- Home
- »
- Medical Devices
- »
-
U.S. Dental Membrane And Bone Graft Substitutes Market, 2033GVR Report cover
![U.S. Dental Membrane And Bone Graft Substitutes Market Size, Share & Trends Report]()
U.S. Dental Membrane And Bone Graft Substitutes Market (2025 - 2033) Size, Share & Trends Analysis Report By Product (Bone Graft Substitutes, Dental Membranes & Soft Tissue Regeneration Materials), By Application (Sinus Lift, Socket Preservation), And Segment Forecasts
- Report ID: GVR-4-68040-206-5
- Number of Report Pages: 80
- Format: PDF
- Historical Range: 2021 - 2024
- Forecast Period: 2025 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
Market Size & Trends
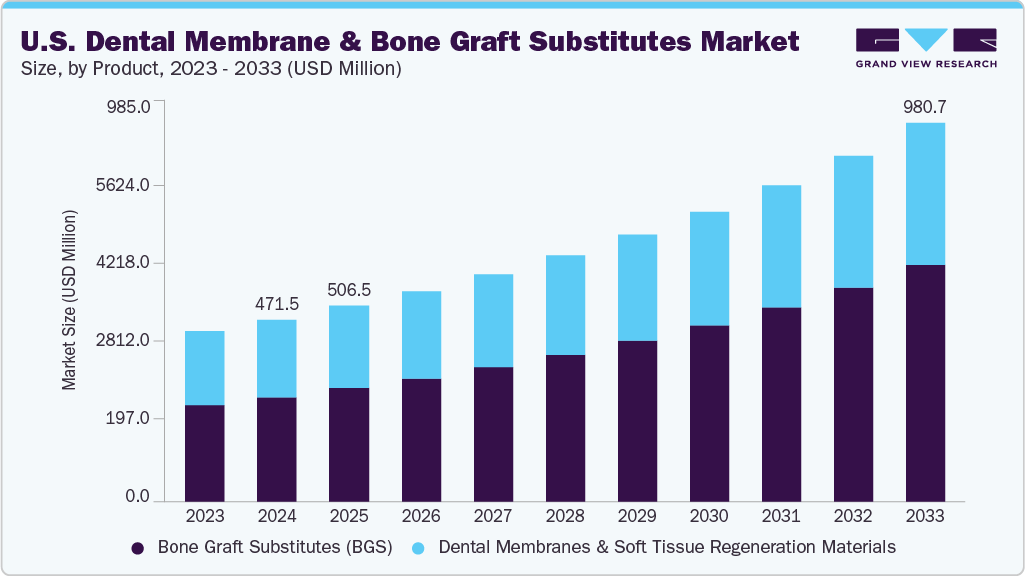
The U.S. dental membrane and bone graft substitutes market size was valued at USD 471.50 million in 2024 and is anticipated to reach USD 980.68 million by 2033, growing at a CAGR of 8.61% from 2025 to 2033. This growth is attributed to the rising prevalence of periodontal diseases, increasing demand for dental implants, and growing awareness of oral health and aesthetics.
Key Market Trends & Insights
- By product, the bone graft substitutes (BGS) segment led the market with the largest revenue share in 2024.
- By application, the socket preservation segment led the market with the largest revenue share in 2024.
Market Size & Forecast
- 2024 Market Size: USD 471.50 Million
- 2033 Projected Market Size: USD 980.68 Million
- CAGR (2025-2033): 8.61%
Advancements in regenerative dental technologies, such as bioresorbable membranes, xenografts, and alloplastic grafts, are improving treatment outcomes, accelerating healing, and expanding the use of these materials in both restorative and cosmetic dental procedures. Additionally, an aging population with higher rates of tooth loss and bone resorption, and the increasing availability of minimally invasive procedures, are further boosting market growth.The rising burden of dental disorders is a key factor driving the growth of the industry. Increasing incidences of conditions such as periodontal disease, tooth loss, and jawbone deterioration are leading to a greater need for regenerative dental procedures. These disorders often result in bone resorption and soft tissue loss, creating the necessity for bone graft substitutes and barrier membranes to restore oral structure and function. Furthermore, the growing demand for dental implants as a preferred tooth replacement solution has intensified the use of these biomaterials to ensure implant stability and successful bone regeneration. As awareness of oral health and access to advanced dental treatments expand, the demand for dental membranes and bone graft substitutes is expected to rise steadily across the country.
