- Home
- »
- Clinical Diagnostics
- »
-
qPCR Instruments Market Size, Share, Industry Report 2033GVR Report cover
![qPCR Instruments Market Size, Share & Trends Report]()
qPCR Instruments Market (2026 - 2033) Size, Share & Trends Analysis Report By Instrument (7500, QuantStudio, CFX, SmartCycler, GeneXpert, StepOne/StepOne Plus, LightCycler, Cobas), By Type, By Region, And Segment Forecasts
- Report ID: GVR-4-68038-339-3
- Number of Report Pages: 156
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
qPCR Instruments Market Summary
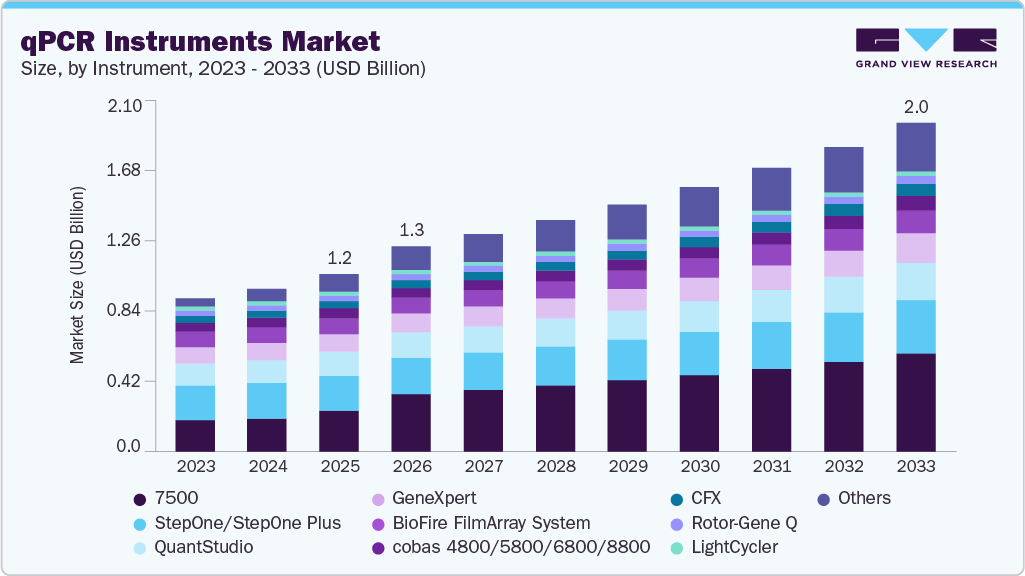
The global qPCR instruments market size was estimated at USD 1.19 billion in 2025 and is expected to reach USD 2.02 billion by 2033, growing at a CAGR of 7.0% from 2026 to 2033. The rising adoption of molecular diagnostics for infectious disease detection, increasing demand for rapid and high sensitivity testing in clinical and research settings, and the expanding use of qPCR in oncology, genetic testing, and precision medicine are identified as the major growth drivers for this market.
Key Market Trends & Insights
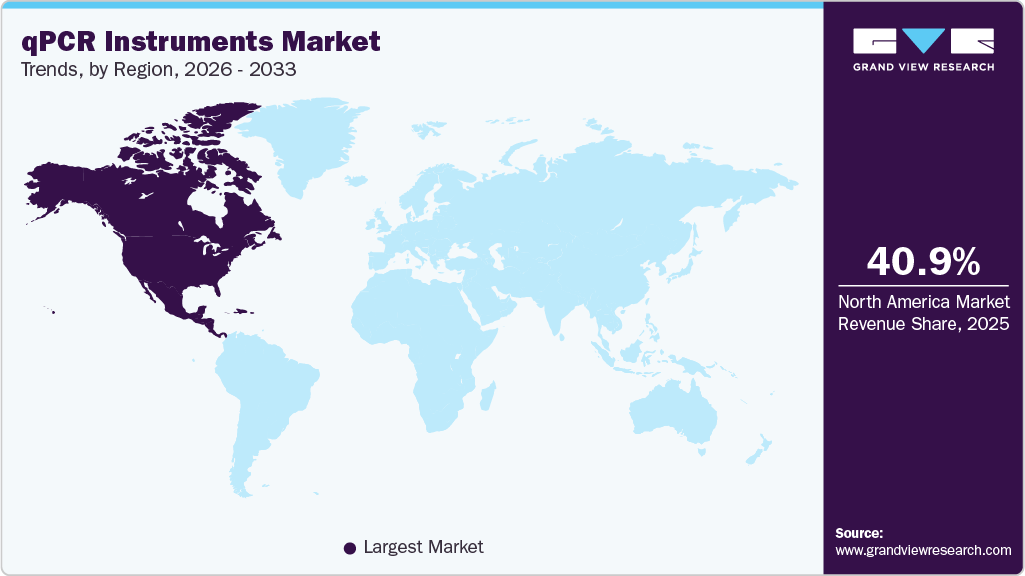
- North America qPCR instruments market held the largest share in 2025.
