- Home
- »
- Pharmaceuticals
- »
-
U.S. Medical Foods Market Size, Industry Report, 2033GVR Report cover
![U.S. Medical Foods Market Size, Share & Trends Report]()
U.S. Medical Foods Market (2026 - 2033) Size, Share & Trends Analysis Report By Route Of Administration (Oral, Enteral), By Product (Powder, Pills, Liquid), By Application (Chronic Kidney Disease, Diabetic Neuropathy, ADHD), By Sales Channel, By Module, And Segment Forecasts
- Report ID: GVR-4-68040-297-0
- Number of Report Pages: 90
- Format: PDF
- Historical Range: 2021 - 2025
- Forecast Period: 2026 - 2033
- Industry: Healthcare
- Report Summary
- Table of Contents
- Segmentation
- Methodology
- Download FREE Sample
-
Download Sample Report
U.S. Medical Foods Market Summary
The U.S. medical foods market size was estimated at USD 6.9 billion in 2025 and is projected to reach USD 10.7 billion by 2033, growing at a CAGR of 5.5% 2026 to 2033. This growth is driven by the rising geriatric population, growing prevalence of chronic diseases, and increasing adoption of disease-specific nutritional formulas.
Key Market Trends & Insights
- Based on product, the powder segment led the market with the largest revenue share of 35.4% in 2025.
- Based on application, the cancer segment led the market with the largest revenue share of 12.25% in 2025.
- Based on route of administration, the oral segment accounted for the largest revenue share in 2025.
Market Size & Forecast
- 2025 Market Size: USD 6.9 Billion
- 2033 Projected Market Size: USD 10.7 Billion
- CAGR (2026-2033): 5.5%
The growing prevalence of chronic diseases further accelerates demand for disease-specific nutritional interventions. According to the data published by the Centers for Disease Control and Prevention in April 2025, approximately 194 million U.S. adults, which is 76.4%, reported at least one chronic condition, while over 51% had multiple chronic conditions. Among older adults, the burden is even higher, with nearly 93% of individuals aged 65+ having at least one chronic disease in 2023. Neurological conditions such as Alzheimer’s are particularly significant, with about 7.2 million Americans currently living with the disease, and projections indicating continued growth. This rising disease burden creates sustained demand for medical foods as adjunct therapy in clinical treatment pathways.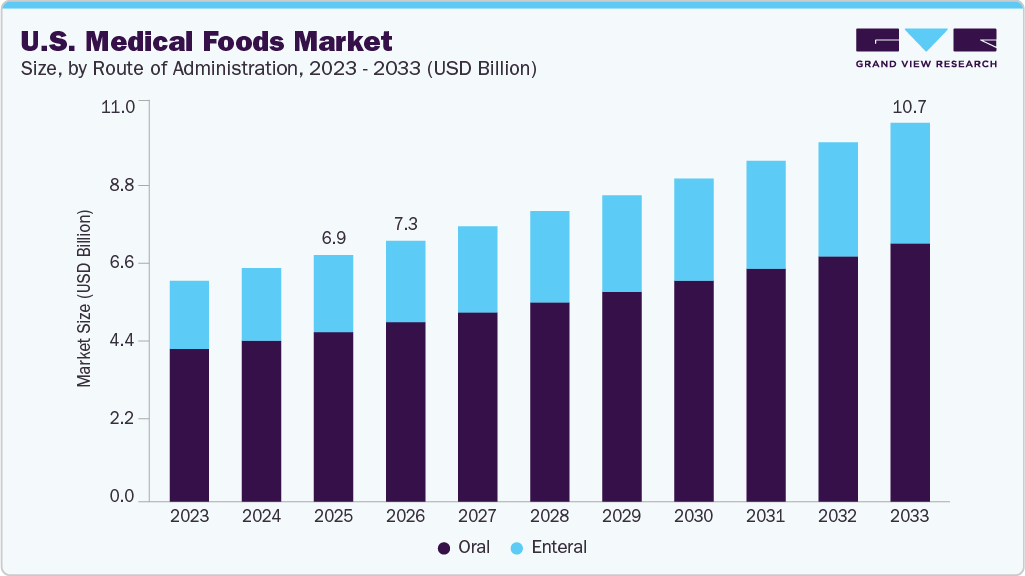
The rising incidence of chronic and rare diseases, along with advances in targeted therapies, is driving demand for disease-specific medical foods. These products are increasingly used in the management of conditions such as cancer, gastrointestinal disorders, and metabolic diseases, where tailored nutrition is essential to support treatment outcomes and avoid drug-nutrient interactions. For instance, gastrointestinal patients require easily digestible, tolerance-focused formulas, while oncology patients benefit from high-protein, energy-dense solutions to manage treatment-related complications. Leading companies such as
-
Abbott Osmolite 1.2 / 1.5 Cal: A fiber-free enteral formula designed for patients with gastrointestinal intolerance, supporting nutrient absorption and tolerance in sensitive patients.