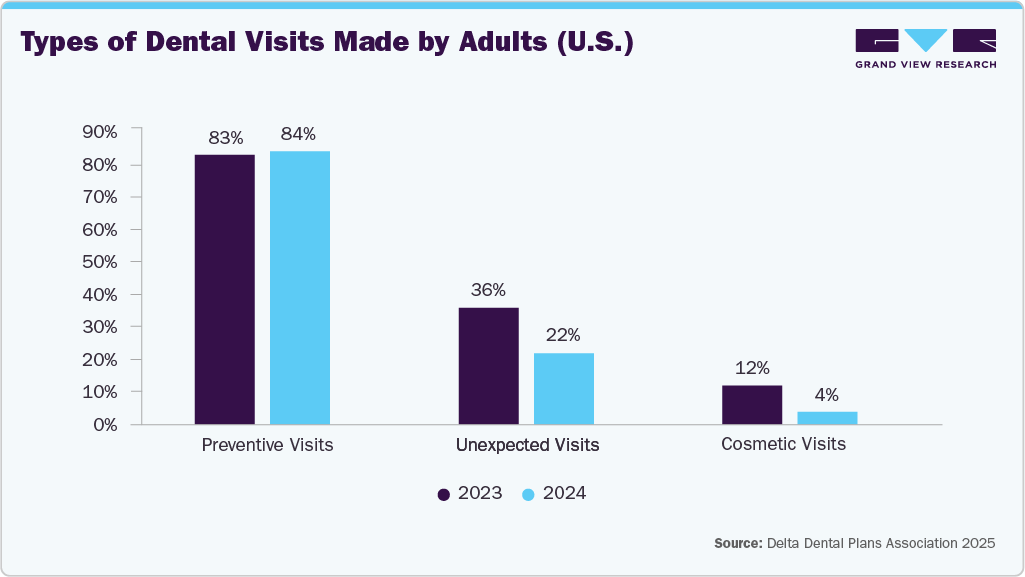
Regular dental visits are pivotal in driving the U.S. dental membrane and bone graft substitutes market, as they directly influence the need for preventive and restorative dental procedures. Many U.S. adults undergo routine dental check-ups annually, promoting early detection of periodontal disease, bone loss, and structural oral defects that often necessitate regenerative interventions using grafts and membranes. This emphasis on preventive care also enhances patient awareness and acceptance of advanced regenerative solutions, supporting sustained market demand. Additionally, unplanned or emergency dental visits, such as those for trauma, infections, or extractions, create immediate clinical needs for bone preservation and augmentation. The steady flow of routine and emergency cases maintains a consistent demand for dental membranes and bone graft substitutes, strengthening market growth across the country.
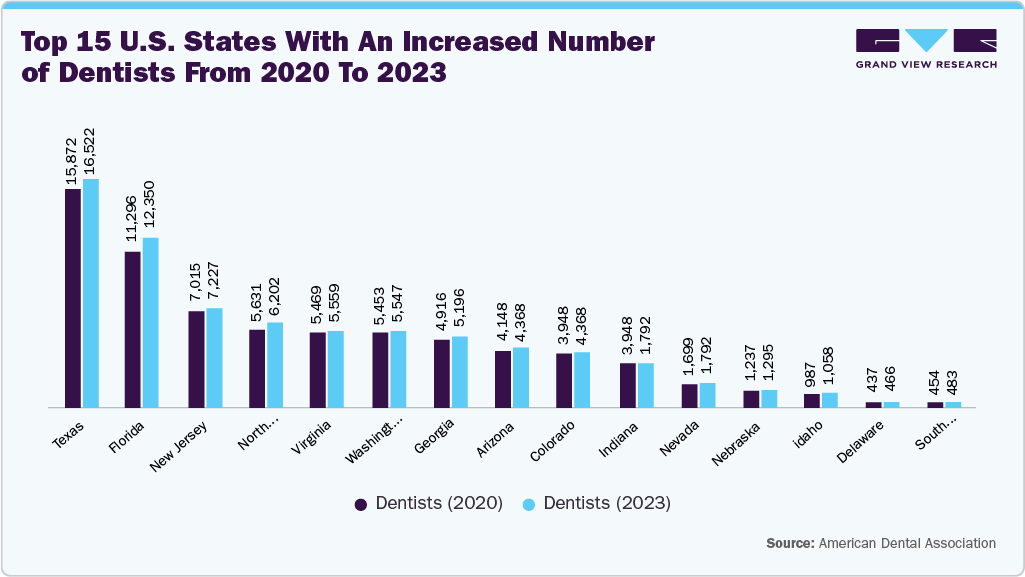
The expanding number of practicing dentists in the U.S. is a key driver of the dental membrane and bone graft substitutes market, as it enhances care accessibility and accelerates the adoption of advanced dental procedures. With more dentists, particularly specialists in periodontics, oral surgery, and implantology, entering the workforce, bone grafting and guided tissue regeneration procedures are becoming more widely available across urban, suburban, and rural settings. This expansion broadens the patient base, leading to more preventive, restorative, and implant-related treatments where bone graft substitutes and membranes are frequently utilized. Moreover, new-generation dentists increasingly embrace regenerative dentistry, digital workflows, and evidence-based biomaterials, contributing to greater integration of grafting materials into everyday practice. Growing competition among dental professionals also encourages diversifying practices and offering advanced regenerative services to attract patients. From a broader perspective, this workforce expansion supports increased patient throughput, more frequent dental visits, and heightened market awareness of regenerative solutions. As the U.S. population continues to age, illustrated by the Census Bureau’s 2024 estimate showing a 3.1% rise in adults aged 65 and older to 61.2 million, the demand for complex restorative and implant procedures is expected to rise, further propelling market growth for dental membranes and bone graft substitutes.
