- Home
- »
- Medical Devices
- »
-
Positive Airway Pressure Devices Market, Industry Report, 2033GVR Report cover
![Positive Airway Pressure Devices Market Size, Share & Trends Report]()
Positive Airway Pressure Devices Market (2026 - 2033) Size, Share & Trends Analysis Report By Product (CPAP, APAP, BiPAP), By Application (Obstructive Sleep Apnea, Respiratory Failures), By End Use (Home Care Settings, Hospitals & Clinics), By Region, And Segment Forecasts
- Report ID: GVR-3-68038-910-4
- Number of Report Pages: 150
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Positive Airway Pressure Devices Market Summary
The global positive airway pressure devices market size was valued at USD 4.82 billion in 2025 and is projected to reach USD 7.40 billion by 2033, growing at a CAGR of 5.48% from 2026 to 2033. This growth is attributed to the increasing prevalence of sleep apnea, advancements in technology, rising awareness and diagnosis of sleep disorders, and improving healthcare infrastructure.
Key Market Trends & Insights
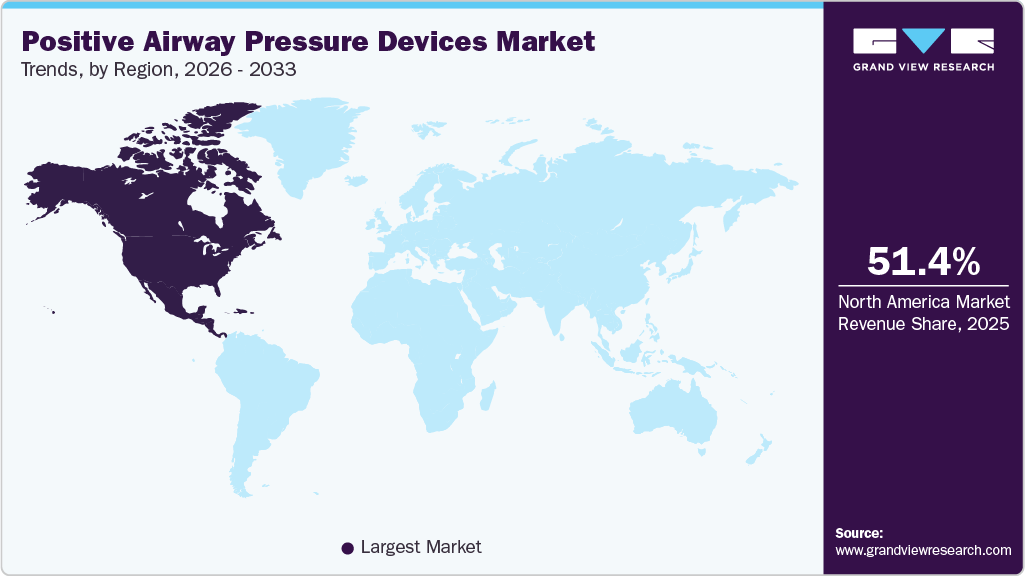
- North America dominated the market and accounted for a 51.39%share in 2025.
- The PAP devices market in the U.S. has seen significant growth over the forecast period due to the growing prevalence of respiratory diseases, such as obstructive sleep apnea, coupled with increasing awareness about the available treatment solutions.
- By product, the Continuous Positive Airway Pressure (CPAP) devices segment dominated the market with a share of 57.4% in 2025.
- By application, the obstructive sleep apnea segment led the market in 2025.
- By end use, the hospitals and clinics segment was the leading one in 2025 and accounted for the largest market share.
