- Home
- »
- Medical Devices
- »
-
Small Molecule Innovator CDMO Market, Industry Report, 2033GVR Report cover
![Small Molecule Innovator CDMO Market Share & Trends Report]()
Small Molecule Innovator CDMO Market (2025 - 2033 ) Share & Trends Analysis Report By Product (Small Molecule API, Small Molecule Drug Product), By Stage Type (Clinical, Commercial), By Therapeutic Area, By Customer Type, By Region, And Segment Forecasts
- Report ID: GVR-4-68039-908-0
- Number of Report Pages: 145
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Small Molecule Innovator CDMO Market Summary
The global small molecule innovator CDMO market size was estimated at USD 41.39 billion in 2024 and is projected to reach USD 69.38 billion by 2033, growing at a CAGR of 6.07% from 2025 to 2033. The growth of the market is due to the increasing outsourcing of drug development and manufacturing by pharmaceutical innovators, particularly in the small molecule space.
Key Market Trends & Insights
- North America small molecule innovator CDMO market held the largest revenue share of 40.88% of the global market in 2024.
- The small molecule innovator CDMO industry in the U.S. is expected to grow significantly over the forecast period.
- By product, the small molecule API segment led the market with the largest revenue share of 61.93% in 2024.
- Based on stage type, the clinical segment led the market with the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 41.39 Billion
- 2033 Projected Market Size: USD 69.38 Billion
- CAGR (2025-2033): 6.07%
- North America: Largest market in 2024
- Asia-Pacific: Fastest growing market
As drug pipelines become more complex and regulatory scrutiny tightens, pharmaceutical companies are relying more on CDMOs to provide specialized services such as API synthesis, formulation development, and clinical-scale production. The market is driven by a growing reliance on outsourcing among pharmaceutical companies aiming to streamline operations, reduce internal manufacturing burdens, and accelerate drug development timelines. Innovator companies, especially emerging biotechs with limited infrastructure, increasingly turn to CDMOs for specialized services ranging from early-phase development to commercial manufacturing. This trend is reinforced by the complexity of regulatory compliance, the need for advanced containment facilities for potent APIs, and the high costs associated with maintaining in-house capabilities. The rise in clinical trials and new molecular entities (NMEs) targeting oncology, infectious diseases, and rare disorders is pushing demand for CDMOs that offer high-quality API production, analytical testing, and formulation development expertise under cGMP conditions.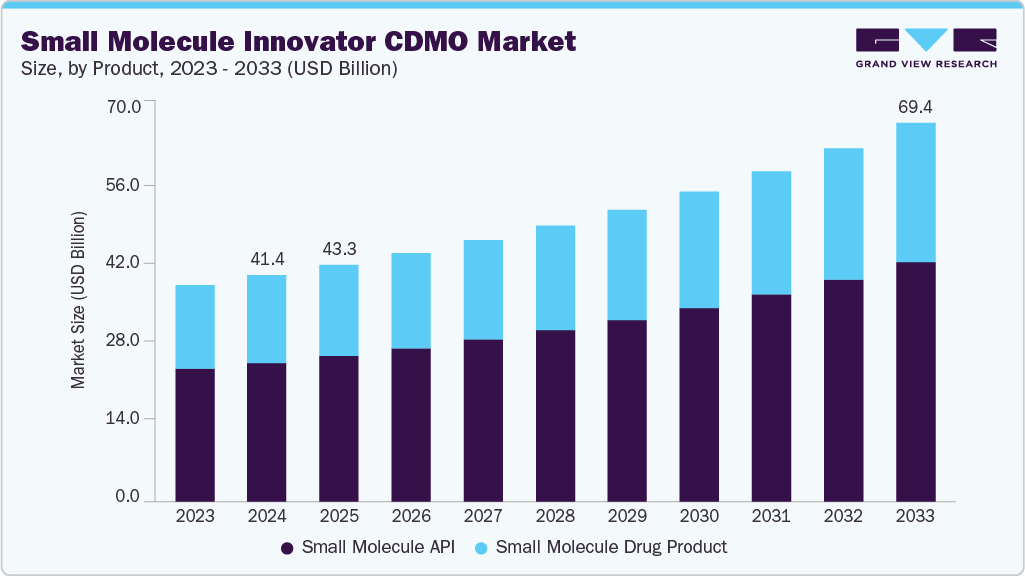
Moreover, the market is benefiting from a surge in demand for niche and targeted small molecule therapies, which require flexible and scalable manufacturing platforms. CDMOs are responding by investing in continuous manufacturing technologies, advanced process analytical tools, and modular production facilities to support fast-track drug development and complex synthesis routes. For instance, companies like Cambrex, Lonza, and Catalent have made significant capital investments to expand their capacity for small molecule API production and oral solid dose manufacturing. In 2024, Lonza expanded its Visp site to offer early-phase development for small molecules, while Piramal Pharma Solutions enhanced its high-potency API manufacturing capabilities in India and the UK. These advancements position CDMOs as strategic partners, not just vendors, and support the long-term growth of the market by aligning pharmaceutical companies’ focus on agility, risk mitigation, and time-to-market efficiency.
