- Home
- »
- Market Trend Reports
- »
-
Aficamten Market Analysis: Clinical Development, Regulatory Status
Report Overview
Aficamten, developed by Cytokinetics, Inc., is an novel oral cardiac myosin inhibitor targeting hypertrophic cardiomyopathy (HCM), a genetic heart condition affecting roughly 1 in 500 individuals globally. By selectively modulating cardiac myosin activity, Aficamten reduces excessive contractility, alleviates left ventricular outflow tract obstruction, and improves diastolic function, offering a precise, disease-targeted therapy. Currently in Phase III clinical trials, it has demonstrated strong efficacy and safety profiles, positioning it as a potential first-line option for both obstructive and non-obstructive HCM. With FDA approval anticipated in 2026, Aficamten is expected to provide a safer and more titratable alternative to existing therapies such as Camzyos (mavacamten). The drug’s market growth is driven by increasing disease awareness, rising genetic screening, and the unmet need for novel treatment options. Analysts project the global Aficamten market could reach multi-billion-dollar levels by 2035, redefining precision cardiology and improving patient outcomes worldwide.
Key Report Deliverables
-
Analyze the Aficamten market landscape, detailing its current status, ongoing clinical development, anticipated market entry, and key trends shaping the hypertrophic cardiomyopathy (HCM) treatment segment.
-
Evaluate the competitive environment, identifying key players such as Cytokinetics, Bristol Myers Squibb (Camzyos), and emerging pipeline candidates, along with strategic partnerships and market positioning.
-
Forecast market growth, projecting adoption trends, revenue potential, and expansion opportunities across obstructive and non-obstructive HCM markets, while assessing potential risks to growth.
-
Identify regulatory and market barriers, providing insights into approval timelines, pricing, reimbursement challenges, and safety monitoring requirements that could impact market expansion.
-
Concurrent Competitive Landscape, examining both direct and indirect competitors, differentiating therapeutic profiles, and analyzing strategic moves shaping the evolving HCM treatment market.
Transforming The Future Of Hypertrophic Cardiomyopathy Treatment
Aficamten, developed by Cytokinetics, Inc., is emerging as a breakthrough therapy for the management of hypertrophic cardiomyopathy (HCM), a genetic cardiac disorder characterized by abnormal thickening of the heart muscle. HCM affects approximately 1 in 500 individuals globally and can lead to symptoms such as shortness of breath, chest pain, and arrhythmias, significantly impacting quality of life. Traditional treatment options, including beta-blockers, calcium channel blockers, and surgical interventions, often provide symptomatic relief but fail to address the underlying molecular mechanisms of the disease. Aficamten, as a selective cardiac myosin inhibitor, offers a novel therapeutic approach by directly modulating cardiac contractility, addressing the root cause of HCM rather than merely alleviating symptoms.
Aficamten’s mechanism of action focuses on selectively inhibiting cardiac myosin ATPase, thereby reducing the number of actin-myosin cross-bridges in the heart muscle. This targeted approach decreases excessive contractility, alleviates left ventricular outflow tract (LVOT) obstruction, and improves diastolic filling, which is critical in patients with both obstructive and non-obstructive HCM. Unlike traditional therapies, Aficamten allows for precision titration based on patient response, minimizing risks such as hypotension or systolic dysfunction. By directly addressing the molecular drivers of hypertrophic remodeling, Aficamten is poised to transform HCM management, offering improved cardiac function, better symptom control, and potentially long-term disease-modifying benefits.
Currently in Phase III clinical trials, Aficamten has demonstrated promising efficacy and safety across multiple patient populations, signaling its potential as a first-in-class therapy for HCM. The drug is expected to receive regulatory approval in the near future, opening the door to commercial adoption in major markets, including the U.S., Europe, and Asia-Pacific regions. Its development marks a significant milestone in precision cardiology, reflecting a shift toward targeted, mechanism-based therapies that go beyond symptomatic management. With increasing disease awareness, genetic screening, and unmet clinical needs, Aficamten is well-positioned to reshape the global HCM treatment landscape and improve outcomes for patients living with this complex cardiovascular condition.