- The U.S. dominated the market in 2025, driven by expanding molecular diagnostics, infectious disease testing, oncology research, technological advancements, and increasing adoption in clinical laboratories.
- By instrument, the QuantStudio segment held the largest revenue share of 20.98% in 2025.
- By type, the standalone qPCR instruments segment held the largest share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1.19 Billion
- 2033 Projected Market Size: USD 2.02 Billion
- CAGR (2026-2033): 7.0%
- North America: Largest market in 2025
The growth has also been supported by advancements in automated and high throughput qPCR platforms, increased public and private funding for life sciences research, and the continued integration of qPCR technologies into public health surveillance and epidemic preparedness programs worldwide.The rising prevalence of respiratory viruses such as SARS-CoV-2, influenza, and RSV has kept molecular diagnostics central to healthcare infrastructure planning. For instance, in February 2025, Thermo Fisher Scientific announced U.S. FDA approval for Applied Biosystems TaqPath Flu A, Flu B, COVID-19, RSV Select Panel. This PCR-based multiplex test ensures reliable detection and differentiation of these prevalent viruses. It identifies co-infections, underlining the ongoing importance of qPCR-based assays for monitoring seasonal outbreaks. These products increase the utilization of qPCR platforms in clinical labs. They allow a single instrument to run multiple clinically relevant assays. The result is higher throughput and more efficient use of qPCR instruments across diagnostic workflows. These further drive instrument demand in markets focused on public health and clinical readiness.
The demand for qPCR equipment and consumables was heightened during the SARS-CoV-2 pandemic. The spike in incidence of COVID-19 added significantly to the number of preventive screenings. This was attributed to the fact that the number of COVID-19 patients cannot be ascertained without proper screening, owing to the large number of asymptomatic cases.
Launch of new products is further expected to change certain market dynamics, as it can increase the threat of internal substitution. Furthermore, increasing preference of consumers towards updated and advanced products is creating a demand for technologically advanced products, such as the Mic qPCR system with Bluetooth connectivity and multiple devices that can be connected to the computer simultaneously to run multiple sample sets.
qPCR is extensively used for gene expression analysis, SNP genotyping, copy number variation studies, and validation of next-generation sequencing (NGS) data in genomics research. The growing emphasis on precision medicine and functional genomics has increased the need for accurate nucleic acid quantification, reinforcing qPCR’s role as a standard laboratory tool. This trend is reflected in ongoing product innovation by major industry players. For instance, in July 2025, Bio-Rad Laboratories expanded its PCR technology portfolio with the launch of advanced Droplet Digital PCR platforms, strengthening its molecular analysis offerings for genomics and oncology research. While digital PCR complements qPCR, such developments underscore the broader expansion of molecular quantification technologies driven by genomics research growth.
Prevalence of chronic diseases, infectious diseases, and genetic disorders such as Alzheimer’s, Turner syndrome, & Parkinson’s, has increased rapidly, which is expected to fuel the market. According to CDC, chronic diseases such as cancer, diabetes, kidney diseases, and respiratory diseases including asthma, are accountable for 7 in 10 deaths in the U.S. each year. Faster detection of diseases with PCR can allow for timely & appropriate treatment.
Increasing adoption of qPCR technologies is expected to propel this market. Adoption of qPCR techniques is increasing, owing to their accuracy, automation, precise results, real-time quantification, and sensitivity. qPCR offers a wide range of input nucleic acids, making it an effective and useful technology in research applications.
Government initiatives and public-private partnerships are accelerating the adoption of molecular diagnostics in emerging economies. Investments in national reference laboratories, public health programs, and pandemic preparedness have expanded PCR-based testing beyond urban centers. International funding agencies and global health organizations are supporting the deployment of molecular diagnostic platforms. For instance, in May 2025, Siemens Healthineers invested USD 150 million in the U.S., expanding manufacturing, supply depots, and a 60,000 sq. ft. Experience Center, creating jobs and enhancing healthcare innovation nationwide. These investments further drive qPCR instrument penetration in resource-limited settings.