Market Size & Forecast
- 2025 Market Size: USD 4.82 Billion
- 2033 Projected Market Size: USD 7.40 Billion
- CAGR (2026-2033): 5.48%
- North America: Largest market in 2025
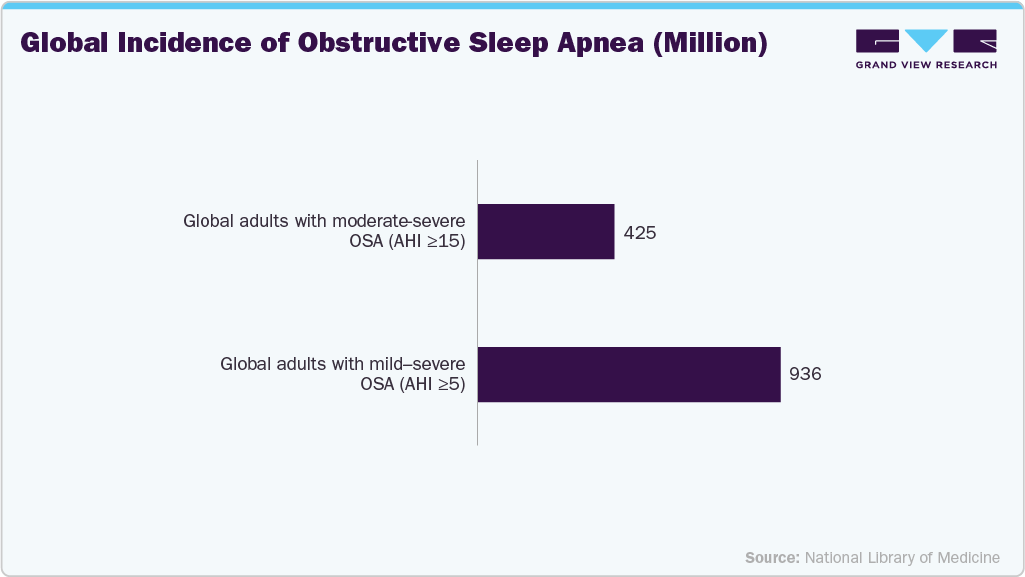
These factors are fueling the demand for PAP devices globally. Growing health concerns, better diagnostic methods, and technological innovations are shaping the market's future growth prospects. Sleep apnea, particularly OSA, has become increasingly common worldwide. The growing prevalence of obesity is a major contributor to the increasing number of sleep apnea cases. Obesity is strongly linked to OSA as excess fat around the neck can obstruct airflow during sleep, leading to interrupted breathing. Sleep apnea cases have seen a sharp increase in regions where obesity rates are high (e.g., the U.S. & Europe). Obesity is a major risk factor for sleep apnea, with up to 40% of obese people also suffering from OSA in the UK. According to an article by the Sleep Apnea Trust in 2023, up to 10 million people in the UK suffer from OSA, of whom around 4 million suffer from moderate or severe OSA. Thus, the increasing prevalence of respiratory diseases in the country is expected to fuel the market over the forecast period. In addition, according to a study published in the Journal of Pulmonary Therapy in July 2022, the most common respiratory symptoms, such as shortness of breath and active cough, affect over 17% of adult citizens.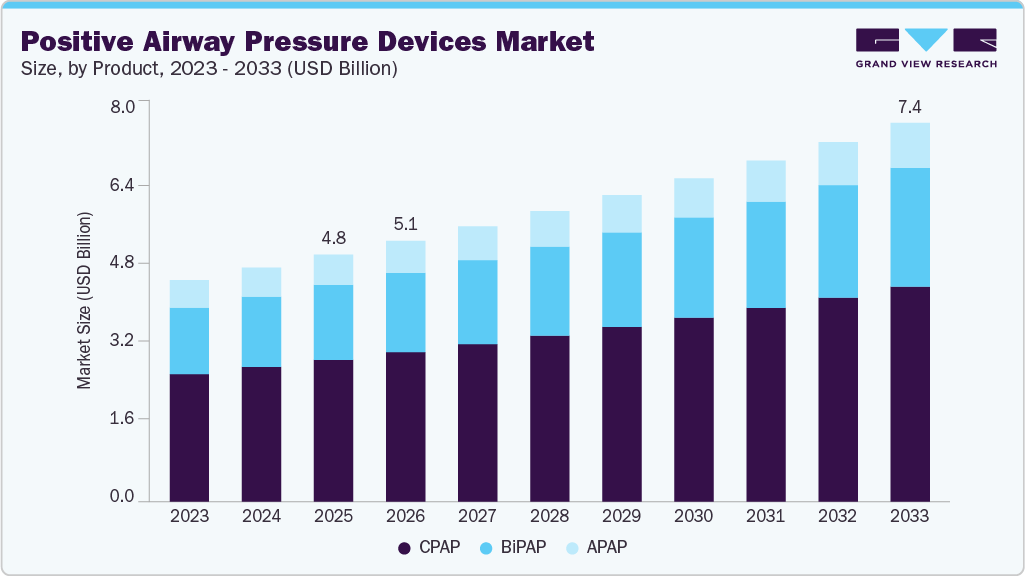
The advanced healthcare infrastructure and high investment in R&D in the U.S. have enabled manufacturers to develop & launch the latest technologies in respiratory devices, making them more efficient, portable, & user-friendly. In January 2022, Cathay Capital made a strategic investment in CPAP.com to support expansion and strengthen its position in the sleep apnea treatment market. The move reflects rising demand for CPAP and PAP devices as obstructive sleep apnea continues to grow significantly due to aging demographics and increasing obesity rates
Rising awareness and diagnosis of sleep disorders are crucial factors propelling market growth. As people become more educated about sleep apnea and its potential risks, they are more likely to seek diagnosis and treatment. Public health campaigns run by organizations such as the American Sleep Apnea Association helped improve awareness. In addition, improved diagnostic methods such as polysomnography and home sleep tests have made identifying sleep apnea in its early stages easier. The increasing recognition of sleep apnea as a significant health concern drives more individuals to pursue PAP therapy, thus fueling market demand.