-
Danone Milupa Product Range: Foods for Special Medical Purposes (FSMPs) intended for the dietary management of inherited metabolic disorders, supporting growth and nutrient requirements under medical supervision.
-
Nestlé BOOST SOOTHE: A clear, high-calorie oral nutritional supplement designed for patients experiencing oral discomfort, taste changes, or mucositis, particularly during cancer treatment. It provides 300 kcal and 10 g of protein per serving, in a soothing, easy-to-drink format, to help maintain weight and nutrition.
Market Concentration & Characteristics
The chart below illustrates the relationship between market concentration, industry characteristics, and industry participants. The x-axis represents the level of industry concentration, ranging from low to high. The y-axis represents various industry characteristics, including industry competition, impact of regulations, level of partnerships & collaborations activities, degree of innovation, and regional expansion. For instance, the medical foods market is consolidated, steadily moving towards fragmentation, with many product & service providers entering the market. The degree of innovation, the level of partnerships & collaboration activities, and the impact of regulations on the industry are high. However, the regional expansion observes moderate growth.
The degree of innovation in the U.S. medical foods market is steadily increasing, driven by advancements in clinical nutrition, personalized medicine, and disease-specific formulations. Companies are focusing on condition-targeted products for neurological disorders, metabolic diseases, and gastrointestinal conditions. Integration of evidence-based ingredients, improved palatability, and novel delivery formats enhance patient adherence. In addition, the use of digital health tools and data analytics is supporting more tailored nutritional interventions and expanding the market’s innovation landscape.
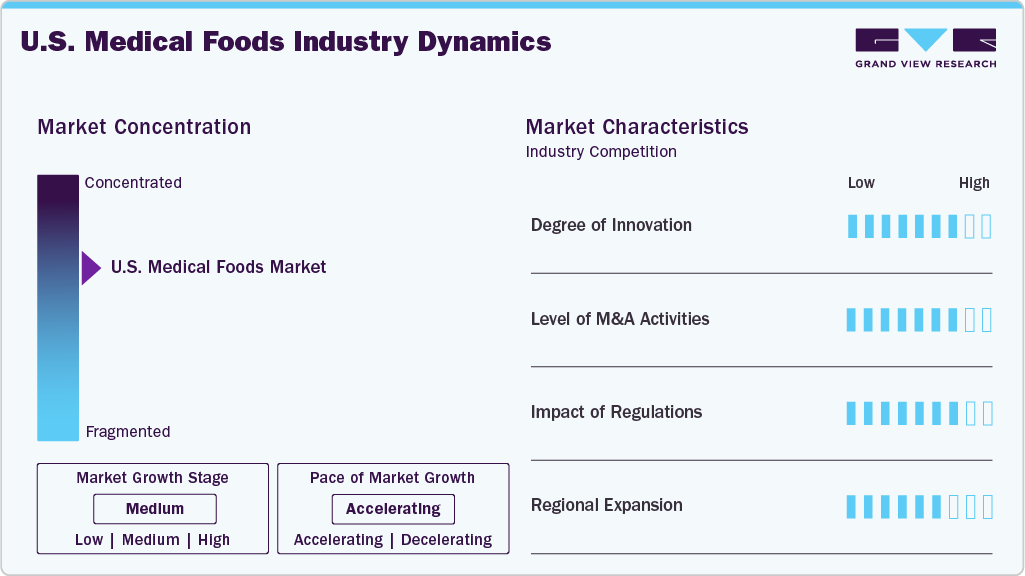
The level of M&A activity in the U.S. medical foods industry is increasing, as companies work with healthcare providers, digital health platforms, and research organizations to expand access to nutrition-based healthcare solutions and improve patient outcomes. For instance, in May 2024, Danone announced the acquisition of Functional Formularies, a U.S.-based whole-foods tube-feeding company. This development is part of Danone’s ‘Renew Danone’ strategy and is expected to strengthen its medical nutrition portfolio in the country through the expansion of its enteral tube feeding range. Functional Formularies offers organic, whole-food-based alternatives to conventional tube feeding formulas and serves institutional customers, including hospitals and long-term care facilities, as well as retail consumers through online channels across the U.S. and Canada.
The impact of regulation on the market is high, as medical foods in the U.S. must comply with specific regulatory frameworks governing foods for special medical purposes and clinical nutrition products. Regulatory authorities such as the U.S. Food and Drug Administration define medical foods under the Orphan Drug Act and set requirements for labeling, manufacturing practices, and intended use under medical supervision. While medical foods are not subject to premarket approval, such as pharmaceuticals, compliance with regulatory standards influences product formulation, marketing claims, and market entry timelines, significantly driving the growth.