Instruments Insights
QuantStudio segment accounted for the largest market share of 20.98% in 2025. The segment is anticipated to maintain its leading position over the forecast period due to its widespread adoption in clinical diagnostics and research laboratories, high sensitivity and reproducibility, compatibility with a broad range of assays, and increasing demand for reliable, high throughput real time PCR systems in infectious disease testing, genomics, and applied research applications.
The Roche LightCycler platform represents one of the earliest and most established families of real-time PCR instruments positioned across both research-use-only (RUO) and in-vitro diagnostic (IVD) applications. The LightCycler portfolio, including systems such as the LightCycler 1.x/2.0, LightCycler 480, and later Dx-configured variants, played a foundational role in the adoption of qPCR in clinical diagnostics and molecular research.
Early market penetration was driven by Roche’s strong diagnostics footprint, proprietary assay chemistry, and early regulatory acceptance in Europe and select global markets. The installed base expanded rapidly within hospital laboratories, reference centers, and public health institutions, particularly in regions where Roche maintained strong clinical relationships. The LightCycler 480 further accelerated adoption by introducing higher throughput (96- and 384-well formats) and improved optical performance.
The QIAGEN Rotor-Gene Q is a bench-top real-time PCR platform known for its rotary thermal cycling design, which delivers high thermal uniformity and reproducibility. Introduced in 2012 as the successor to earlier Rotor-Gene models, the system was positioned across research-use-only (RUO) and diagnostic/IVD applications, particularly in laboratories prioritizing assay precision over high throughput. Adoption was driven by its analytical robustness, compact footprint, and strong compatibility with QIAGEN’s sample-to-answer workflows, including nucleic acid extraction and assay kits.
Type Insights
Standalone qPCR instruments segment accounted for the largest market share of 57.49% in 2025 and is expected to grow at the fastest CAGR over the forecast period. Key growth drivers include increasing molecular research activity, expansion of precision medicine, biomarker validation studies, and decentralized mid-complexity testing in hospital labs. Technological trends include improved thermal uniformity, multiplexing capability, digital connectivity, cloud-based data management, and compact benchtop formats. Representative platforms include the QuantStudio 5 Real-Time PCR System, CFX96 Real-Time PCR Detection System, and LightCycler 480, which are commonly used for infectious disease research, oncology assays, gene expression analysis, and genotyping workflows.
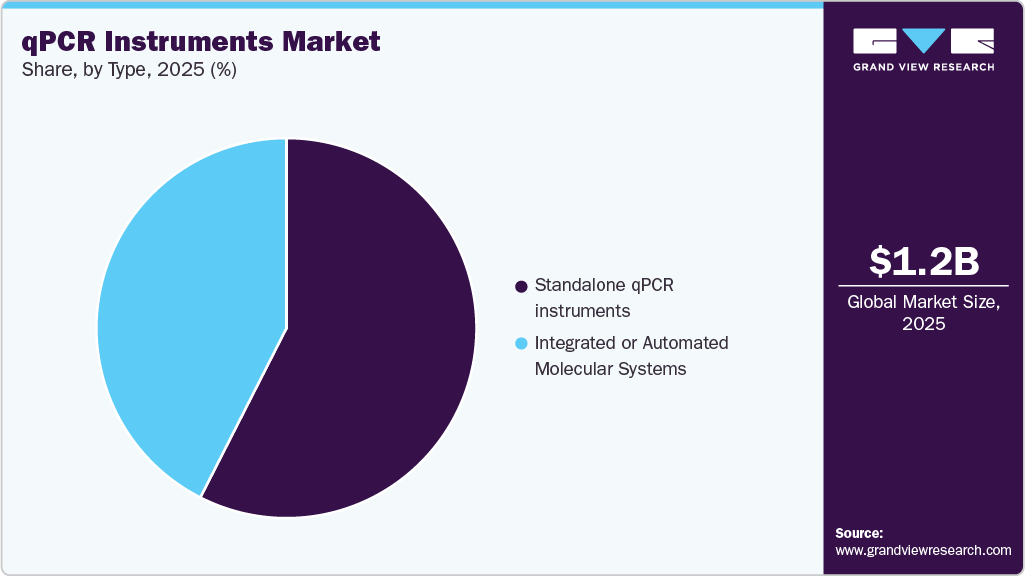
The integrated or automated molecular systems held a significant share in 2025, and the market growth is driven by demand for rapid turnaround in infectious disease diagnostics, decentralization of molecular testing to near-patient settings, CLIA-waived expansion, and syndromic panel adoption. Current trends include cartridge-based automation, high-throughput consolidation in centralized labs, and expansion into antimicrobial resistance and respiratory panels. Prominent examples include the GeneXpert System, cobas 6800 System, and BioFire FilmArray, which are widely implemented in hospital laboratories and reference labs for standardized clinical testing workflows.
