- Home
- »
- Clinical Diagnostics
- »
-
Rare Disease Genetic Testing Market Size Report, 2033GVR Report cover
![Rare Disease Genetic Testing Market Size, Share & Trends Report]()
Rare Disease Genetic Testing Market (2026 - 2033) Size, Share & Trends Analysis Report By Disease (Neurological Disorders), By Technology (Next Generation Sequencing, Array Technology, PCR-based Testing), By Specialty, By End Use, By Region, And Segment Forecasts
- Report ID: GVR-4-68038-695-0
- Number of Report Pages: 120
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Interactive Charts
- Methodology
- Download FREE Sample
-
Download Sample Report
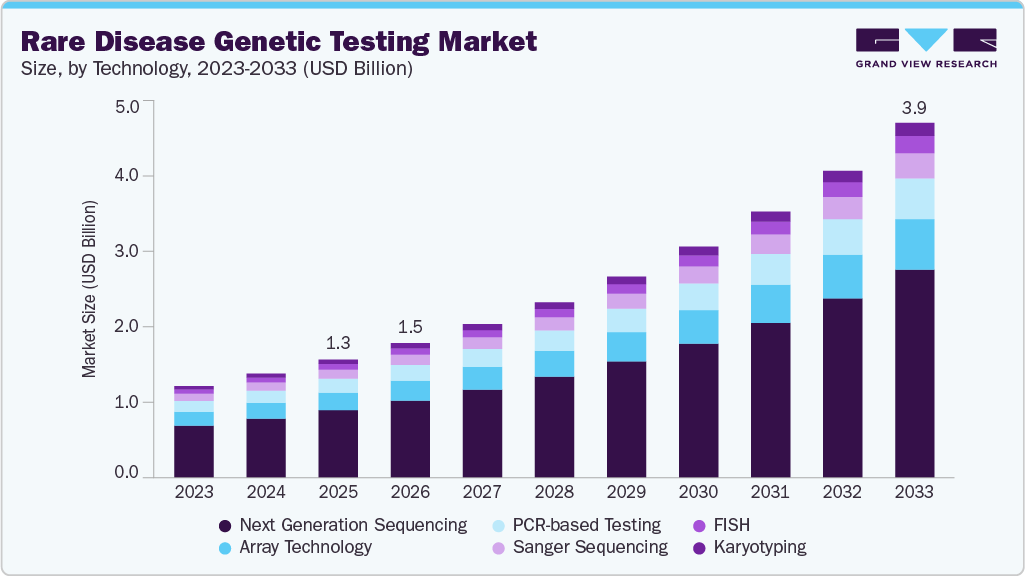
Rare Disease Genetic Testing Market Summary
The global rare disease genetic testing market size was estimated at USD 1,293.15 million in 2025 and is projected to reach USD 3,883.76 million by 2033, growing at a CAGR of 14.87% from 2026 to 2033. The market is driven by advancements in next-generation sequencing (NGS), increased awareness of rare genetic disorders, and rising demand for personalized medicine.
Key Market Trends & Insights
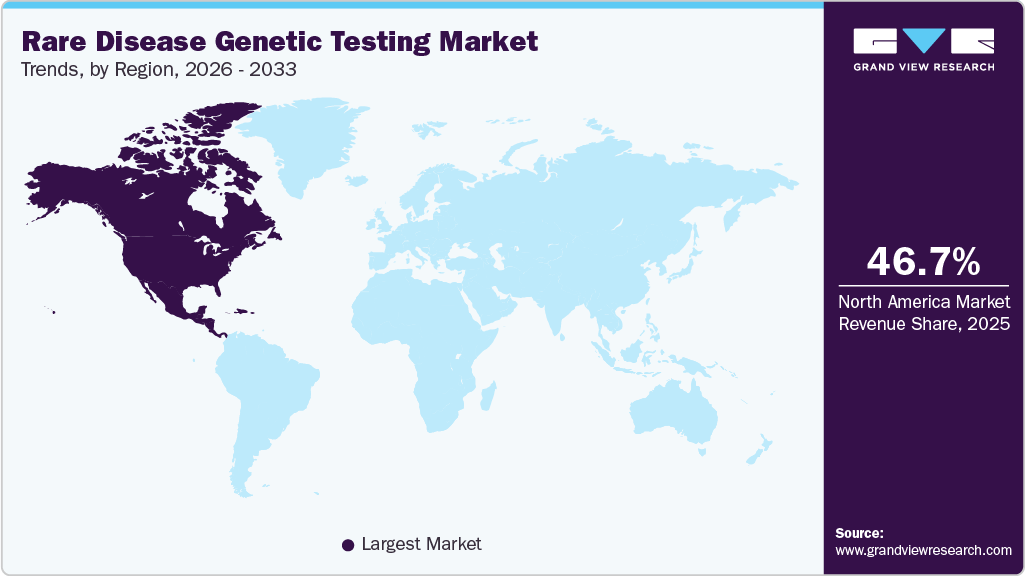
- The North America rare disease genetic testing market dominated the global market with a share of 46.73% in 2025.