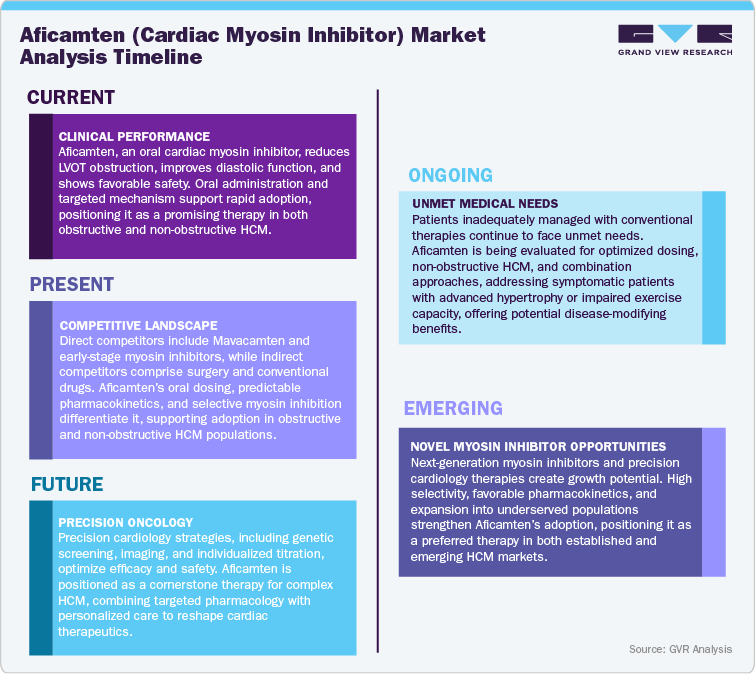
Current Market Scenarios
The global hypertrophic cardiomyopathy (HCM) therapeutics landscape is evolving rapidly, driven by increasing awareness of the disease, improvements in early diagnosis, and rising demand for innovative treatment options. Traditional therapies, such as beta-blockers, calcium channel blockers, and invasive surgical interventions, primarily manage symptoms without addressing the underlying molecular mechanisms of HCM. In this context, Aficamten, an oral cardiac myosin inhibitor developed by Cytokinetics, Inc., represents a novel therapeutic approach. By selectively modulating cardiac contractility, it targets the root cause of HCM, reducing left ventricular outflow tract obstruction and improving diastolic function. The drug’s development reflects a shift toward precision cardiology and mechanism-based treatment strategies in cardiovascular medicine.
Recent clinical trials, including MAPLE-HCM, have highlighted Aficamten’s efficacy and safety profile in patients with obstructive HCM. Results demonstrated significant improvements in exercise capacity, symptom reduction, and cardiac performance compared to standard therapies. The ability to titrate doses according to patient response enhances safety and optimizes outcomes, setting it apart from conventional therapies that often have systemic side effects. Both obstructive and non-obstructive HCM patients stand to benefit from this targeted approach, which has been recognized by cardiology experts for its potential to transform disease management. Ongoing research and regulatory progress indicate growing confidence in Aficamten’s role as a first-line therapeutic option.
Looking ahead, Aficamten is expected to reshape the global HCM market, driven by unmet clinical needs, favorable trial outcomes, and strategic collaborations for international commercialization. Partnerships and licensing agreements have expanded its reach, reinforcing its commercial potential and future adoption across major markets. Its mechanism-based approach represents a milestone in precision cardiology, promising long-term benefits in disease management and patient quality of life. As the market shifts toward targeted therapies, Aficamten is positioned to set a new standard of care, offering clinicians a more effective and safer alternative to traditional treatment strategies, and potentially redefining therapeutic expectations for hypertrophic cardiomyopathy patients worldwide.

Mechanism of Action: Targeted Myosin Inhibition for Cardiac Precision
Aficamten is a selective cardiac myosin inhibitor designed to directly modulate the molecular mechanisms driving hypertrophic cardiomyopathy (HCM). In HCM, excessive cardiac contractility caused by hyperactive myosin proteins leads to left ventricular outflow tract (LVOT) obstruction, impaired relaxation, and increased cardiac workload. Aficamten binds specifically to cardiac myosin ATPase, reducing the formation of actin-myosin cross-bridges and lowering contractile force. By targeting this fundamental pathological process, the drug addresses the root cause of the disease rather than merely mitigating symptoms. This mechanism allows Aficamten to provide precise control over cardiac contractility, reducing obstruction while maintaining overall cardiac function, which differentiates it from conventional therapies like beta-blockers or calcium channel blockers.
The enhancement of diastolic function is a key benefit of Aficamten. By reducing hypercontractility, the left ventricle can relax more effectively during diastole, allowing improved filling and better oxygen delivery to tissues. This results in improved exercise tolerance, reduced symptoms such as shortness of breath, and lower risk of adverse cardiovascular events. Unlike traditional therapies, which often have systemic effects or rely on non-specific modulation of heart rate and contractility, Aficamten’s targeted approach ensures a tunable, patient-specific therapeutic effect, optimizing both safety and efficacy. Its ability to precisely control cardiac output while alleviating obstruction highlights its potential as a disease-modifying therapy rather than solely a symptomatic treatment.