Regional Insights
North America qPCR instruments market accounted for the largest revenue share of 40.95% in 2025 and is expanding at a CAGR of 6.8% due to rising demand for rapid, accurate molecular diagnostics across the United States, Canada, and Mexico, especially for respiratory infections, pandemic preparedness, and population surveillance. Public health agencies and clinical laboratories increasingly rely on real time PCR and RT qPCR platforms to detect influenza, SARS CoV 2, and other pathogens with high sensitivity.
U.S. qPCR Instruments Market Trends
The qPCR instruments market in the U.S. has experienced significant growth due to increasing demand for rapid, accurate molecular diagnostics, particularly for respiratory infections and pandemic preparedness. For instance, in September 2025, the Centers for Disease Control and Prevention (CDC) reported that its Influenza SARS-CoV‑2 (Flu SC2) Multiplex Assay was developed as a real-time reverse-transcription polymerase chain reaction (rRT‑PCR) test capable of detecting and differentiating influenza A, influenza B, and SARS‑CoV‑2 from respiratory specimens.
Canada qPCR instruments market has expanded significantly due to increasing demand for sensitive and high-throughput molecular diagnostics, particularly for pathogen detection in public health and environmental monitoring.
Europe qPCR Instruments Market Trends
The qPCR instruments market in the Europe is expanding due to increasing investment in high-throughput molecular diagnostics, public health surveillance, and outbreak preparedness. In the United Kingdom, government-led initiatives such as the December 2025 UK Health Security Agency contract of £267,606.33 with Thermo Fisher Scientific for 96‑well real-time PCR instruments reflect strong support for mobile and field-deployable testing.
qPCR Instruments Market in the UK has experienced growth due to increasing investment in advanced molecular diagnostics and rapid pathogen detection infrastructure. The government-led procurement, coupled with advanced assay development, illustrates how regulatory support, high specificity, and operational demands collectively propel the adoption of qPCR instruments, strengthening the market for both equipment and related consumables across the United Kingdom.
Germany qPCR instruments market has grown significantly due to increasing demand for high-throughput, multiplex molecular diagnostics for respiratory viruses. The adoption of high-precision PCR technologies has further stimulated market growth by supporting detailed viral quantification and surveillance.
qPCR instruments market in Francehas grown steadily due to increasing demand for high-precision molecular diagnostics in clinical and infectious disease applications. The adoption of RT‑qPCR over conventional methods has further strengthened market growth by offering enhanced sensitivity and accuracy, especially for low pathogen loads.
Asia Pacific qPCR Instruments Market Trends
qPCR instruments market in APAC is expanding due to rising adoption of molecular diagnostics across healthcare, research, and public health sectors, driven by epidemic preparedness and routine clinical testing. In Japan, increasing reliance on high-throughput, sensitive qPCR systems enable rapid detection of viral pathogens, supporting clinical decision-making and national surveillance.
Japan qPCR Instruments Market is growing due to the increasing reliance on molecular diagnostics for epidemic preparedness and routine clinical testing. The country’s public health and research institutions prioritize rapid and precise detection of viral pathogens to guide timely interventions and inform public health strategies.
qPCR instruments market in China has been experiencing significant growth, driven primarily by the expanding application of molecular diagnostics in public health monitoring, especially in the post-COVID-19 era. The surge in demand for high-throughput and accurate testing solutions has been fueled by nationwide initiatives such as the Chinese Urban Wastewater Surveillance System, which was officially initiated in December 2022.
India qPCR instruments market has been experiencing significant growth due to the increasing adoption of molecular diagnostic tools for infectious disease monitoring, particularly in response to the COVID-19 pandemic. Given India’s national context of over 45 million COVID-19 cases and 533,300 deaths, the need for precise, rapid, and scalable diagnostic tools has directly fueled the India qPCR Instruments Market, making it essential for ongoing public health monitoring, research, and clinical applications.