The U.S. medical foods industry is experiencing steady expansion, driven by the rising prevalence of chronic diseases and the growing demand for disease-specific nutritional management. Companies are strengthening their presence across the country by expanding distribution networks, particularly through hospitals, long-term care facilities, and home healthcare channels. In addition, Manufacturers are leveraging e-commerce and healthcare partnerships to improve access, supporting wider adoption of medical foods across patient populations.
Route of Administration Insights
The oral segment accounted for the largest revenue share of the U.S. medical foods market in 2025. Nutrient and vitamin administration via the oral method is widely practiced across healthcare institutions in the U.S., with products available in various forms such as liquids, powders, puddings, pills, and pre-thickened formulations. Most patients requiring Oral Nutritional Supplements (ONS) are treated using standard products with a calorific value of 1.5 to 2.4 Kcal/mL, offered in multiple flavors to improve patient compliance.
This mode of administration is convenient, cost-effective, and highly preferred, which has driven its widespread adoption. In addition, the rising prevalence of chronic diseases and increasing demand for patient-friendly, easy-to-consume nutritional solutions, particularly among pediatric and geriatric populations. For instance, in March 2026, Nestlé introduced Compleat Paediatric Oral Blends, an oral nutritional supplement in an on-the-go pouch designed for children with specialized medical nutrition needs, highlighting the increasing focus on convenient and specialized oral formulations.
The enteral segment is expected to grow at the fastest CAGR during the forecast period. Enteral formulas are designed for diverse patient groups, including geriatric and pediatric populations, and are regulated by the U.S. Food and Drug Administration to ensure quality and safety. Growth is driven by the rising prevalence of chronic diseases such as cancer and cardiovascular disorders, which limit oral intake. In addition, advancements in feeding devices and the introduction of easy-to-administer formulations are supporting increased adoption across healthcare settings in the U.S.
Product Insights
The powdered form of medical foods accounted for the leading revenue share of 35.4% in 2025. Medical food products are extensively available in powder form that can be easily administered via the enteral or oral route by combining them with water or milk, as per the directions of the physician. Increasing consumer preference for powdered formulas, driven by their ease of use, is encouraging manufacturers to develop new product categories. Companies such as Danone, Abbott, and Nestlé have developed a range of medical foods in powdered form that address various nutritional requirements, disorders, and deficiencies.