Market Concentration & Characteristics
The degree of innovation in the PAP devices industry is high. Manufacturers are introducing advanced features such as mobile app connectivity, improved comfort, and quieter operation. For instance, in January 2025, ResMed introduced the AirSense 11 CPAP device in India, adding features like Personal Therapy Assistant and Care Check-In along with AirView and myAir connectivity to support higher adherence and ease patients into sleep apnea treatment.
The industry has a moderate level of partnerships and collaborations. Companies partner with regional players to expand their market reach and incorporate innovative technologies. For instance, in February 2025, Koninklijke Philips N.V. partnered with Compumedics Ltd. to expand sleep solutions in Australia, supplying Somfit devices and electrodes to pharmacy-based sleep programs nationwide.
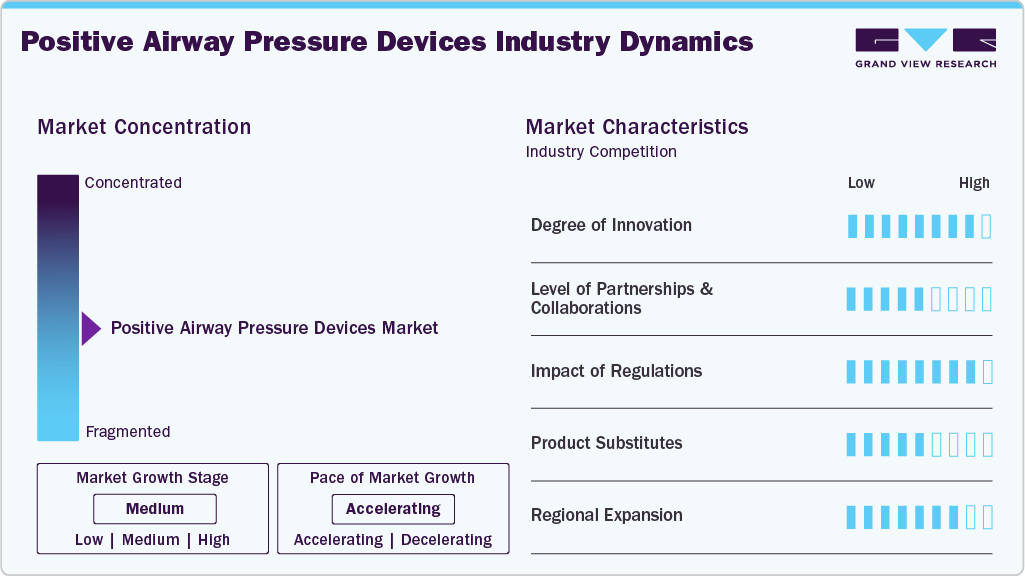
Regulations in the industry have a high impact, as strict standards ensure product safety, efficacy, and patient outcomes. Regulatory bodies such as the FDA and EMA closely evaluate new devices, with strong emphasis on clinical validation and real-world performance;
The presence of product substitutes is moderate in the PAP device market, particularly with the introduction of pharmacological treatments for obstructive sleep apnea. For instance, in December 2024, the FDA approved the first medication for treating obstructive sleep apnea. In addition, the emerging role of GLP-1 (glucagon-like peptide) receptor, which regulates respiratory function and has been linked to improved airway patency, presents a potential substitute for traditional PAP devices, particularly for patients who may not tolerate continuous positive airway pressure (CPAP) therapy. However, PAP devices remain essential for many patients, especially those who prefer non-invasive treatment options or have severe cases that require continuous airway pressure support.