- Based on disease, the neurological disorders segment held the largest share of 29.09% in 2025 and is expected to continue the trend during the forecast period.
- Based on technology, the Next Generation Sequencing segment held the largest share of 57.13% in 2025.
- Based on specialty, the molecular genetic tests segment dominated the market for rare disease genetic testing market and accounted for the largest revenue share of 68.43% in 2025.
- Based on end use, the diagnostics laboratories segment dominated the market for rare disease genetic testing market and accounted for the largest revenue share of 46.39% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 1,293.15 Million
- 2033 Projected Market Size: USD 3,883.76 Million
- CAGR (2026-2033): 14.87%
- North America: Largest market in 2025
In addition, strict regulations from various government bodies requiring alcohol and substance testing for safety reasons, along with increased initiatives to monitor and address substance abuse, are anticipated to further enhance market growth.The expansion and maturation of rare disease patient registries have emerged as a critical driver for the growth of the rare disease genetic testing market. These registries systematically aggregate phenotypic data, longitudinal clinical histories, genomic sequencing results, and patient-reported outcomes, thereby creating a centralized, structured data ecosystem. By transforming fragmented clinical information into cohesive datasets, registries enable clinicians and diagnostic laboratories to identify genotype-phenotype correlations more effectively, refine variant interpretation, and improve diagnostic accuracy.
This development is particularly significant given the scale and complexity of rare diseases, which affect over 300 million individuals globally, with approximately 25 to 30 million cases in the United States alone. Despite advances in diagnostics, the average diagnostic journey still exceeds six years, with only around 40% of patients receiving a confirmed diagnosis. Moreover, nearly 80% of rare diseases are genetic in origin, and about half of diagnosed cases occur in children, highlighting the urgent need for efficient and accurate genetic testing solutions.
The continuous decline in genome sequencing costs represents another major driver accelerating the growth of the rare disease genetic testing market. Over the past two decades, sequencing has evolved from an extremely expensive and time-intensive process-costing nearly USD 3 billion per genome in the early 2000s to a highly accessible technology, with costs projected to reach approximately USD 200 per genome by 2025. This dramatic reduction has been driven by advances in next-generation sequencing technologies and by increasing competition among key industry players, such as Illumina and Ultima Genomics.
Lower sequencing costs have far-reaching implications beyond affordability. They enable laboratories to scale up testing volumes while simultaneously investing in improvements across the diagnostic workflow. On the upstream side, laboratories are enhancing sample processing capabilities through faster extraction techniques and streamlined workflows. Downstream, there is increased investment in advanced bioinformatics, including automated variant calling, continuous reanalysis pipelines, and integrated clinical decision support systems.
Advancements in next-generation sequencing (NGS) technologies are playing a pivotal role in strengthening the capabilities and adoption of genetic testing for rare diseases. NGS enables high-throughput, comprehensive analysis of large genomic regions, enabling the identification of disease-causing variants with greater speed and accuracy than traditional diagnostic methods. Technologies such as whole exome sequencing (WES) and whole genome sequencing (WGS) have become essential tools, particularly for patients with complex or undiagnosed conditions where conventional approaches fail to provide answers.
Ongoing innovations in sequencing chemistry, automation, and bioinformatics are further enhancing the performance of NGS platforms. These improvements are reducing turnaround times, increasing diagnostic yield, and facilitating the integration of genomic testing into routine clinical practice. In 2025, companies such as PacBio have expanded the clinical adoption of long-read sequencing technologies, such as HiFi sequencing, which enable more accurate detection of structural variants and complex genomic regions that are often missed by short-read sequencing methods. Similarly, Element Biosciences has advanced high-throughput sequencing with its AVITI platform, enabling scalable, cost-efficient genomic testing in clinical laboratories.
Several industry leaders are actively expanding their offerings in this space
-
March 2025 - MyOme, a frontrunner in clinical whole-genome analysis and polygenic risk assessment, launched its first rare disease diagnostic product at the American College of Medical Genetics and Genomics (ACMG) Annual Meeting. By leveraging WGS, MyOme aims to increase diagnostic accuracy and improve patient access to genetic testing.
-
February 2025 - MedGenome introduced the #CarefortheRare campaign ahead of Rare Disease Day, focusing on raising awareness about undiagnosed inherited diseases and the importance of early genetic testing. The campaign featured a documentary highlighting the emotional and financial impact of rare diseases on families.