Technological Advancements
The technological landscape of the small molecule innovator CDMO market is defined by the integration of continuous manufacturing, high-potency API containment, and advanced process automation. Continuous manufacturing is gaining ground for its ability to streamline production, reduce costs, and ensure consistent product quality, making it a preferred choice for fast-tracked and commercial-scale drug programs. CDMOs are also investing in state-of-the-art containment systems to safely handle potent compounds, particularly in oncology and specialty therapeutics.
In parallel, the adoption of process analytical technology (PAT), real-time monitoring, and data analytics is enabling better control over critical parameters, improving yields, and regulatory compliance. The use of green chemistry and biocatalysis is advancing sustainable synthesis routes, while AI and machine learning are starting to enhance process development and optimization workflows. These innovations are transforming CDMOs into technologically agile partners capable of supporting complex small molecule programs from discovery to market.
Pricing Analysis
In the Small Molecule Innovator CDMO market, pricing is typically based on a multi-variable cost-plus and value-based hybrid model, influenced by project complexity, development phase, volume, regulatory requirements, and customization level. CDMOs apply tiered pricing structures across early-phase development, clinical trial material manufacturing, and commercial production. Early-phase projects (e.g., preclinical to Phase I) often have higher per-unit costs due to small batch sizes, non-standardized processes, and intensive analytical development. As volume scales in later phases (Phase III and commercial), cost efficiencies increase, leading to more competitive per-unit pricing.
Product Insights
The market is classified into small molecule API and small molecule drug product. The small molecule API segment accounted for the largest revenue share of 61.93% in the small molecule innovator CDMO industry in 2024. The growth of the segment is due to the increasing complexity of active pharmaceutical ingredients, rising demand for high-potency APIs (HPAPIs), and the expanding pipelines of innovator pharmaceutical companies targeting oncology, infectious diseases, and rare disorders. As drug developers prioritize the outsourcing of chemically complex and resource-intensive API synthesis, CDMOs with advanced capabilities in multi-step synthesis, containment, and regulatory compliance are witnessing increased contract volumes.
The small molecule drug product segment is anticipated to grow at a considerable CAGR during the forecast period. The segment growth is driven due to the increasing demand for oral solid dosage forms, the rising number of novel drug approvals, and the need for advanced formulation technologies. Innovator pharmaceutical companies are increasingly outsourcing formulation development, clinical trial material production, and commercial-scale manufacturing to CDMOs to accelerate time-to-market and reduce internal infrastructure costs.