The liquid-formulated medical food segment is anticipated to grow at the fastest CAGR from 2026 to 2033. Liquid products are administered to patients diagnosed with dysphagia or when there are limitations to their oral physiology. Moreover, these formulations are widely adopted by the geriatric and pediatric populations when solid formulations are not possible or are limited. The rising commercial viability of liquid medical foods and their rising adoption owing to ease of administration advance segment growth
Application Insights
The cancer application segment dominated the U.S. medical foods industry, accounting for the largest share of 12.2% in 2025. This is driven by the high disease prevalence and associated risk of malnutrition. According to the American Cancer Society, approximately 2 million new cancer cases are expected in 2025. With 40-80% of cancer patients experiencing malnutrition, the demand for medical foods in oncology care is significantly increasing. In addition, the growing adoption of nutritional support as part of cancer treatment protocols and the rising focus on improving patient outcomes are further driving the demand for specialized medical foods in oncology care.
The Parkinson’s disease segment is expected to grow at the fastest CAGR over the forecast period. According to the Parkinson’s Foundation 2022, about 90,000 new cases are diagnosed annually. The increasing use of medical foods containing omega-3 fatty acids, coenzyme Q, and vitamins supports nutritional management. Dietary approaches such as ketogenic diets and probiotics are also gaining traction as complementary therapies. This further drives demand for specialized medical nutrition in the management of Parkinson’s disease.
Module Insights
The protein modules segment held the largest share of the U.S. medical foods market in 2025, driven by the rising prevalence of chronic conditions, including cancer and chronic kidney disease. Increased use in clinical settings for post-surgery recovery, critical care, and dialysis patients, along with the growing geriatric population prone to sarcopenia, is supporting segment growth.
The hypoallergenic module segment is expected to witness significant growth during the forecast period. The segment growth is driven by the increasing prevalence of food allergies and intolerances, particularly among infants and patients with gastrointestinal disorders. Rising awareness and demand for specialized formulations, including amino acid-based and hydrolyzed proteins, are further boosting adoption.
Sales Channel Insights
The DTC sales channel held the largest market share in 2025. This growth is driven by the increasing adoption of digital health platforms and e-commerce, allowing companies to directly engage with patients and caregivers while bypassing traditional retail and institutional channels. The DTC channels enable manufacturers to provide personalized product recommendations, detailed nutritional information, and condition-specific guidance, improving patient awareness and adherence. The expansion of online pharmacies, subscription-based nutrition services, and home healthcare trends is further supporting the strong growth of this segment.
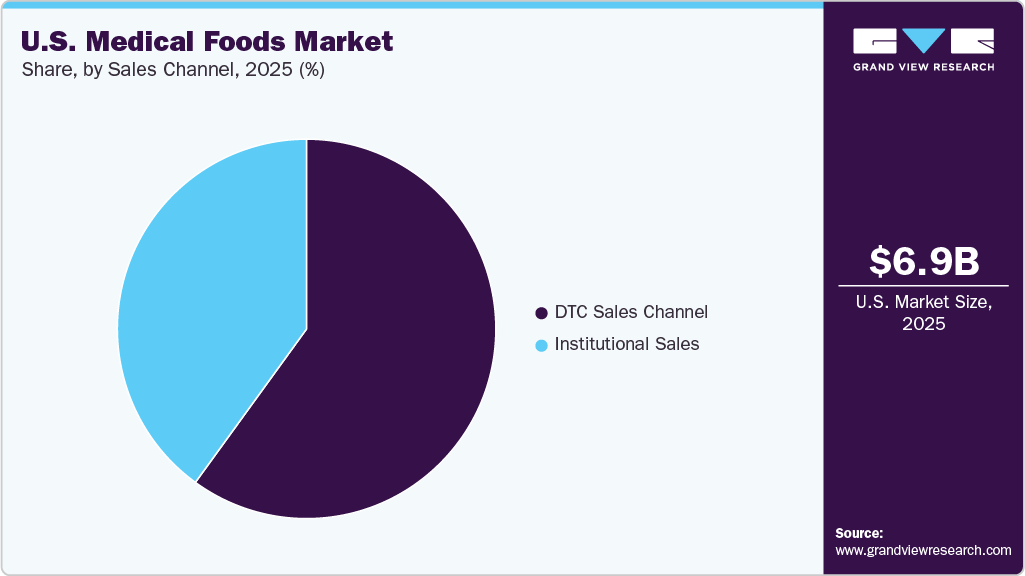
The institutional sales segment is expected to grow significantly over the forecast period. This growth is due to the hospitals, long-term care centers, clinics, and hospices account for the highest revenue share, as medical foods are prescribed under medical supervision. It is driven by the increasing number of healthcare facilities and the rising prevalence of chronic diseases. According to the data published by the American Hospital Association in 2024, 6,120 hospitals in the U.S. provided healthcare services to 33,679,935 patients. In addition, the collaboration between manufacturers and healthcare institutions in the U.S. has led to improved product visibility and accessibility within clinical settings.