Regional expansion is high, particularly in emerging markets such as Asia Pacific and Latin America. As awareness of sleep disorders increases, companies target these regions for growth opportunities. Factors such as urbanization, a growing middle-class population, and a higher prevalence of sleep apnea contribute to market growth. For instance, in November 2022, ResMed opened its advanced manufacturing facility in Singapore. This strategy helped the company expand its AirSense 11 device and mask production.
Impact of GLP-1 Medications on the Positive Airway Pressure (PAP) Devices Market
The introduction of GLP-1 receptor agonists, such as Eli Lilly's Zepbound, has shown promise in reducing the severity of obstructive sleep apnea (OSA) in individuals with obesity. Clinical studies indicate that these medications can significantly decrease the number of apnea events per hour, even when used alongside traditional continuous positive airway pressure (CPAP) therapy.
Despite these findings, the impact of GLP-1 drugs on the Positive Airway Pressure (PAP) devices market is expected to be nuanced. As per the Informa Markets, May 2024 studies, some analysts suggest that while GLP-1 therapies may reduce the incidence of OSA by promoting weight loss, they could also increase awareness and diagnosis of sleep apnea, potentially leading to higher demand for PAP devices.
Industry leaders like ResMed have observed that patients on GLP-1 medications exhibit a higher propensity to initiate and adhere to PAP therapy. This trend suggests that GLP-1 drugs may complement rather than replace existing sleep apnea treatments. For instance, in January 2025, following the U.S. Food and Drug Administration's approval of Zepbound. ResMed intensified its outreach to high-volume GLP-1 drug prescribers, emphasizing that PAP therapy continues to be the gold standard treatment.
“We are looking forward to addressing the educational gap on the prevalence and treatment of obstructive sleep apnea with continuing medical education or CME programs that we are aiming specifically at primary care physicians. And we're especially targeting those who are currently high-volume GLP-1 prescribers. They will be the frontline for the patients that pharmaceutical companies will attract as they ramp up their own consumer advertising throughout calendar year 2025 and beyond.”
-said Mick Farrell, chairman and CEO
Moreover, ResMed's research unveiled that as of January 2025, they track 1.2 million patients, indicating that individuals with prescriptions for both GLP-1 medications and PAP therapy are over 10% more likely to initiate PAP treatment. In addition, after one year, these patients demonstrate a more than 3% higher adherence rate to PAP therapy.
Product Insights
The Continuous Positive Airway Pressure (CPAP) devices segment dominated the market with a share of 57.4% in 2025. This can be attributed to the rising prevalence of sleep apnea, particularly obstructive sleep apnea (OSA), which has significantly increased the demand for CPAP devices as the primary treatment option. Technological advancements, such as auto-adjusting pressure settings, integrated humidifiers, and enhanced comfort features, are improving patient compliance and driving adoption. Moreover, the introduction of portable and travel-friendly CPAP devices, along with the integration of smart connectivity features for remote monitoring, is enhancing user experience and increasing market penetration.
Bi-level positive airway pressure (BiPAP) devices segment is anticipated to grow at the fastest rate of 5.94% over the forecast years, owing to the rising incidence of obstructive sleep apnea-associated comorbidities and the widespread adoption of PAP devices in disease management. BiPAP devices are the second line of treatment for patients who face difficulties using CPAP devices. BiPAP devices offer a more efficient breathing pattern and a variable/alternate airflow. The availability of technologically advanced PAP devices and favorable reimbursement policies are a few driving factors anticipated to strengthen the adoption and growth of BiPAP devices.
Application Insights
The Obstructive Sleep Apnea segment held the largest market share in 2025. The rising prevalence of Obstructive Sleep Apnea (OSA), which is estimated to affect millions worldwide, is an effective driver for this market. The condition occurs when the muscles in the throat relax excessively during sleep, causing airway obstruction and disrupting breathing patterns. PAP devices, particularly CPAP, provide a constant air flow to keep the airway open, improving sleep quality and preventing related health issues like heart disease, hypertension, and stroke. The growing awareness of OSA symptoms and the importance of treatment, coupled with advances in PAP technology, is driving the segment's growth.
The respiratory failure segment is driven by the increasing prevalence of conditions that cause respiratory insufficiency, such as central sleep apnea and other chronic lung diseases. Respiratory failure occurs when the lungs fail to provide adequate oxygen to the body or remove carbon dioxide, and PAP devices, especially BiPAP, play a critical role in managing these patients. BiPAP delivers two pressure levels: a higher pressure during inhalation and a lower pressure during exhalation, offering better support for individuals with respiratory failure. The segment is benefiting from the rising incidence of conditions such as obesity hypoventilation syndrome (OHS) and the aging population, which is more prone to respiratory disorders.