Strategic partnerships and investments play a significant role in driving the U.S. dental membrane and bone graft substitutes market by enabling companies to expand their market presence, accelerate product innovation, and improve distribution networks. Collaborations between dental biomaterial manufacturers, research institutions, and dental service providers facilitate the development and commercialization of advanced grafting materials and membrane technologies, ensuring faster adoption among dental professionals. For instance, in January 2025, NovaBone Products, one of the leaders in bioactive synthetic bone graft solutions, announced a strategic distribution partnership with BEGO, a renowned provider of innovative implantology solutions. This collaboration will introduce NovaBone’s advanced Dental Putty with Cartridge Dispenser to the European market, representing a major advancement in providing advanced dental graft options to practitioners throughout the region.
Some of the key opinions
Name & Role
Key Message
Market Impact (U.S.)
Nevin Mathai, DMD - Dental Director, Delta Dental of Missouri
Establishing good oral health habits early in childhood sets the foundation for a lifetime of healthy smiles. Educating children on a healthy diet, proper brushing, and flossing techniques can prevent cavities and gum disease, ensuring strong, healthy teeth as they grow. These habits play a key role in maintaining good overall health and well-being.
Drives demand for preventive dentistry. More early dental visits, higher awareness of oral care, and long-term need for restorative and implant-related procedures boost bone graft substitutes and membrane use later in life.
Cindy McGregor, DDS - VP of Dental Services, Delta Dental of Minnesota
Building on positive experiences is a good way to ease some potential concerns from parents and children. Having children start dental visits early - before their first birthday or within six months after their first tooth appears - with parents and taking the opportunity to highlight and reinforce oral hygiene is key. For those children who need intervention early in their dental visits, using descriptive, kid-friendly language of the feeling of the procedure and the tell-show-do technique is especially helpful.
Encourages pediatric dental visits. Expands patient pool from a younger age, prevents severe untreated cases, but also increases early identification of bone or dental defects and may raise long-term adoption of grafting and membranes as complex needs arise.
Amisha Singh, DDS, EdD - Dental Director, Delta Dental of Colorado
Oral health during pregnancy is a cornerstone of both maternal and infant well-being. The connection between a mother’s oral health and her baby’s development is profound. By prioritizing dental care, we can reduce the risks of complications such as pre-term birth and low birth weight, paving the way for a healthier start in life. Embracing comprehensive oral health care empowers mothers to protect their own health and that of their babies, safeguarding against systemic illnesses, fostering a better quality of life, and promoting holistic health.