Stage Type Insights
The market is segregated into preclinical, clinical, and commercial. The clinical segment held the largest revenue share in 2024 due to the rising number of small molecule candidates advancing into clinical development, coupled with the increasing reliance of pharmaceutical innovators on CDMOs for GMP-compliant manufacturing, formulation development, and supply of clinical trial materials across Phases I to III.
The commercial segment is anticipated to grow at the fastest CAGR over the forecast period. The growth of the segment is due to the increasing number of small molecule drug approvals, rising demand for large-scale production capabilities, and the shift toward long-term strategic partnerships between pharmaceutical innovators and CDMOs. As more investigational drugs successfully progress through clinical trials and receive regulatory approvals, innovators are turning to CDMOs for cost-effective, scalable, and compliant commercial manufacturing solutions.
Customer Type Insights
The market is segregated into pharmaceutical and biotechnology. The pharmaceutical segment held the largest revenue share in 2024 due to the extensive outsourcing of small molecule development and manufacturing activities by large and mid-sized pharmaceutical companies aiming to streamline operations, reduce costs, and focus on core competencies such as drug discovery and commercialization. These companies maintain broad pipelines across multiple therapeutic areas, requiring robust CDMO support for API synthesis, formulation development, clinical trial material supply, and commercial production.
The biotechnology segment is anticipated to grow at a considerable CAGR over the forecast period. The growth of the segment is due the increasing number of small and mid-sized biotech firms developing novel small molecule therapeutics, particularly in niche and high-value therapeutic areas such as oncology, rare diseases, and neurology. These companies often operate with limited in-house manufacturing infrastructure and rely heavily on CDMOs for end-to-end development and production support from preclinical synthesis and formulation to clinical trial material supply and commercialization.
Therapeutic Area Insights
The market is segregated into cardiovascular diseases, oncology, respiratory disorders, neurology, metabolic disorders, infectious diseases, and others. The oncology segment held the largest revenue share in 2024 due to the rapidly growing global prevalence of cancer and the continuous introduction of novel targeted therapies and small molecule anticancer drugs. The high unmet medical need for effective and personalized cancer treatments has driven significant investment in oncology drug development, resulting in a robust pipeline of innovative molecules requiring specialized CDMO services.
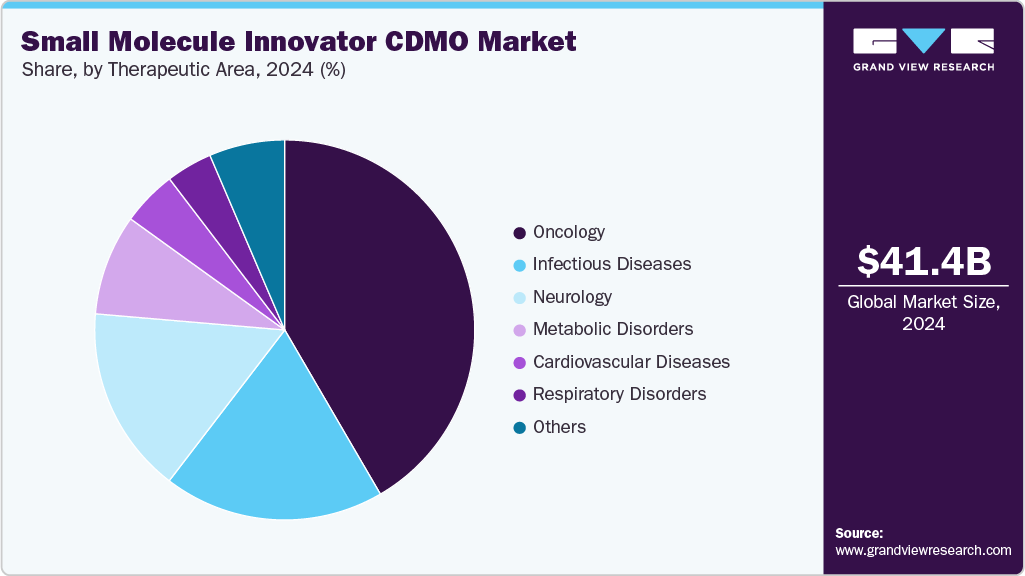
The infectious diseases segment is anticipated to grow at a considerable CAGR over the forecast period. The growth of the segment is due to the ongoing global focus on pandemic preparedness, the emergence of drug-resistant pathogens, and the continuous demand for antiviral and antibacterial small molecule therapies. Pharmaceutical and biotech companies are accelerating R&D efforts to develop novel therapeutics targeting infections such as COVID-19, influenza, tuberculosis, and multidrug-resistant bacterial strains.
