabled molecular diagnostic platforms, advancing portable point-of-care testing with real-time, high-accuracy results.
-
In June 2025, QuidelOrtho in the U.S. announced a strategic shift in molecular diagnostics, opting to acquire LEX Diagnostics after FDA clearance while discontinuing Savanna platform development to accelerate growth.
-
In May 2025, Siemens Healthineers expanded U.S. operations with a USD 150 million investment, boosting manufacturing, strengthening supply-chain resilience, and enhancing customer support through new and upgraded facilities across multiple states.
-
In February 2025, BD in the U.S. announced plans to separate its Biosciences and Diagnostic Solutions business to sharpen strategic focus, accelerate growth investments, and create greater long-term shareholder value.
Point Of Care Diagnostics Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 22.64 billion
Revenue forecast in 2033
USD 29.94 billion
Growth rate
CAGR of 4.07% from 2026 to 2033
Actual years
2021 - 2024
Forecast years
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Germany; UK; France; Italy; Spain; Russia; Denmark; Sweden; Norway; Japan; China; India; South Korea; Australia; Thailand; Brazil; Mexico; Argentina; South Africa; Saudi Arabia; UAE; Kuwait
Key companies profiled
F. Hoffmann-La Roche Ltd.; Qiagen; Danaher Corporation; Becton Dickinson (BD); bioMérieux; Abbott; Siemens Healthcare GmbH; Werfen; Nova Biomedical; Trividia Health, Inc.; QuidelOrtho Corporation; Trinity Biotech; Sekisui Diagnostics; Orasure Technologies, Inc.; Spectral Medical, Inc.; EKF Diagnostics Holdings plc.; Anbio Biotechnology Co., Ltd.; AccuBioTech Co., Ltd; ALPHA LABORATORIES
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Point Of Care Diagnostics Market Report Segmentation
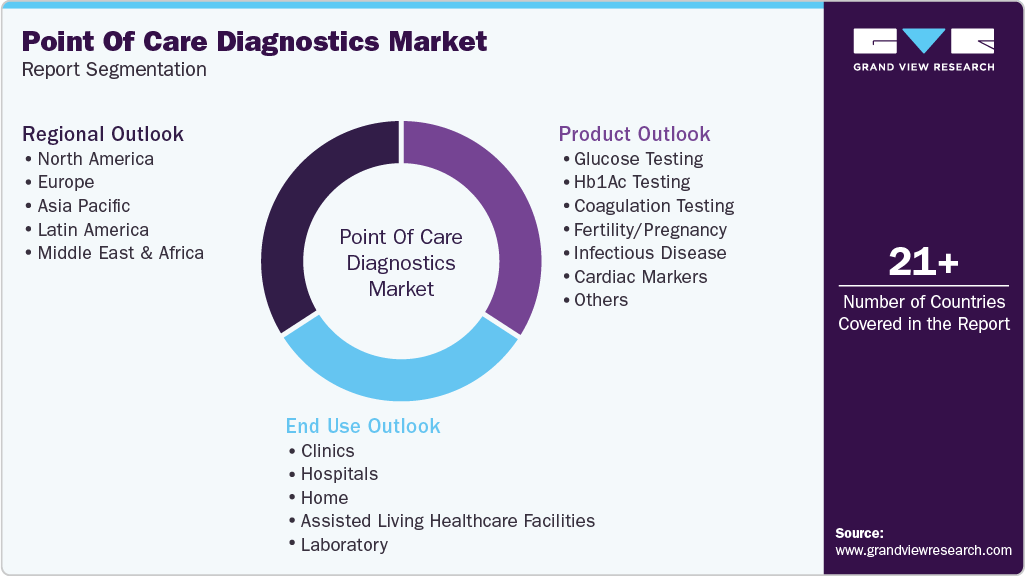
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global point of care diagnostics market report based on product, end use, and region.
-
Product Outlook (Revenue, USD Billion, 2021 - 2033)
-
Glucose Testing
-
Hb1Ac Testing
-
Coagulation Testing
-
Fertility/Pregnancy
-
Infectious Disease
-
HIV POC
-
Clostridium Difficile POC
-
HBV POC
-
Pneumonia or Streptococcus Associated Infections
-
Respiratory Syncytial Virus (RSV) POC
-
HPV POC
-
Influenza/Flu POC
-
HCV POC
-
MRSA POC
-
TB and Drug-resistant TB POC
-
HSV POC
-
COVID-19
-
Other Infectious Diseases
-
-
Cardiac Markers
-
Thyroid Stimulating Hormone
-
Hematology
-
Primary Care Systems
-
Decentralized Clinical Chemistry
-
Feces
-
Lipid Testing
-
Cancer Marker
-
Blood Gas/Electrolytes
-
Ambulatory Chemistry
-
Drug of Abuse (DOA) Testing
-
Autoimmune Diseases
-
Urinalysis/Nephrology
-
-
End-use Outlook (Revenue, USD Billion, 2021 - 2033)
-
Clinics
-
Pharmacy & Retail Clinics
-
Physician Office
-
Urgent Care Clinics
-
Non-practice Clinics
-
-
Hospitals
-
Home
-
Assisted Living Healthcare Facilities
-
Laboratory
-
-
Regional Outlook (Revenue, USD Billion, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
Germany
-
U.K.
-
France
-
Italy
-
Spain
-
Russia
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global point of care diagnostics market size was estimated at USD 21.79 billion in 2025 and is expected to reach USD 22.64 billion in 2026.
b. The global point of care diagnostics market is expected to grow at a compound annual growth rate of 4.07% from 2026 to 2033 to reach USD 29.94 billion by 2033.
b. The infectious disease segment dominated the point of care diagnostics market with a share of 30.53% in 2026. The rising prevalence of HIV/AIDS, influenza, RSV, and other diseases is leading to an increase in the number of people being diagnosed.
b. Some key players operating in the POC diagnostics market include F. Hoffmann-La Roche Ltd.; Abbott; Siemens; Danaher; bioMérieux SA; Johnson and Johnson; Abaxis, Inc.; QIAGEN; Nova Biomedical; Trividia Health, Inc.; Quidel Corporation; OraSure Technologies Inc.; Becton Dickinson and Company; Spectral Medical, Inc.; and Nipro.
b. Key factors that are driving the point of care diagnostics market growth include robust government initiatives, the presence of favorable regulations, and the advent of next-generation point of care diagnostics technologies.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