The Chronic Obstructive Pulmonary Disease (COPD) segment drives the demand for PAP devices. COPD, a group of lung diseases including emphysema and chronic bronchitis, is characterized by long-term respiratory issues, making it difficult for patients to breathe. As COPD is a leading cause of respiratory-related hospitalizations worldwide, the use of PAP devices, particularly BiPAP and CPAP, is typical for managing patients with COPD exacerbations. These devices help reduce the work of breathing and improve oxygenation by providing continuous or bilevel pressure to keep the airways open. The segment’s growth is influenced by the increasing prevalence of COPD, particularly in regions with high smoking rates or aging populations.
End Use Insights
The hospitals and clinics segment dominated the market in 2025, driven by the increasing prevalence of sleep apnea and the rising demand for professional diagnosis and treatment. These healthcare facilities are crucial for accurate diagnosis through polysomnography and other sleep studies, where PAP devices play a central role in therapy. Hospitals and sleep labs provide a controlled environment for sleep therapy initiation and monitoring, ensuring better patient compliance and treatment outcomes. The segment's growth is supported by advances in healthcare infrastructure, enabling more comprehensive sleep disorder services.
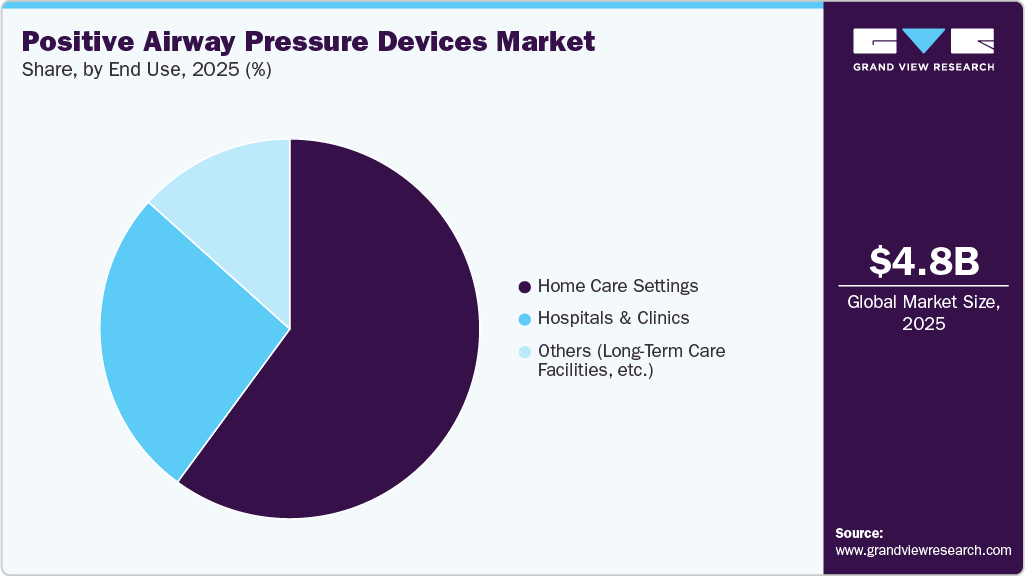
The home care settings segment for PAP devices is experiencing substantial growth, driven by the increasing preference for at-home healthcare and its convenience to patients. Many individuals with sleep apnea opt for home-based treatments as PAP devices become more user-friendly, portable, and equipped with remote monitoring capabilities. This segment benefits from the growing trend of telemedicine and digital health solutions, allowing healthcare providers to track patient progress and adjust therapy as needed remotely. The rising awareness of sleep apnea and the comfort of receiving treatment at home fuel the market.
Regional Insights
North America dominated the market and accounted for a 51.39% share in 2025. North America positive airway pressure devices market is witnessing substantial growth, driven by the increasing prevalence of respiratory conditions, such as chronic obstructive pulmonary disease (COPD), respiratory failure, and sleep apnea, which is driving the PAP devices market in the region. According to an article published by the American Lung Association in September 2024, over 35 million individuals in the U.S. suffer from chronic lung diseases, including COPD and asthma.