Links oral health to maternal and infant well-being. Greater emphasis on dental check-ups during pregnancy, expands awareness of oral-systemic health, and increases market growth through preventive care and future restorative demand.
Source: GVR Analysis
Market Concentration & Characteristics
The U.S. market demonstrates a high degree of innovation driven by advancements in biomaterials, regenerative dentistry, and tissue engineering. Companies are increasingly developing bioactive and biocompatible materials, such as collagen-based, xenograft, alloplastic, and growth factor-enriched grafts that accelerate natural bone regeneration and improve patient outcomes. Innovations like resorbable membranes, 3D-printed grafts, and stem cell-based regenerative solutions are gaining traction, supported by strong clinical research and FDA approvals. Additionally, digital dentistry integration, including custom graft design through CAD/CAM technologies, enhances procedural precision, reducing surgery time and improving implant success rates. Overall, continuous R&D investments and collaborations between biotech firms, dental manufacturers, and academic institutions are driving rapid product modernization in the U.S. market.
The U.S. market is witnessing a high level of mergers and acquisitions (M&A) activity as leading companies pursue strategic expansion, product portfolio diversification, and global market penetration. Major players are acquiring innovative regenerative biomaterial firms to strengthen their clinical capabilities and meet increasing demand for advanced bone regeneration solutions in implant dentistry. For instance, in April 2024, Geistlich entered into an agreement to acquire Bionnovation Biomedical, a Brazil-based medical technology company, to expand its regenerative product portfolio and increase access to affordable grafting solutions across Latin America and other emerging markets. Such M&A strategies enhance product innovation and supply chain efficiency and accelerate the adoption of advanced biomaterials in the U.S. market, positioning companies for a stronger competitive advantage.

Regulatory oversight has a significant impact on the U.S. market, shaping product development, approval timelines, and commercialization strategies. The U.S. Food and Drug Administration (FDA) classifies these products as Class II or Class III medical devices, requiring rigorous 510(k) clearance or Premarket Approval (PMA) based on clinical safety and performance. Regulations emphasize biocompatibility, sterility, and long-term safety, especially for biologically derived materials such as allografts and xenografts. While stringent regulations may slow market entry for emerging innovators and increase R&D investments, they ultimately foster high-quality, clinically proven products, improving patient safety and supporting sustained adoption in dental implantology and oral reconstruction procedures.
The threat of product substitutes in the U.S. market is moderate, as alternative treatment approaches are available but often lack the same regenerative efficacy. Non-grafting techniques such as guided tissue regeneration without membranes, socket preservation using platelet-rich fibrin (PRF) or platelet-rich plasma (PRP), and bone morphogenetic protein (BMP)-based biologics, like Medtronic’s INFUSE, offer substitute options in certain clinical scenarios. Additionally, minimally invasive techniques and short or narrow dental implants can sometimes reduce the need for grafting procedures altogether. However, these substitutes may not provide sufficient structural support for complex defects, long-term stability, or predictable bone regeneration outcomes. As a result, while substitutes offer cost and procedural convenience, dental membranes and bone graft materials remain the preferred choice for reliable regeneration in implant dentistry and oral reconstructive surgery.
Product Insights
The bone graft substitutes (BGS) segment held the largest share in 2024, and is also anticipated to grow at the fastest CAGR of 9.66% from 2025 to 2033. The rising prevalence of periodontal diseases, tooth loss, and an aging population seeking restorative dental solutions has driven demand for grafting materials to rebuild bone structure. BGS products, including allografts, xenografts, and synthetic alloplasts, offer improved biocompatibility, osteoconductive properties, and predictable clinical outcomes, making them widely preferred by dental surgeons. Continuous advancements such as bioactive ceramics, growth factor-enhanced grafts, and customized 3D bone grafts further enhance treatment success rates.
Additionally, the availability of off-the-shelf graft materials reduces surgical complexity and eliminates donor-site morbidity associated with autografts, contributing to broader adoption. Strong clinical efficacy, expanding dental implant procedures, and growing patient preference for minimally invasive and regenerative therapies together reinforce the dominance of the BGS segment in the U.S. market.