-
August 2024 - Ambry Genetics, a subsidiary of REALM IDx, Inc., launched ExomeReveal, a multiomic exome sequencing test that integrates RNA analysis to enhance rare disease detection. Unlike conventional DNA-based sequencing, ExomeReveal performs functional RNA studies, resolving variants of uncertain significance and improving diagnostic accuracy. Initial trials showed clinically meaningful results in 1 in 50 patients, with an additional 5% benefiting from long-term genomic reanalysis via the Ambry Patient for Life program.
Navigating The Landscape Of Clinical Genetic Testing: Insights And Challenges In Rare Disease Diagnostics
Clinical genetic testing is essential for diagnosing rare diseases and offers cytogenetic, molecular, and biochemical testing methods. Cytogenetic tests detect chromosomal abnormalities using techniques like microarrays (for unbalanced rearrangements) and FISH (for structural variations). Molecular testing, the most common, includes Sanger sequencing (high-accuracy single-gene analysis), PCR (gene amplification), MLPA (deletion/duplication detection), and NGS (comprehensive genome analysis). Biochemical tests, such as Southern blotting, remain relevant for specific disorders.
Test selection depends on factors like cost, turnaround time, and detection capability. While NGS offers broad coverage, it is not the gold standard for detecting CNVs or STRs. A precise clinical diagnosis ensures targeted testing, reducing time and costs. For example, Fragile X syndrome is confirmed via Southern blotting or PCR, while Alport syndrome requires COL4A5 sequencing and MLPA.
Comparative Characteristics of Different Genetic Testing Methodologies
Method
Range
Common Indication
Turnaround Time (TAT)
Cost
Example
Karyotype
Genome-wide
CNVs, structural variations
< 1 month
Low
Down syndrome
Chromosomal Microarray (CMA)
Genome-wide
CNVs, UPD (SNP platform)
< 1 month
Average
Challenging cases
FISH (Fluorescence In Situ Hybridization)
Targeted
CNVs, structural variations
< 1 week
Low
Angelman syndrome
Target PCR
Targeted
SNVs, repeat expansions
< 1 week
Low
-
MLPA (Multiplex Ligation-dependent Probe Amplification)
Targeted
Small CNVs (exon level)
> 1 month
Low
Duchenne muscular dystrophy
Southern Blot
Targeted
Small CNVs, repeat expansions
> 1 month
Low
Fragile X syndrome
Sanger Sequencing
Targeted
SNVs
> 1 month
Average
Cystic fibrosis
Gene Panel
Targeted (wide)
SNVs
> 2 months
High
Long QT syndrome
Exome/Genome Sequencing
Genome-wide
SNVs, CNVs (mainly WGS)
> 2 months
High
Challenging cases
Market Concentration & Characteristics
The rare disease genetic testing market is witnessing significant innovation, driven by advancements in next-generation sequencing (NGS), AI-driven data analysis, and multi-omic approaches. Technologies such as whole-genome sequencing (WGS) and RNA sequencing enhance diagnostic accuracy by identifying previously undetectable genetic mutations. Ambry Genetics' ExomeReveal, for example, integrates RNA analysis with exome sequencing, improving variant classification and diagnostic yield. Similarly, MyOme's whole-genome analysis leverages polygenic risk scores to expand detection capabilities. These innovations reduce diagnostic timelines, lower costs, and improve patient outcomes, addressing the longstanding challenge of undiagnosed rare diseases. Ongoing R&D and collaborations continue to push the boundaries of genetic diagnostics.
The market has seen a high level of mergers and acquisitions (M&A) as companies seek to expand capabilities, enhance technology portfolios, and strengthen market presence. Key drivers include the rising demand for advanced diagnostics, integration of AI-driven genomic analysis, and cost efficiencies. For example, Invitae Corporation acquired ArcherDX to improve precision oncology and rare disease diagnostics, while Labcorp acquired Personal Genome Diagnostics to enhance its genomic profiling services. Similarly, Eurofins acquired DNA Diagnostics Center (DDC) to strengthen its genetic testing portfolio.