U.S. Positive Airway Pressure Devices Market Trends
The positive airway pressure devices market in the U.S. is encouraged by favorable healthcare reimbursement policies and rising healthcare spending, which makes treatment more accessible. In addition, the aging population in the U.S. is another factor driving the demand for PAP devices, as older adults are to suffer from sleep apnea, further driving the market growth. According to the Population Reference Bureau, Americans aged 65 and older are expected to grow from 58 million in 2022 to 82 million by 2050, representing a 47% increase. Hence, the proportion of this age group in the overall population is projected to rise from 17% to 23%.
Asia Pacific Positive Airway Pressure Devices Market Trends
The positive airway pressure devices market in Asia Pacific is growing due to increasing healthcare awareness and the rising prevalence of sleep apnea. Innovation in the market focuses on developing quieter, more comfortable machines with advanced features, including smart connectivity for remote monitoring. These technological advancements aim to enhance patient compliance and treatment efficacy, addressing diverse consumer needs across the region. For instance, in January 2025, ResMed announced the launch of AirSense 11 in India, a next-generation Continuous PAP (CPAP) device aimed at making it easier for individuals with Obstructive Sleep Apnea (OSA) to begin and maintain therapy.
China positive airway pressure devices market is expanding rapidly, driven by increasing awareness of sleep apnea and the rising adoption of advanced treatment options. A key trend is the continuous innovation in PAP devices, with manufacturers enhancing comfort through quieter machines, improved ergonomics, and the integration of advanced remote monitoring features. In addition, the expansion of healthcare services, including improved access to sleep apnea diagnosis, is fueling market growth. Another significant factor is China’s growing aging population, which is driving demand as older adults are more prone to sleep disorders.
The positive airway pressure devices market in India is growing due to rising awareness of sleep disorders and an increasing focus on healthcare. A study by AIIMS New Delhi, published by Bennett, Coleman & Co. in October 2023, reveals that 11% of Indian adults suffer from OSA, with men at higher risk. Approximately 104 million people are affected, including 47 million with moderate to severe OSA. The market is further driven by expanding healthcare infrastructure and improved access to sleep disorder diagnosis.
Europe Positive Airway Pressure Devices Market Trends
Favorable government initiatives for respiratory care are anticipated to propel the positive airway pressure devices market in Europe due to the rising prevalence of sleep apnea and rising prevalence of COPD among the European population, attributed to aging demographics, increasing obesity rates, and lifestyle factors such as smoking and alcohol consumption in European countries. For instance, in June 2025, the WHO European Region report highlighted that chronic respiratory diseases (CRDs), particularly COPD and asthma, remain a major health burden despite declining mortality. Nearly 80% of CRD deaths are due to COPD, and cases are projected to rise by 23% globally by 2050, disproportionately affecting women and people in low- and middle-income countries, with regional productivity losses estimated at USD 20.7 billion.
High demand for PAP devices is anticipated to drive the market owing to the rising prevalence of chronic respiratory diseases in the UK. According to an article published by the Sleep Apnea Trust in 2023, up to 10 million people in the UK suffer from the most prevalent type of OSA. Among these, around 4 million people suffer from either moderate or severe OSA. In November 2024, an article published in BMJ Open reported that sleep-disordered breathing affects approximately 1.6 million individuals in the UK, highlighting the significant clinical need for effective PAP therapy. The study reinforced that positive airway pressure treatment substantially improves quality of life and health outcomes when the interface is optimally fitted.
The positive airway pressure devices market in Germany is driven by the rising number of sleep apnea patients and the availability of reimbursement coverage. In addition, enhanced patient compliance and a high prevalence of respiratory diseases are expected to have a positive impact on the CPAP devices market. For instance, in April 2024, a study in Sleep analyzed 146,227 OSA patients in Germany, mostly older males with comorbidities such as hypertension and diabetes. PAP therapy was the main treatment. These findings underscore the importance of improving patient adherence and streamlining the diagnostic process. Continued advancements in CPAP technology and patient support programs could help bridge these gaps and enhance treatment outcomes.
Latin America Positive Airway Pressure Devices Market Trends
The positive airway pressure devices market in Latin America is gaining momentum due to improved healthcare infrastructure and increased awareness of sleep apnea. The rising prevalence of the condition, particularly among the aging population, is a key driver for the market. Manufacturers are introducing affordable, user-friendly devices to cater to the region's diverse needs. Expansion of healthcare access, particularly in rural areas, is enhancing diagnosis and treatment options.