Application Insights
The socket preservation segment dominated the U.S. market in 2024 due to its critical role in maintaining alveolar bone volume and architecture following tooth extraction. By using bone grafts and barrier membranes immediately after extraction, clinicians can prevent bone resorption, preserve the socket for future implant placement, and improve long-term esthetic and functional outcomes. The high adoption of dental implants in the U.S. and increasing patient awareness of minimally invasive procedures and improved post-extraction care have driven strong demand for socket preservation products. Additionally, advancements in graft materials, such as synthetic, allograft, and xenograft options, and bioactive membranes that enhance bone regeneration have further reinforced this segment’s market leadership, making it a preferred choice among dental professionals for predictable implant site preparation.
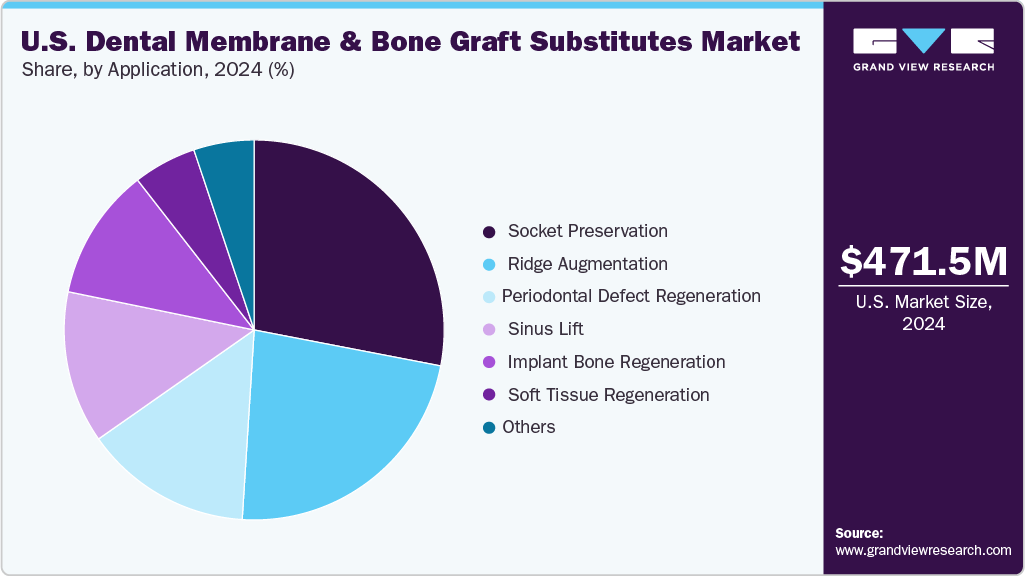
Sinus lifts are expected to grow at the fastest CAGR due to the anatomical complexities of the upper posterior jaw, where limited bone height and the proximity to the sinus cavity pose challenges for implant placement. Sinus lift procedures create adequate vertical bone volume, improving implant stability and success rates. The segment’s growth is further supported by the increasing adoption of dental implants, the rising prevalence of edentulism among the aging population, and the growing demand for long-term tooth replacement solutions.
Key U.S. Dental Membrane And Bone Graft Substitutes Companies Insights
Key players operating in the U.S. dental membrane and bone graft substitutes market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key U.S. Dental Membrane And Bone Graft Substitutes Companies:
- Dentsply Sirona
- Bioteck S.p.A.
- Institut Straumann AG
- Maxigen Biotech Inc.
- Nobel Biocare Services AG (Envista)
- Geistlich Pharma North America, Inc.
- ZimVie Inc.
- NovaBone (Halma)
- Regenity
- Medtronic
- BioHorizons
- Curasan, Inc.
- DentiumUSA
- Unicare Biomedical, Inc.
- Neoss
Recent Developments
-
In February 2025, Geistlich announced a strategic investment in ReOss Ltd, a company renowned for its patient-specific solutions that integrate digital patient data with 3D-printing technology. This equity stake underscores Geistlich's dedication to enhancing its portfolio with cutting-edge regenerative solutions. Geistlich is making a major move to reinforce its leadership in medical regeneration.