Regional Insights
The North America small molecule innovator CDMOdominated the market with the largest revenue share of 40.88% in 2024, owing to the increasing R&D investments, presence of global players, and rising efforts to come up with newer patents. In addition, the increasing number of clinical trials is also fueling market growth.
U.S. Small Molecule Innovator CDMO Market Trends
The small molecule innovator CDMOmarket in the U.S. held the largest revenue share in 2024. This is attributed to the increasing number of clinical trials being performed in the U.S. For instance, according to the National Clinical Trials Registry (NCT), around 12,326 clinical trials were undergoing across various phases for the treatment of cancer in 2022.
Europe Small Molecule Innovator CDMO Market Trends
The Europe small molecule innovator CDMOmarket is expected to grow significantly due to rising focus on clinical trial studies, favorable government initiatives and policies, compliance with the Good Clinical Practice (GCP) standards, larger patient pools, and rising number of clinical trials are some of the key factors fueling the market growth.
The small molecule innovator CDMO market in Germany is driven by the factors such as advanced medical expertise and easy access to patients, low rate of saturation of competing trials, and high number of participants contributing to the clinical research studies.
The small molecule innovator CDMO market in the UK is driven by factors such as rising government spending on clinical research and rising number of clinical trial studies in the region. According to the National Institute for Health Research (NIHR), UK, clinical research is worth USD 3.74 billion each year and includes USD 2.07 billion from commercial sources, which supports around 47,000 jobs.
Asia Pacific Small Molecule Innovator CDMO Market Trends
The Asia Pacific is expected to grow at the fastest CAGR over the forecast period due togrowing investment by developed countries in the regions and various amendments made by the regulatory agencies to facilitate local manufacturing & contract services.
The small molecule innovator CDMO market in China held the largest revenue share in 2024. The growth is attributed to the government initiatives to regulate the clinical trials as per the global standards and better infrastructure, which includes 4,000 hospital beds. Initially in China, Centre of Drug Evaluation (CDE) was responsible was regulating the clinical trials
The small molecule innovator CDMO market in Japan is expected to grow over the forecast period. The regulatory process is supportive of faster clinical trial approval. For instance, clinical trials can be started once the probable benefits are demonstrated by carrying out a relatively smaller number of clinical trials.
The small molecule innovator CDMO market in India is anticipated to grow at the lucrative CAGR over the forecast period. This can be attributed tothe increasing government support by launching several funding initiatives. The government of India focuses to expand affordable healthcare facilities in the country. This propelled the growth of clinical trials sector in India. In addition, local Indian players such as Vitas Pharma (Hyderabad) and Bugworks (Bengaluru) ventured into drug development activities.
Key Small Molecule Innovator CDMO Company Insights
Several key players are acquiring various strategic initiatives to strengthen their market position offering diverse services to customers. The prominent strategies adopted by companies are service launches, mergers & acquisitions/joint ventures merger, partnership & agreements, expansions, and others to increase market presence & revenue and gain a competitive edge drives the market growth.
Key Small Molecule Innovator CDMO Companies:
The following are the leading companies in the small molecule innovator CDMO market. These companies collectively hold the largest market share and dictate industry trends.
- Piramal Pharma Solutions
- CordenPharma International
- Wuxi AppTec
- Cambrex Corporation
- Recipharm AB
- Thermo Fisher Scientific, Inc.