Key U.S. Medical Foods Company Insights
Key players in the U.S medical foods market are adopting innovative strategies, focusing on specialized formulations for chronic diseases, plant-based and sustainable nutrition, and patient-tailored solutions to improve outcomes and adherence. Companies are investing in clinical research and digital health integration to strengthen evidence-based nutrition and enhance patient engagement. Strategic partnerships and product innovations are further supporting market expansion and competitive positioning in the U.S.
Key U.S. Medical Foods Companies:
- Danone
- Nestlé
- Abbott
- Targeted Medical Pharma, Inc.
- Fresenius Kabi AG
- Primus Pharmaceuticals, Inc.
- Mead Johnson & Company, LLC (Acquired by Reckitt Benckiser Group PLC)
- Alfasigma USA, Inc.
Recent Developments
-
In July 2025, Danone completed the acquisition of a majority stake in Kate Farms, a leading U.S. plant-based medical nutrition company, strengthening its specialized nutrition portfolio in North America. The deal enhances Danone’s ability to expand access to plant-based and medical nutrition solutions across hospitals, homecare, and retail channels in the U.S.
“Becoming part of Danone - an organization that shares our mission to improve lives through nutrition - is a really proud moment for Kate Farms and will make a positive difference to the people, families and communities we serve. Together, we are in a better position to provide high-quality nutrition to those with a wide range of health needs. With Danone’s global expertise, broad U.S. footprint and deep commitment to science-based, patient-centered care, we will reach more people, expand our impact, and continue building an organization rooted in strong values, innovation and compassion. I’m honored to step into this new role leading the combined business and excited to work alongside our world-class teams at both Danone and Kate Farms to bring our hearts and expertise to support more people on their health journey. Together, we’ll ensure our brands flourish, support the important work healthcare professionals do every day and help people thrive.”
- Brett Matthews, Danone North America Medical Nutrition Chairman and Chief Executive Officer
-
In April 2024, Brain Ritual, a Switzerland-based wellness brand, announced the launch of the medical food product MigraKet, which aims to address the root cause of migraines. The product has been developed by Elena Gross, PhD, a well-known neuroscientist and former chronic migraineur, and has been released to customers in the U.S.
-
In January 2024, Abbott announced the launch of the company’s PROTALITY brand, with the first product of this brand being a high-protein nutrition shake aimed at supporting adults who are looking to pursue weight loss while maintaining good nutrition and muscle mass. The product features a mix of slow- and fast-digesting protein that can feed the muscles for up to 7 hours. Other notable nutrients include eight essential B vitamins, 4 grams of comfort fiber, 150 calories, 1 gram of sugar, and 25 minerals and vitamins.