-
In September 2024, Regenity, one of the global leaders in regenerative medicine and a portfolio company of Linden Capital Partners, announced that it had obtained regulatory approval from the NMPA of China for its innovative crosslinked, bioresorbable, implantable collagen dental membrane designed for oral surgical procedures. This marks the company's first dental product approval in China. Additionally, the approval is supported by a Level 1 randomized clinical study demonstrating its effectiveness, representing the first-ever head-to-head clinical comparison between Regenity dental membranes and Geistlich dental membranes in the Chinese market.
-
In January 2025, NovaBone Products, one of the leaders in bioactive synthetic bone graft solutions, announced a strategic distribution partnership with BEGO, a renowned provider of innovative implantology solutions. This collaboration will introduce NovaBone’s advanced Dental Putty with Cartridge Dispenser to the European market, representing a major advancement in providing advanced dental graft options to practitioners throughout the region.
-
In May 2024, Geistlich achieved MDR (Medical Device Regulation) certification from TÜV SÜD Product Service GmbH for its proven product lines-Geistlich Bio-Gide, Geistlich Fibro-Gide, and Geistlich Mucograft-ensuring compliance with the updated EU regulations. Despite the more stringent quality and evidence standards introduced by the MDR, all existing indications for these products, used in various regenerative procedures, have been fully reaffirmed.
U.S. Dental Membrane And Bone Graft Substitutes Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 506.50 million
Revenue forecast in 2033
USD 980.68 million
Growth rate
CAGR of 8.61% from 2025 to 2033
Actual data
2021 - 2024
Forecast period
2025 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Segments covered
Product, application, region
Report coverage
Revenue, competitive landscape, growth factors, and trends
Key companies profiled
Dentsply Sirona; Bioteck S.p.A.; Institut Straumann AG; Maxigen Biotech Inc.; Nobel Biocare Services AG (Envista); Geistlich Pharma North America, Inc.; ZimVie Inc.; NovaBone (Halma); Regenity; Medtronic; BioHorizons; Curasan, Inc.; DentiumUSA; Unicare Biomedical, Inc.; Neoss.
Customization scope
Free report customization (equivalent to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Dental Membrane And Bone Graft Substitutes Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. dental membrane and bone graft substitutes market report on the basis of product and application:
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Bone Graft Substitutes (BGS)
-
Allograft
-
Demineralized Bone Matrix
-
Others
-
-
Autograft
-
Xenograft
-
Bovine Derived
-
Porcine Derived
-
Others
-
-
Synthetic
-
Hydroxyapatite (HA)
-
Tricalcium Phosphate (TCP)
-
Bioactive Glass
-
Others
-
-
-
Dental Membranes and Soft Tissue Regeneration Materials
-
Resorbable Membranes
-
Bovine -derived collagen based
-
Porcine derived collagen based
-
Others
-
-
Non- Resorbable Membrane
-
Expanded PTFE (ePTFE)
-
Dense PTFE (dPTFE)
-
Titanium-Reinforced PTFE
-
-
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Ridge Augmentation
-
Sinus Lift
-
Periodontal Defect Regeneration
-
Implant Bone Regeneration
-
Socket Preservation
-
Soft Tissue Regeneration
-
Others
-
Frequently Asked Questions About This Report
b. The U.S. dental membrane and bone graft substitutes market size was estimated at USD 471.50 million in 2024 and is expected to reach USD 506.50 million in 2025.
b. The U.S. dental membrane and bone graft substitutes market is expected to grow at a compound annual growth rate of 8.61% from 2025 to 2033 to reach USD 980.68 million by 2033.
b. The bone graft substitutes (BGS) segment dominated the market with a revenue share of 57.30% in 2024. This is due to its wide clinical adoption for bone regeneration, high success rates, and increasing use in dental implant procedures.
b. Some of the key companies in the U.S. dental membrane and bone graft substitutes market include Dentsply Sirona; Bioteck S.p.A.; Institut Straumann AG; Maxigen Biotech Inc.; Nobel Biocare Services AG (Envista); Geistlich Pharma North America, Inc.; ZimVie Inc.; NovaBone (Halma); Regenity; Medtronic; BioHorizons; Curasan, Inc.; DentiumUSA; Unicare Biomedical, Inc.; Neoss.
b. The U.S. market is expected to witness lucrative growth due to the increasing geriatric population and the presence of strong infrastructure for advanced treatment options such as guided tissue regeneration with CAD/CAM software solutions & digital radiography.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