Aficamten’s mechanism offers advantages over conventional treatment strategies by combining efficacy with precision. Traditional medications, while useful in symptom management, do not modify the underlying myocardial hypercontractility or prevent disease progression. In contrast, Aficamten directly intervenes at the molecular level, potentially reducing long-term remodeling of the heart muscle. Its selective action minimizes adverse effects such as hypotension or bradycardia often seen with standard therapies. Furthermore, the drug’s titratable dosing regimen allows clinicians to adjust therapy based on individual patient response, providing a personalized treatment approach. By addressing both obstructive and non-obstructive HCM, Aficamten represents a new era in precision cardiology, offering improved outcomes and enhanced quality of life for patients.
Clinical Development and Trial Insights
Aficamten’s clinical development has been highlighted by robust studies, including the Phase II REDWOOD-HCM and Phase III SEQUOIA-HCM trials, providing critical insights into its safety, efficacy, and therapeutic potential. The Phase II REDWOOD-HCM trial focused on patients with obstructive HCM, evaluating the drug’s ability to reduce left ventricular outflow tract (LVOT) gradients and improve exercise capacity. Results showed consistent reduction in obstruction, improved functional outcomes, and relief from symptoms such as shortness of breath and fatigue. Adverse events were mostly mild to moderate, reflecting a favorable safety profile. These findings emphasize Aficamten’s potential as an effective alternative to traditional therapies, which generally offer only symptomatic relief without addressing the underlying molecular drivers of hypertrophic cardiomyopathy.
The Phase III SEQUOIA-HCM trial further assessed Aficamten in a broader patient population, including non-obstructive HCM. The study confirmed improvements in diastolic function, cardiac filling, and overall cardiac performance, demonstrating the drug’s mechanism-based effect. Dose optimization was a central aspect, showing that individualized titration maximizes benefit while minimizing risks such as hypotension or excessive reduction in cardiac output. This precise, patient-specific dosing reinforces Aficamten’s role as a personalized therapy, allowing clinicians to tailor treatment based on patient response, which is crucial for managing a heterogeneous condition like hypertrophic cardiomyopathy.
Data from the Phase II and Phase III trials strongly support Aficamten as a potential first-line therapy for HCM. Evidence demonstrates improvements in exercise tolerance, symptom management, and quality of life, addressing the limitations of existing treatments. By directly targeting hypercontractility, Aficamten also shows promise in modifying disease progression, a benefit not provided by conventional therapies. The consistent safety profile, flexible dosing options, and efficacy across obstructive and non-obstructive populations establish a strong foundation for regulatory approvals and commercial adoption, positioning Aficamten as a transformative agent in the emerging field of precision cardiology.
Regulatory Milestones and Commercial Readiness
Aficamten has made significant progress in its regulatory journey, reflecting its potential as a first-in-class therapy for hypertrophic cardiomyopathy (HCM). The Phase III SEQUOIA-HCM trial data supported submission of the New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA). The FDA has set a target action date for review, and approval is anticipated in the near future, positioning Aficamten for commercial launch in the United States shortly thereafter. Regulatory filings in Europe and Asia-Pacific regions are also underway, following positive trial outcomes and established safety and efficacy profiles. The structured regulatory strategy underscores Cytokinetics’ commitment to achieving timely global access for patients with both obstructive and non-obstructive HCM.
Commercial readiness for Aficamten is supported by robust strategic planning and market preparation. Cytokinetics has entered partnerships to enhance regional distribution and maximize reach, including collaborations for Japan and select Asia-Pacific markets. These agreements facilitate regulatory submissions, local manufacturing, and market access strategies, ensuring seamless entry upon approval. Marketing and education initiatives are being designed to train cardiologists and healthcare providers on patient selection, dose titration, and monitoring. By aligning regulatory approvals with commercial strategies, Cytokinetics aims to rapidly position Aficamten as a leading therapy in the HCM treatment landscape, emphasizing precision cardiology, personalized dosing, and patient-centered outcomes.