U.S. Medical Foods Market Report Scope
Report Attribute
Details
Market size value in 2026
USD 7.3 billion
Revenue forecast in 2033
USD 10.7 billion
Growth rate
CAGR of 5.5% from 2026 to 2033
Actual data
2021 - 2025
Forecast period
2026 - 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2026 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Route of administration, product, application, module, sales channel, region
Key companies profiled
Danone; Nestlé; Abbott; Fresenius Kabi AG; Targeted Medical Pharma, Inc.; Fresenius Kabi AG; Primus Pharmaceuticals, Inc.; Mead Johnson & Company, LLC (Acquired by Reckitt Benckiser Group PLC); Alfasigma USA, Inc.
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
U.S. Medical Foods Market Report Segmentation
This report forecasts revenue growth at the country level and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2033. For this study, Grand View Research has segmented the U.S. medical foods market report based on route of administration, product, application, module, sales channel, and region:
-
Route of Administration Outlook (Revenue, USD Million, 2021 - 2033)
-
Oral
-
Enteral
-
-
Product Outlook (Revenue, USD Million, 2021 - 2033)
-
Powder
-
Pills
-
Liquid
-
Other
-
-
Application Outlook (Revenue, USD Million, 2021 - 2033)
-
Chronic Kidney Disease
-
Minimal Hepatic Encephalopathy
-
Chemotherapy-Induced Diarrhea
-
Pathogen-Related Infections
-
Diabetic Neuropathy
-
ADHD
-
Depression
-
Alzheimer's Disease
-
Nutritional Deficiency
-
Orphan Diseases
-
Tyrosinemia
-
Eosinophilic Esophagitis
-
FPIES
-
Phenylketonuria
-
MSUD
-
Homocystinuria
-
Others
-
-
Wound Healing
-
Chronic Diarrhea
-
Constipation Relief
-
Protein Booster
-
Dysphagia
-
Pain Management
-
Parkinson's Disease
-
Epilepsy
-
Other cancer-related treatments
-
Severe Protein Allergy
-
Cancer
-
Cachexia
-
Other
-
-
Module Outlook (Revenue, USD Million, 2021 - 2033)
-
Amino Acid Module
-
Protein Module
-
Vitamin & Mineral Modules
-
Fatty Acid-based Modules
-
Carbohydrate Modules
-
Fiber Modules
-
Ketogenic Modules
-
Peptide-based Modules
-
Hypoallergenic Modules
-
Others (Electrolyte Module, etc.)
-
-
Sales Channel Outlook (Revenue, USD Million, 2021 - 2033)
-
DTC Sales Channel
-
Online Sales
-
Retail Sales
-
-
Institutional Sales
-
Hospitals
-
Others (Long-term Care Facilities, etc.)
-
-
Frequently Asked Questions About This Report
b. The U.S. medical foods market size was estimated at USD 6.9 billion in 2025 and is expected to reach USD 7.3 billion in 2026.
b. The U.S. medical foods market is expected to grow at a compound annual growth rate of 5.5% from 2026 to 2033 to reach USD 10.7 billion by 2033.
b. Based on product the powdered form of medical foods accounted for the leading revenue share of 35.4% in 2025. Increasing consumer preference for powdered formulas on account of their ease of consumption is encouraging manufacturers to focus on developing different product categories.
b. Some key players operating in the U.S. medical foods market include Danone, Nestlé, Abbott, Fresenius Kabi AG, Targeted Medical Pharma, Inc., Fresenius Kabi AG, Primus Pharmaceuticals, Inc., Mead Johnson & Company, LLC (Acquired by Reckitt Benckiser Group PLC), Alfasigma USA, Inc.
b. Key factors that are driving the market growth include by the rising geriatric population, growing prevalence of chronic diseases, and increasing adoption of disease-specific nutritional formulas.
Share this report with your colleague or friend.
Need a Tailored Report?
Customize this report to your needs — add regions, segments, or data points, with 20% free customization.

ISO 9001:2015 & 27001:2022 Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
Trusted market insights - try a free sample
See how our reports are structured and why industry leaders rely on Grand View Research. Get a free sample or ask us to tailor this report to your needs.