Middle East & Africa Positive Airway Pressure Devices Market Trends
The positive airway pressure devices market in the Middle East & Africa is characterized by expanding healthcare facilities, particularly in the Gulf Cooperation Council (GCC) countries, with a growing emphasis on healthcare modernization. This expansion is increasing the availability of sleep disorder diagnoses and treatments. Innovation in device design, including enhanced portability and quieter operation, is also becoming a key trend. Moreover, the aging population and rising rates of chronic conditions such as sleep apnea are contributing to increased demand for PAP devices. Companies are pursuing strategic partnerships to expand their regional presence and enhance technology integration. For instance, in March 2021, Philips partnered with the Middle East Healthcare Company (MEAHCO) to introduce advanced sleep disorder management services in Saudi Arabia via the Saudi German Health Group.
Key Positive Airway Pressure Devices Companies Insights
Key players focus on product innovation, incorporating features such as enhanced comfort, noise reduction, portability, and connectivity with mobile apps for remote monitoring. In addition, strategic collaborations, acquisitions, and regional expansions help maintain a strong foothold in developed and emerging markets. For instance, in February 2021, Philips and the Dutch SAZ group entered a strategic partnership to offer a complete suite of monitoring, observation, and self-management services to patients diagnosed with obstructive sleep apnea. The market is characterized by increasing investments in research and development to improve PAP devices' efficacy and user experience.
Key Positive Airway Pressure Devices Companies:
The following key companies have been profiled for this study on the positive airway pressure market.
- Koninklijke Philips N.V.
- SEFAM
- Trudell Medical (Vyaire)
- Löwenstein Medical UK Ltd
- Transcend Inc. (Somnetics International, Inc.)
- Fisher & Paykel Healthcare Limited
- ResMed
- Drive DeVilbiss Healthcare
- React Health (3B Medical)
- BMC Medical
- Armstrong Medical Inc.
- Wellell (Apex Medical)
- Breas Medical
Exit Strategies of Key Market Players
-
Koninklijke Philips N.V.
In June 2021, Philips recalled certain BiPAP, CPAP, and mechanical ventilator devices due to potential health hazards linked to the polyester-based polyurethane (PE-PUR) sound-dampening foam utilized in these products. Since the recall, Philips, the FDA, and other organizations have provided various resources.
In March 2024, Philips Respironics announced that it would discontinue several products, including the NightBalance device for positional sleep apnea therapy and the DreamStation Go travel CPAP machine. In addition, the company reached a consent agreement with the U.S. Department of Justice, which is anticipated to halt the sale of all Respironics CPAP devices for the next few years.
-
Fisher & Paykel Healthcare Limited.
In April 2024, Fisher & Paykel Healthcare decided to stop selling its F&P SleepStyle CPAP device in the U.S. The SleepStyle CPAP, designed to treat OSA, was positioned as a convenient and user-friendly option for patients seeking at-home therapy. Due to regulatory hurdles, market challenges, and evolving company strategies, Fisher & Paykel discontinued this product in the U.S. market. This move comes amid increasing competition from major players such as ResMed and Philips, major players in the PAP device market with highly advanced and widely adopted products.
-
Drive DeVilbiss Healthcare
Drive DeVilbiss Healthcare stopped producing its DV5 CPAP units in August 2021, just a few months after the large-scale recall of Philips CPAP devices. On June 29, 2021, the company acknowledged that their machines contained polyester-based polyurethane foam. The company chose not to initiate a voluntary recall, as they had not received any reports of health issues linked to the devices.
This decision was driven by challenges such as raw material shortages, difficulties sourcing necessary components, and rising parts prices. In light of these issues, Drive DeVilbiss shifted its focus to its leading oxygen products, responding to the increased demand during the pandemic.
Recent Developments
-
In December 2025, SleepRes, Inc. received FDA 510(k) clearance for its Kricket PAP device, powered by proprietary Kairos Positive Airway Pressure (KPAP) technology, for treating obstructive sleep apnea in patients weighing over 66 lbs.
-
In May 2025, ResMed acquired VirtuOx, an independent diagnostic testing facility, to expand virtual care, improve access to sleep and respiratory diagnostics, and enhance the digital experience for patients and providers.
-
In June 2024, React Health, a manufacturer and distributor of sleep & respiratory devices, introduced the IntelliPAP feature for its V+Pro, V*Home, V+C, and VC+Pro ventilators. This feature enables physicians to automatically adjust PEEP levels during noninvasive ventilation for patients with respiratory insufficiency and OSA in response to detected sleep-disordered breathing events.