Global commercialization of Aficamten is expected to benefit from growing awareness of HCM and the unmet need for effective, mechanism-based therapies. Launch strategies are being tailored to each region, considering local healthcare infrastructure, reimbursement policies, and market dynamics. Europe and Asia-Pacific markets are prioritized for early adoption, leveraging strategic partnerships to ensure regulatory compliance and smooth product rollout. Commercial readiness includes pre-launch medical education programs, patient support initiatives, and distribution logistics. By integrating regulatory milestones with global market strategies, Cytokinetics aims to optimize adoption, achieve strong market penetration, and establish Aficamten as a standard-of-care therapy. This coordinated approach reflects a comprehensive plan for long-term success in precision cardiology.

Market Dynamics and Growth Drivers
The hypertrophic cardiomyopathy (HCM) market is expanding due to the increasing prevalence of the disease and rising global awareness. HCM is a genetic cardiac disorder affecting a significant number of individuals, many of whom remain undiagnosed or misdiagnosed due to subtle or variable symptoms. Advances in diagnostic techniques, including echocardiography, cardiac MRI, and genetic testing, are improving early detection and enabling timely intervention. Early diagnosis allows patients to benefit from novel therapies like Aficamten, which directly target the underlying molecular mechanisms. The growing population of diagnosed patients, combined with improved access to healthcare, is creating a strong demand for innovative treatments that go beyond symptomatic management.
Unmet clinical needs are a major driver of market growth, as traditional HCM therapies primarily address symptoms without modifying disease progression. Beta-blockers, calcium channel blockers, and surgical interventions provide limited relief and often carry risks of adverse effects. Aficamten, as a precision-targeted cardiac myosin inhibitor, addresses hypercontractility, improves diastolic function, and reduces left ventricular obstruction, offering a mechanism-based approach. Clinicians are increasingly seeking therapies that can improve patient outcomes, reduce long-term complications, and enhance quality of life. The recognition of these unmet needs has accelerated research and development efforts, positioning Aficamten to fill a critical gap in HCM treatment.
The growing focus on precision cardiology further supports market expansion, as healthcare systems emphasize personalized and targeted treatments. Aficamten’s ability to be titrated based on individual patient response exemplifies this trend, allowing for optimized therapy with minimal risks. In addition, patient education, genetic screening programs, and guideline updates are facilitating wider adoption of advanced therapies. Strategic collaborations and partnerships in key regions are also enhancing market access, ensuring the drug reaches patients efficiently. Collectively, these factors-rising prevalence, unmet clinical needs, improved diagnostics, and the shift toward precision medicine are driving robust growth and shaping the future landscape of the HCM therapeutics market.
Aficamten vs. Mavacamten and Beyond
Aficamten and mavacamten are both cardiac myosin inhibitors designed for hypertrophic cardiomyopathy (HCM), but they differ in pharmacological profile and clinical outcomes. Aficamten binds to a unique allosteric site on the cardiac myosin catalytic domain, stabilizing a weak actin-binding state and reducing ATPase activity. This selective inhibition improves diastolic function and reduces left ventricular outflow tract (LVOT) obstruction in obstructive HCM patients. Mavacamten targets a different myosin site, also reducing ATPase activity, but may carry a higher risk of left ventricular ejection fraction (LVEF) decline or arrhythmias. Clinical comparisons suggest that aficamten has a more favorable safety profile, with fewer incidences of atrial fibrillation and heart failure compared to mavacamten.
Pharmacokinetically, aficamten provides distinct advantages over mavacamten. It has a shorter half-life of approximately 3-4 days, allowing more flexible dosing and easier titration. This shorter half-life also reduces the risk of prolonged drug exposure in case of adverse events. Aficamten exhibits a predictable pharmacokinetic profile, which simplifies dose adjustments and monitoring in clinical practice. In contrast, mavacamten’s longer half-life of 6-9 days may require more careful titration and extended monitoring, especially in patients at higher cardiovascular risk. These differences highlight aficamten’s potential for safer and more individualized therapy.
Looking toward the future, several other cardiac myosin inhibitors are in development, aiming to expand treatment options for HCM. These emerging agents target alternative myosin sites or utilize different mechanisms to modulate cardiac contractility. While promising, they have not yet demonstrated the same degree of clinical efficacy and safety as aficamten or mavacamten. Given its demonstrated benefits, including efficacy, safety, and pharmacokinetic flexibility, aficamten currently holds a competitive edge in the HCM therapeutics landscape. Its profile positions it as a preferred option for clinicians seeking targeted, precision-based therapy for patients with both obstructive and non-obstructive HCM.