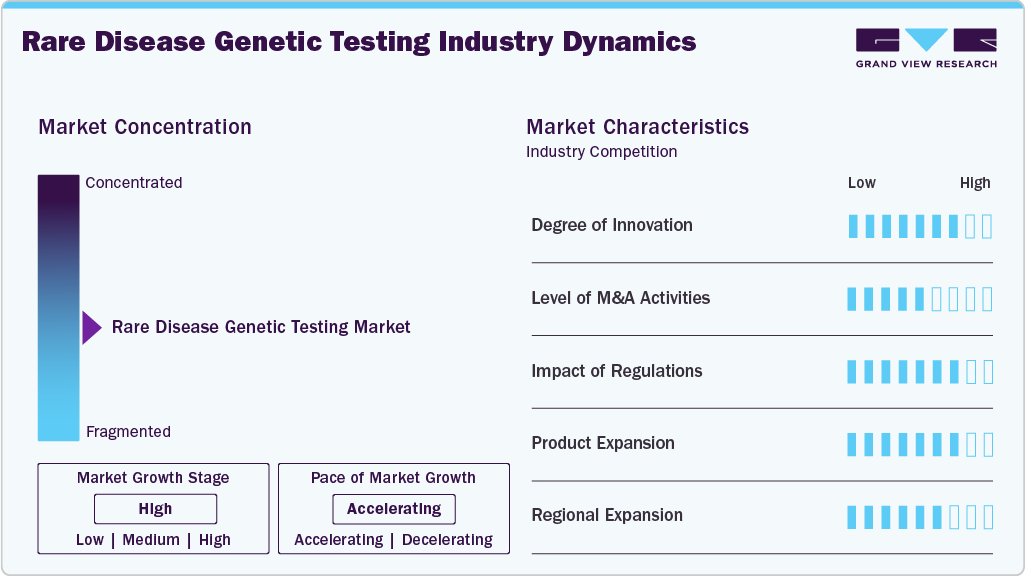
Regulatory frameworks play a crucial role in shaping the rare disease genetic testing market, ensuring test accuracy, patient safety, and ethical compliance. Stringent regulations from authorities such as the FDA (U.S.), EMA (Europe), and CFDA (China) impact test development, validation, and commercialization. For example, the EU In Vitro Diagnostic Regulation (IVDR) has increased compliance costs, requiring stricter clinical evidence for genetic tests. In the U.S., FDA oversight of laboratory-developed tests (LDTs) affects market entry timelines.
Product substitutes are limited due to the uniqueness of genetic diagnostics. However, alternative diagnostic approaches exist, such as biochemical testing that detects metabolic abnormalities linked to genetic disorders, such as enzyme assays for lysosomal storage diseases. Imaging techniques, including MRI, CT scans, and ultrasound, assist in diagnosing structural abnormalities in congenital disorders. Electrophysiological tests, such as EEG and EMG, play a role in conditions like muscular dystrophies and neurological disorders. Clinical diagnosis and family history analysis remain traditional methods based on symptom observation and hereditary patterns. Despite these alternatives, genetic testing remains superior in precision, early detection, and personalized treatment planning.
The rare disease genetic testing industry is experiencing significant geographical expansion, driven by increasing awareness, technological advancements, and government initiatives. North America leads due to strong healthcare infrastructure, regulatory support, and high adoption of next-generation sequencing (NGS). Europe follows closely, with the implementation of the EU In Vitro Diagnostic Regulation (IVDR) shaping market growth. The Asia Pacific region is witnessing rapid expansion, fueled by rising investments in genomics, expanding healthcare access, and initiatives such as China's Precision Medicine Initiative. Emerging markets in Latin America and the Middle East are also gaining traction as governments promote genetic research and diagnostic accessibility, further broadening market reach.
Disease Insights
The neurological disorders segment captured the largest revenue share at 29.09% in 2025, driven by high unmet needs and rapid scientific progress. Rare neurological diseases often present with overlapping symptoms, making diagnosis extremely difficult through clinical evaluation alone. This challenge has created strong demand for advanced genetic testing solutions, contributing a significant share of the overall rare disease testing market. A major driver of this momentum is innovation in sequencing technologies. In July 2025, Athena Diagnostics (a Quest Diagnostics company) introduced a long-read gene-sequencing test to detect repeat expansions and complex variants in ataxia and related neurological disorders. Standard short-read methods often miss these subtle but critical mutations; the adoption of long-read sequencing represents a leap forward in diagnostic precision and is poised to redefine testing standards in neurology. Equally important are strategic partnerships that embed genetic testing deeper into neurological care pathways. In March 2025, Baylor Genetics joined the Child Neurology Society as an official genetic testing partner. This collaboration highlights the growing recognition that genetic insights are indispensable for managing pediatric neurological disorders, while also expanding Baylor’s reach into specialist networks that directly impact patient care.
The endocrine & metabolism diseases segment is expected to grow at the fastest CAGR of 17.84% during the forecast period. A key factor driving this momentum is the combination of industry partnerships and expanded diagnostic access. In March 2024, CENTOGENE GmbH strengthened its long-standing global collaboration with Takeda Pharmaceutical Company to continue offering diagnostic testing services for patients with lysosomal storage disorders, including Fabry disease, Gaucher disease, and Hunter syndrome. This collaboration demonstrates how partnerships between diagnostic companies and pharmaceutical organizations can broaden patient access to testing while supporting therapeutic development and treatment monitoring. By reinforcing its position as a global diagnostic provider, CENTOGENE highlights how such alliances strengthen the rare disease ecosystem from diagnosis to therapy.