- Lonza
- Catalent Inc.
- Siegfried Holding AG
- Boehringer Ingelheim
- Labcorp
Recent Developments
-
In October 2024, Thermo Fisher Scientific, Inc. launched its Accelerator Drug Development offerings designed to support both emerging biotech firms and major pharmaceutical companies. This comprehensive suite encompasses manufacturing, clinical research, and supply chain services across various modalities, including small molecules covering the entire journey from preclinical stages through to commercial launch.
-
In June 2024, Siegfried plans to acquire an early-phase CDMO site in Wisconsin, U.S., specializing in highly potent APIs. The site will serve as a hub for early-phase development and manufacturing services, strengthening Siegfried's global capabilities.
Small Molecule Innovator CDMO Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 43.31 billion
Revenue forecast in 2033
USD 69.38 billion
Growth rate
CAGR of 6.07% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Report updated
August 2025
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, stage type, customer type, therapeutic area, and region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; Mexico; UK; Germany; France; Italy; Spain; Norway; Denmark; Sweden; Japan; China; India; Australia; South Korea; Thailand; Brazil; Argentina; South Africa; Saudi Arabia; Kuwait; UAE
Key companies profiled
Piramal Pharma Solutions; CordenPharma International; Wuxi AppTec; Cambrex Corporation; Recipharm AB; Thermo Fisher Scientific, Inc.; Lonza; Catalent Inc.; Siegfried Holding AG; Boehringer Ingelheim; Labcorp
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global Small Molecule Innovator CDMO Market Report Segmentation
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the global small molecule innovator CDMO market report based on product, stage type, customer type, therapeutic area, and region.
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Small Molecule API
-
Small Molecule Drug Product
-
Oral Solid Dose
-
Semi-Solid Dose
-
Liquid Dose
-
Others
-
-
-
Stage Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Preclinical
-
Clinical
-
Phase I
-
Small
-
Medium
-
Large
-
-
Phase II
-
Small
-
Medium
-
Large
-
-
Phase III
-
Small
-
Medium
-
Large
-
-
-
Commercial
-
-
Customer Type Outlook (Revenue, USD Million, 2021 - 2033)
-
Pharmaceutical
-
Small
-
Medium
-
Large
-
-
Biotechnology
-
-
Therapeutic Area Outlook (Revenue, USD Million, 2021 - 2033)
-
Cardiovascular Diseases
-
Oncology
-
Respiratory Disorders
-
Neurology
-
Metabolic Disorders
-
Infectious Diseases
-
Others
-
-
Regional Outlook (Revenue, USD Million, 2021 - 2033)
-
North America
-
U.S.
-
Canada
-
Mexico
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Norway
-
Sweden
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
South Korea
-
Thailand
-
-
Latin America
-
Brazil
-
Argentina
-
-
Middle East and Africa (MEA)
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global small molecule innovator CDMO market size was estimated at USD 41.39 billion in 2024 and is expected to reach USD 43.31 billion in 2025.
b. The global small molecule innovator CDMO market is expected to grow at a compound annual growth rate of 6.07% from 2025 to 2033 to reach USD 69.38 billion by 2033.
b. North America dominated the small molecule innovator CDMO market with a share of 40.8% in 2024. This is attributable to rising healthcare awareness coupled with cloud-based technologies acceptance and constant research and development initiatives.
b. Some key players operating in the small molecule innovator CDMO market include Lonza Group Ltd., Thermo Fisher Scientific Inc, Cambrex Corporation, Catalent Inc., Siegfried holdings AG, Recipharm AB, Corden Pharma International, Boehringer Ingelheim, Piramal Pharma Solutions, Covance Inc.
b. The main drivers for this small molecule innovator CDMO market are increasing pharmaceutical R&D investment, growing demand for small molecules, and rising incidence of cancer & age-related disorders.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