Latin America qPCR Instruments Market Trends
qPCR instruments market in Latin America is expanding due to increasing demand for rapid, sensitive, and high-throughput molecular diagnostic technologies across clinical, research, and environmental applications. In Brazil, the rising prevalence of infectious diseases such as rotavirus, norovirus, and SARS‑CoV‑2 has driven adoption of real-time PCR systems in hospitals, public health laboratories, and research centers.
MEA qPCR Instruments Market Trends
qPCR instruments market in MEA is expanding due to increasing demand for rapid, sensitive, and accurate molecular diagnostics across clinical, environmental, and research applications. Rising prevalence of infectious diseases, including SARS‑CoV‑2, dengue, and rotavirus, has accelerated the adoption of real-time PCR technologies in hospitals, public health laboratories, and veterinary facilities. Studies from South Africa, Saudi Arabia, UAE, and Kuwait highlight the critical role of qPCR instruments in detecting viral RNA, monitoring variant prevalence, and guiding public health interventions.
Key qPCR Instruments Company Insights
Some key players in the qPCR instruments market included Thermo Fisher Scientific, Inc., F. Hoffmann-La Roche Ltd., Bio-Rad Laboratories, Inc., Danaher, QIAGEN, Agilent Technologies, Inc., Abbott, BIOMÉRIEUX, Quantabio, Azure Biosystems Inc., and Bio Molecular Systems. Key players were adopting inorganic growth strategies, such as mergers, collaborations, and strategic partnerships, to expand their product portfolios and strengthen their technological capabilities.
Real-time PCR has various significant external substitutes, including advanced instruments such as dPCR and newly developed nanoplasmonic PCR. Key players are making use of their existing consumer pool in the region to provide additional products and generate more business. This strategy is useful for brands that have already built trust in the market.
Key qPCR Instruments Companies:
The following key companies have been profiled for this study on the qpcr instruments market.
- Thermo Fisher Scientific, Inc.
- F. Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc
- Danaher Corporation
- Qiagen
- Agilent Technologies, Inc.
- Abbott
- bioMérieux SA
- Quantabio
- Azure Biosystems, Inc.
- Bio Molecular Systems
qPCR Instruments Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1.26 billion
Revenue forecast in 2033
USD 2.02 billion
Growth rate
CAGR of 7.0% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD Million/Billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Instruments, type, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; China; India; Japan; Australia; South Korea; Brazil; Argentina; South Africa; UAE; Saudi Arabia; Kuwait
Key companies profiled
Thermo Fisher Scientific, Inc.; F. Hoffmann-La Roche Ltd.; Bio-Rad Laboratories, Inc.; Danaher Corporation; Qiagen; Agilent Technologies, Inc.; Abbott; bioMérieux SA; Quantabio; Azure Biosystems, Inc.; Bio Molecular Systems
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global qPCR Instruments Market Report Segmentation
This report forecasts revenue growth at the global, regional, and country levels and provides an analysis of the latest industry trends and opportunities in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global qPCR instruments market report based on instruments, type, and region:

-
Instruments Outlook (Revenue, USD Million, 2021 - 2033)
-
7500
-
QuantStudio
-
StepOne/StepOne Plus
-
LightCycler
-
Cobas 4800/5800/6800/8800
-
GeneXpert
-
Rotor-Gene Q
-
BioFire FilmArray Systems
-
BMS Mic System
-
Cobas Z480
-
CFX
-
Others
-
-
Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Standalone qPCR Instruments
-
Integrated/Automated Molecular Systems
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global qPCR instruments market size was estimated at USD 1.0 billion in 2022 and is expected to reach USD 1.02 billion in 2023.
b. The global qPCR instruments market is expected to grow at a compound annual growth rate of 7.0% from 2024 to 2030 to reach USD 1.64 billion by 2030.
b. North America dominated the global qPCR instruments market with a share of 42.73% in 2022. This is attributable to growing regulatory support and increasing launches of novel products. The large number of instruments installed in North America, as compared to other regions, demonstrates its high diagnostic capacity.
b. Some key players operating in the global qPCR instruments market include Thermo Fisher Scientific, Inc., F. Hoffmann-La Roche Ltd., Bio-Rad Laboratories, Inc., Danaher Corporation, Qiagen, Agilent Technologies, Inc., Abbott, bioMérieux SA, Quantabio, Azure Biosystems, Inc., BioMolecular Systems.
b. Key factors that are driving the qPCR instruments market growth include the growing prevalence of diseases, technological advancements, increasing demand for qPCR instruments, and increased focus on diagnostics attributed to the spread of COVID-19.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