Technology Insights
The Next Generation Sequencing segment accounted for the largest share of 57.13% in 2025. The integration of next-generation sequencing (NGS) technologies has significantly advanced the field of rare disease genetic testing, enabling more precise, comprehensive, and timely diagnoses. Improvements in sequencing platforms, bioinformatics tools, and genomic interpretation have expanded the ability of clinicians and researchers to detect pathogenic variants responsible for rare inherited disorders. As sequencing costs continue to decline and accessibility improves, NGS is increasingly being adopted across clinical laboratories, research institutions, and public healthcare systems to support earlier detection and improved management of rare genetic diseases. For instance, in October 2024, Illumina introduced the MiSeq i100 gene sequencers, compact benchtop sequencing devices designed to broaden access to NGS technology. The systems were priced at approximately USD 49,000 for the MiSeq i100 and USD 109,000 for the MiSeq i100 Plus, allowing smaller research laboratories and diagnostic facilities to perform in-house sequencing. By reducing the cost and complexity associated with genomic testing, these platforms aim to expand the adoption of NGS-based diagnostics in rare disease testing workflows.
Array-based technologies have emerged as a critical component in rare disease genetic testing, providing high-resolution analysis of the genome for the detection of copy number variations (CNVs), chromosomal imbalances, and other submicroscopic genetic alterations. These platforms, including comparative genomic hybridization (CGH) arrays and single-nucleotide polymorphism (SNP) arrays, allow for the simultaneous examination of thousands of genetic markers, enabling clinicians to identify anomalies that may not be detectable through traditional karyotyping or sequencing approaches. The ability to rapidly screen the genome with such precision makes array-based technology particularly valuable in diagnosing complex rare diseases, intellectual disabilities, and congenital anomalies.
Specialty Insights
The molecular genetic tests segment dominated the market in 2025 and is anticipated to grow at the fastest CAGR during the forecast period, powered by innovative technologies, global collaborations, and rising demand for precision medicine. A notable instance is the partnership between Myriad Genetics and SOPHiA GENETICS in September 2025 to develop an innovative liquid biopsy companion diagnostic (CDx) test. This collaboration combines Myriad’s advanced laboratory capabilities in the U.S. with SOPHiA’s extensive decentralized network in countries, ensuring broad global deployment. The initial focus is on MSK-ACCESS, powered with SOPHiA DDM, developed with Memorial Sloan Kettering Cancer Center. This test detects actionable genomic alterations from a simple blood draw, providing an advanced tumor-profiling solution that could accelerate oncology clinical trials and expand patient access to personalized treatments.
Chromosomal genetic testing is advancing rapidly, with innovations transforming diagnostics, treatment, and patient access across oncology, prenatal health, and rare disease care. In October 2024, NIH launched the myeloMATCH precision medicine trial to test tailored drug combinations for acute myeloid leukemia (AML) and myelodysplastic syndromes (MDS). By matching therapies to genetic changes in patients’ blood cancers, the initiative aimed to accelerate targeted treatment strategies.
End Use Insights
The diagnostic laboratories segment dominated the market in 2025. These laboratories offer specialized testing services and often serve as referral centers for hospitals, clinics, and research institutions. With advancements in automation, bioinformatics, and multiplex testing, diagnostic laboratories have significantly increased their throughput and diagnostic accuracy. They also play a crucial role in providing confirmatory testing, variant interpretation, and genetic counseling services to patients and clinicians. The further reason for market growth is the rising number of partnership and collaboration activities of diagnostic laboratories with genetic testing companies. In November 2023, Genomenon and Alexion, AstraZeneca Rare Disease announced a strategic collaboration projected to make important information for the treatment and diagnosis of rare diseases more readily available. The goal of the collaboration is to empower the genetic testing laboratories with the data they need for the diagnosis of rare diseases.