“React Health is committed to advancing healthcare solutions that contribute to improved patient outcomes and operational efficiency. The most recent launch of IntelliPAP, along with the new E0468 HCPCS code for our V+C ventilator with integrated cough assist (ICAT™), further reflects that commitment to our partners and patients.”
-Jeff Ward, EVP of Ventilation for React Health.
-
In February 2024, ResMed introduced its AirCurve 11 series bilevel devices, offering inspiratory and expiratory pressure support. These devices integrate digital technology, simplifying sleep apnea treatment for healthcare providers while helping patients initiate and maintain effective therapy.
“Patient care is our top priority, and that includes their comfort and compliance with PAP therapy. We’ve seen patient compliance improve from 70% to 87% through the use of coaching features and the ability to track and view their nightly sleep data via MyAir and AirView, so we’ve incorporated these digital health applications into the new AirCurve11 series to enable maximum comfort and support throughout the therapy experience.”
-ResMed chief medical officer Carlos M. Nunez, MD
-
In July 2024, the FDA issued a final rule on October 19, 2018, reclassifying positive airway pressure (PAP) delivery systems from Class III to Class II, though they remain available by prescription only.
Positive Airway Pressure Devices Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 5.09 billion
Revenue forecast in 2033
USD 7.40 billion
Growth rate
CAGR of 5.48 % from 2026 to 2033
Actual data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Volume in (000’ Units); revenue in USD billion/million, and CAGR from 2026 to 2033
Report coverage
Volume & revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; Spain; France; Italy; Sweden; Denmark; Norway; Asia Pacific; Japan; China; India; Australia; South Korea; Thailand; Latin America; Brazil; Argentina; Middle East & Africa; South Africa; Saudi Arabia; UAE
Key companies profiled
Koninklijke Philips N.V.; SEFAM; Trudell Medical (Vyaire); Löwenstein Medical UK Ltd; Transcend Inc. (Somnetics International, Inc.); Fisher & Paykel Healthcare Limited; ResMed; Drive DeVilbiss Healthcare; React Health (3B Medical); BMC Medical; Armstrong Medical Inc.; Wellell (Apex Medical); Breas Medical
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Positive Airway Pressure Devices Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global positive airway pressure devices market report based on product, application, end use, and region.

-
Product Outlook (Volume, 000` Units; Revenue, USD Million, 2021 - 2033)
-
CPAP
-
Connected
-
Bluetooth-Connected CPAP
-
Wi-Fi & Cellular Connected CPAP
-
Hybrid-Connected
-
-
Non-Connected
-
-
APAP
-
Bi-PAP
-
-
Application Outlook (Volume, 000`Units; Revenue, USD Million, 2021 - 2033)
-
Obstructive Sleep Apnea
-
Respiratory Failures
-
COPD
-
-
End Use Outlook (Volume, 000`Units; Revenue, USD Million, 2021 - 2033)
-
Home Care Settings
-
Hospitals & Clinics
-
Others (Long-Term Care Facilities, etc.)
-
-
Regional Outlook (Volume, 000`Units; Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Sweden
-
Denmark
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa
-
Saudi Arabia
-
South Africa
-
UAE
-
-
Frequently Asked Questions About This Report
b. Key factors that are driving the market growth include the rising sleep apnea awareness, advanced treatment adoption, continuous device innovation, improved ergonomics, remote monitoring features, expanding healthcare services, better diagnosis access, and an aging population more prone to sleep disorders.
b. Some key players operating in the positive airway pressure devices market include Koninklijke Philips N.V., SEFAM, Trudell Medical (Vyaire), Löwenstein Medical UK Ltd, Transcend Inc. (Somnetics International, Inc.), Fisher & Paykel Healthcare Limited, ResMed, Drive DeVilbiss Healthcare, React Health (3B Medical), BMC Medical, Armstrong Medical Inc., Wellell (Apex Medical), and Breas Medical
b. The global positive airway pressure devices market size was estimated at USD 4.82 billion in 2025 and is expected to reach USD 5.09 billion in 2026.
b. The global positive airway pressure devices market is expected to grow at a compound annual growth rate of 5.48% from 2026 to 2033 to reach USD 7.40 billion by 2033.
b. CPAP devices dominated the positive airway pressure devices market with a share of 57.4% in 2025. This is attributable to the increasing adoption of these devices due to their low cost as compared to other PAP devices.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