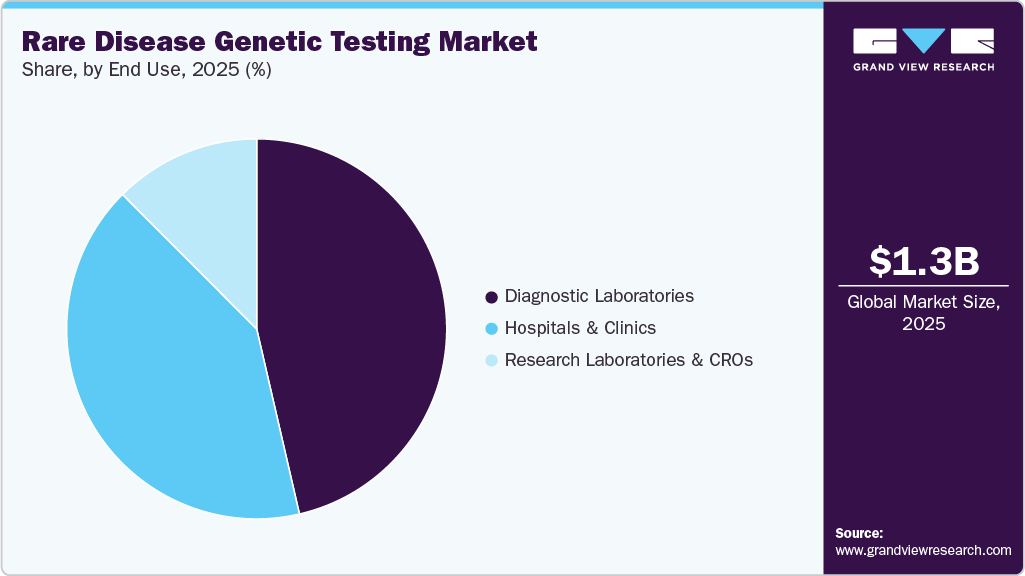
The hospitals & clinics segment is projected to witness the fastest growth rate in the market. The integration of genetic testing into routine clinical workflows has become increasingly important for early detection, personalized treatment planning, and risk assessment. Hospitals are investing in in-house genetic testing capabilities, particularly for conditions with high clinical urgency such as neonatal or pediatric rare diseases. In 2024, Mayo Clinic launched an advanced rare disease genomic testing program, leveraging AI-driven analytics for faster detection of genetic variants. Additionally, the integration of cloud-based bioinformatics platforms, such as Fabric Genomics' AI-powered variant interpretation tool, is enhancing diagnostic efficiency. Growing investment in precision medicine and newborn screening programs is further fueling market expansion.
Regional Insights
The North America rare disease genetic testing industry dominated the global market with a share of 46.73% in 2025. The market is experiencing significant growth due to advanced healthcare infrastructure, high adoption of next-generation sequencing (NGS), and strong government support for rare disease research. The U.S. leads the market, driven by initiatives such as the Rare Diseases Clinical Research Network (RDCRN) and the Orphan Drug Act, which incentivizes research and development. Companies like Invitae and Ambry Genetics offer expanded genetic testing solutions for rare conditions, improving early diagnosis. In 2023, Illumina launched the TruSight Software Suite, enhancing the interpretation of rare disease variants. Increasing patient awareness and collaborations between biotech firms and research institutes further propel market expansion.
U.S. Rare Disease Genetic Testing Market Trends
The rare disease genetic testing industry in the U.S. is witnessing substantial growth due to rising demand for early diagnosis and advancements in multiomic testing approaches. The increasing prevalence of rare genetic disorders has led to greater adoption of whole-genome sequencing (WGS) and RNA sequencing for more precise diagnostics. In 2024, Mayo Clinic launched an advanced rare disease genomic testing program, leveraging AI-driven analytics for faster detection of genetic variants. Additionally, the integration of cloud-based bioinformatics platforms, such as Fabric Genomics' AI-powered variant interpretation tool, is enhancing diagnostic efficiency. Growing investment in precision medicine and newborn screening programs is further fueling market expansion.
Europe Rare Disease Genetic Testing Market Trends
The rare disease genetic testing industry in Europe is expanding due to strong government support, increased funding for genomic research, and advancements in next-generation sequencing (NGS). Countries like Germany, the UK, and France are leading the market, driven by national genomic initiatives such as the UK’s 100,000 Genomes Project and France’s Genomic Medicine Plan 2025. In 2023, Centogene partnered with Takeda to enhance genetic testing for rare metabolic disorders, improving early detection. Additionally, the European Reference Networks (ERNs) are fostering cross-border collaboration for rare disease diagnostics. Growing accessibility to whole-exome sequencing (WES) and AI-driven bioinformatics platforms is further driving market growth.
The France rare disease genetic testing industry is growing due to strong government initiatives, increasing adoption of next-generation sequencing (NGS), and expanding precision medicine programs. The France Genomic Medicine Plan 2025 has played a crucial role in integrating genetic testing into the healthcare system, boosting accessibility to whole-exome sequencing (WES) and whole-genome sequencing (WGS). In 2023, SeqOIA, a leading genomic medicine center, expanded its rare disease testing capabilities to improve early diagnosis. Additionally, collaborations between research institutions and biotech firms, such as Genethon’s partnership with hospitals for rare disease gene therapy research, are accelerating advancements in the market.
The rare disease genetic testing industry in Germany is expanding due to strong government support, technological advancements, and increased adoption of whole-genome sequencing (WGS) and next-generation sequencing (NGS). The German Human Genome-Phenome Archive (GHGA) is enhancing genomic data accessibility, facilitating rare disease research. In 2023, CeGaT launched an advanced multiomics-based genetic testing service, improving diagnostic accuracy. Additionally, the German National Cohort (NAKO) study is driving innovation by integrating genetic testing into epidemiological research. Collaborations between biotech firms and healthcare providers, such as BioNTech’s expansion into rare disease diagnostics, are further fueling market growth and enhancing patient access to genetic testing.
Asia Pacific Rare Disease Genetic Testing Market Trends
The rare disease genetic testing industry in the Asia Pacific is experiencing rapid growth due to increasing government initiatives, expanding healthcare infrastructure, and rising awareness of genetic disorders. Countries like China, Japan, and India are leading the market with strong investments in next-generation sequencing (NGS) and whole-genome sequencing (WGS). In 2023, China’s National Rare Disease Registry expanded genetic testing programs, improving early diagnosis. Japan’s Initiative on Rare and Undiagnosed Diseases (IRUD) is driving advancements in clinical genomics. Meanwhile, MedGenome launched a nationwide genetic testing awareness campaign in India to enhance accessibility. Growing collaborations between biotech firms and research institutions are further accelerating market expansion.
The Japan rare disease genetic testing industry is growing due to strong government support, advancements in whole-genome sequencing (WGS) and next-generation sequencing (NGS), and increasing collaborations between research institutions and biotech firms. The Initiative on Rare and Undiagnosed Diseases (IRUD), launched by the Japan Agency for Medical Research and Development (AMED), has significantly improved genetic diagnosis for rare diseases. In 2023, Takara Bio expanded its genetic testing services, enhancing precision diagnostics. Additionally, Japan’s Genomic Medicine Initiative is driving the integration of AI-based bioinformatics for the detection of rare diseases. Increased investment in newborn screening programs is further fueling market growth.
Latin America Rare Disease Genetic Testing Market Trends
The rare disease genetic testing industry in Latin America is expanding due to increasing awareness, improved healthcare infrastructure, and growing adoption of next-generation sequencing (NGS) and whole-exome sequencing (WES). Countries like Brazil, Mexico, and Argentina are leading the market, supported by government initiatives and private sector investments. In 2023, Brazil’s Fiocruz expanded its genetic testing programs, enhancing early diagnosis for rare conditions. Mexico’s National Institute of Genomic Medicine (INMEGEN) is advancing precision medicine initiatives. Additionally, partnerships between global biotech firms and local healthcare providers, such as Blueprint Genetics’ expansion into Latin America, are improving access to advanced genetic testing solutions.
The Brazil rare disease genetic testing industry is growing due to government initiatives, expanding genetic research, and increased adoption of next-generation sequencing (NGS) and whole-exome sequencing (WES). The Rare Diseases National Policy, established by the Brazilian Ministry of Health, has improved access to genetic testing and early diagnosis. In 2023, Fiocruz expanded its genomic research programs, enhancing rare disease detection capabilities. Additionally, the Genomas Brasil Initiative is driving advancements in precision medicine. Collaborations with biotech firms, such as Illumina’s partnership with Brazilian research institutions, are further strengthening the country’s genetic testing capabilities and accessibility.
Middle East & Africa Rare Disease Genetic Testing Market Trends
The rare disease genetic testing industry in the Middle East & Africa is rapidly evolving, driven by government-led genomic initiatives and strategic collaborations. For instance, the UAE Genomics Council has prioritized genetic research, leading to increased adoption of next-generation sequencing (NGS) and whole-exome sequencing (WES). In 2023, M42 expanded its partnerships with global biotech firms to enhance diagnostic capabilities. Additionally, the country is integrating AI-driven bioinformatics into genetic analysis, improving accuracy and efficiency. Collaborations with international players, such as Illumina’s alliance with UAE research centers, are further strengthening the market, ensuring broader accessibility to genetic testing for rare disease patients across the region.
The Saudi Arabia rare disease genetic testing industry is making significant strides in the market, driven by large-scale genomic initiatives and technological advancements. The Saudi Human Genome Program (SHGP) has been instrumental in identifying genetic variations associated with rare diseases, leading to improved diagnostic accuracy. In 2023, the Ministry of Health partnered with international biotech firms to integrate cutting-edge genetic testing into public healthcare. With the increasing adoption of AI-powered bioinformatics and next-generation sequencing (NGS), the country is enhancing early detection capabilities. Collaborations with global players like Illumina and BGI Genomics are further strengthening Saudi Arabia’s position in precision medicine.
Key Rare Disease Genetic Testing Company Insights
Some of the key market players include Quest Diagnostics, Inc., Centogene N.V., Invitae Corp., 3billion, Inc., and Arup Laboratories, among others. These players are undertaking various strategic initiatives to increase their share in the market. New product development, collaborations, and partnerships are some such endeavors.
Key Rare Disease Genetic Testing Companies:
The following key companies have been profiled for this study on the rare disease genetic testing market.
- Quest Diagnostics Inc.
- Centogene N.V.
- Invitae Corp.
- 3billion, Inc.
- Arup Laboratories
- Eurofins Scientific
- Strand Life Sciences
- Ambry Genetics
- Perkin Elmer, Inc.
- Realm IDX, Inc.
- Macrogen, Inc.
- Baylor Genetics
- Color Genomics, Inc.
- Health Network Laboratories
- PreventionGenetics
- Progenity, Inc.
- Coopersurgical, Inc.
- Fulgent Genetics Inc.
- Myriad Genetics, Inc.
- Laboratory Corporation of America Holdings
- Opko Health, Inc.
- Artemis DNA
Recent Developments
-
In February 2026, Ambry Genetics announced the completion of its one-millionth DNA/RNA genetic test, highlighting the growing use of paired DNA-RNA diagnostics to improve the detection of hereditary diseases and rare genetic conditions.
-
In February 2026, Illumina, Inc. announced the launch of TruPath Genome, a new solution designed to provide high-quality and comprehensive whole-genome insights for genetic diseases. TruPath Genome delivers enhanced accuracy and resolution, including in challenging “dark regions” of the genome, enabling researchers to gain a more complete understanding of genomic alterations associated with genetic disorders.
-
In July 2025, Athena Diagnostics (a Quest company) launched a long-read gene-sequencing lab-developed test to improve detection of repeat expansions and complex variants in ataxia and related neurological (rare) disorders, addressing variants missed by standard short-read methods.
Rare Disease Genetic Testing Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 1,472.10 million
Revenue forecast in 2033
USD 3,883.76 million
Growth rate
CAGR of 14.87% from 2026 to 2033
Base year for estimation
2025
Historical data
2021 - 2024
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Disease, specialty, technology, end use, region
Regional scope
North America; Europe; Asia Pacific; Latin America; Middle East and Africa
Country scope
U.S.; Canada; Mexico; Germany; UK; France; Spain; Italy; Denmark; Sweden; Norway; China; Japan; India; South Korea; Australia; Thailand; Brazil; Argentina; Saudi Arabia; Kuwait; UAE; South Africa
Key companies profiled
Quest Diagnostics, Inc.; Centogene N.V.; Invitae Corp.; 3billion, Inc.; Arup Laboratories; Eurofins Scientific; Strand Life Sciences; Ambry Genetics; Perkin Elmer, Inc.; Realm IDX, Inc.; Macrogen, Inc.; Baylor Genetics; Color Genomics; Inc.; Health Network Laboratories; PreventionGenetics; Progenity; Inc.; Coopersurgical; Inc.; Fulgent Genetics Inc.; Myriad Genetics; Inc.; Laboratory Corporation Of America Holdings; Opko Health; Inc.; Artemis DNA
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options Global Rare Disease Genetic Testing Market Report Segmentation
This report forecasts revenue growth at global, regional & country levels and provides an analysis on the industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global rare disease genetic testing market report on the basis of disease, specialty, technology, end use and region:
-
Disease Outlook (Revenue, USD Million, 2021 - 2033)
-
Neurological Disorders
-
Immunological Disorders
-
Hematology Diseases
-
Endocrine & Metabolism Diseases
-
Cancer
-
Musculoskeletal Disorders
-
Cardiovascular Disorders
-
Dermatology Diseases
-
Other Rare Diseases
-
-
Technology Outlook (Revenue, USD Million, 2021 - 2033)
-
Next Generation Sequencing
-
Whole Exome Sequencing
-
Whole Genome Sequencing
-
-
Array Technology
-
PCR-based Testing
-
FISH
-
Sanger Sequencing
-
Karyotyping
-
-
Specialty Outlook (Revenue, USD Million, 2021 - 2033)
-
Molecular Genetic Tests
-
Chromosomal Genetic Tests
-
Biochemical Genetic Tests
-
-
End Use Outlook (Revenue, USD Million, 2021 - 2033)
-
Research Laboratories & CROs
-
Hospitals & Clinics
-
Diagnostic Laboratories
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
China
-
Japan
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. Some key players operating in the rare disease genetic testing market include Quest Diagnostics, ARUP Laboratories, Perkin Elmer Inc., and Centogene.
b. Key factors that are driving the rare disease genetic testing market growth include plummeting sequencing cost, expanding patient registry for rare disease, & launch of programs to advance in rare disease diagnosis.
b. The global rare disease genetic testing market size was estimated at USD 1,293.15 million in 2025 and is expected to reach USD 1,472.10 million in 2026.
b. The global rare disease genetic testing market is expected to grow at a compound annual growth rate of 14.87% from 2026 to 2033 to reach USD 3,883.76 million by 2033.
b. Next Generation Sequencing (NGS) dominated the rare disease genetic testing market with a revenue share of 57.13% in 2025. High penetration of Whole Exome Sequencing (WES) coupled with introduction of Whole Genome Sequencing (WGS)-based new tests has driven the segment share in 2025.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