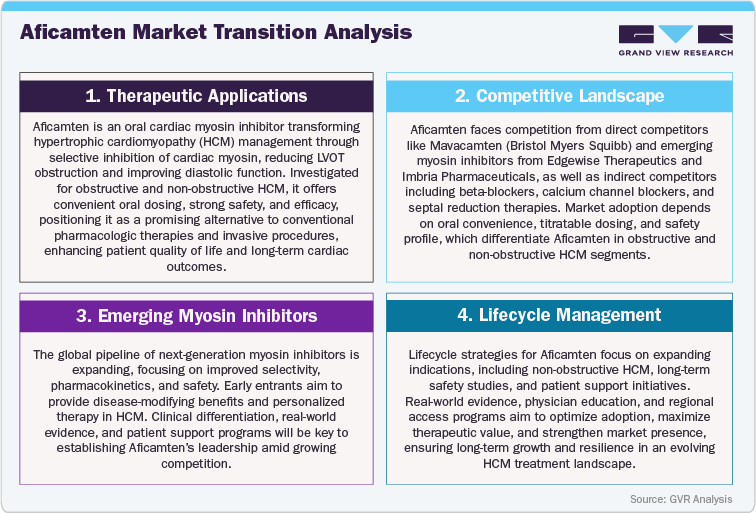
Opportunities and Strategic Challenges
The Aficamten market presents significant opportunities driven by increasing demand for precision therapies in hypertrophic cardiomyopathy (HCM). The drug’s novel mechanism allows differentiation from traditional treatments, providing both symptomatic relief and potential disease-modifying benefits. Early adoption by cardiologists is supported by robust clinical data demonstrating efficacy, a favorable safety profile, and predictable pharmacokinetics. Market expansion is also influenced by rising awareness of HCM, improved diagnostic rates, and the unmet need for targeted therapies. Cytokinetics’ strategic focus on patient education and medical outreach further strengthens its competitive position. However, careful planning is required to optimize pricing and reimbursement strategies in order to ensure both patient accessibility and commercial sustainability.
Long-term safety remains a critical consideration for Aficamten’s adoption. While clinical trials show favorable outcomes, ongoing post-marketing surveillance will be necessary to monitor potential adverse effects over extended periods. Regulatory authorities may require additional real-world evidence to support broader approval and labeling. Cytokinetics’ continued investment in R&D allows for refinement of dosing protocols, evaluation of combination therapies, and exploration of new patient populations. Strategic collaboration with healthcare providers and payers will be essential to balance affordability with commercial viability. Ensuring comprehensive long-term data and effective risk management plans will enhance physician confidence and patient adherence, driving sustainable growth for Aficamten.
Differentiation strategies will be pivotal to maintain Aficamten’s competitive edge in a landscape that includes mavacamten and emerging pipeline candidates. Cytokinetics’ focus on targeted therapy, flexible titration, and precision-based treatment aligns with evolving cardiology trends. Potential combination approaches with complementary medications could further expand therapeutic utility and market penetration. Strategic pricing, global commercialization partnerships, and efficient supply chain management are additional factors influencing adoption and profitability. By addressing clinical, commercial, and regulatory challenges, Cytokinetics aims to position Aficamten as a preferred therapy for both obstructive and non-obstructive HCM, maximizing its impact on patient outcomes and long-term market success.
Market Forecast and Future Outlook (2025-2035)
The global Aficamten market is poised for substantial growth over the next decade, driven by increasing adoption in hypertrophic cardiomyopathy (HCM) management. With anticipated approval in major markets, including the U.S., Europe, and Asia-Pacific, the drug is expected to achieve rapid penetration among both obstructive and non-obstructive HCM patients. Rising awareness, improved diagnostic capabilities, and a strong focus on precision cardiology will further accelerate uptake. Cytokinetics’ strategic partnerships and global commercialization efforts are designed to facilitate efficient distribution and access, supporting sustainable revenue expansion. Analysts view Aficamten as a transformative therapy capable of redefining treatment paradigms, positioning it as a cornerstone in the growing global HCM therapeutics landscape.
Adoption trends indicate increasing utilization in both newly diagnosed and existing HCM patients. Clinicians are expected to favor aficamten due to its efficacy, flexible dosing, and favorable safety profile. Expansion into non-obstructive HCM represents a significant market opportunity, tapping into a previously underserved patient population. Real-world evidence, ongoing post-marketing studies, and educational initiatives will reinforce confidence in treatment outcomes. The combination of targeted therapy, personalized titration, and patient support programs will drive broader acceptance and long-term adherence. These factors collectively strengthen Aficamten’s position as a preferred therapy, supporting both clinical adoption and commercial sustainability in the evolving HCM treatment landscape.
Looking ahead, Aficamten is projected to become a multi-billion-dollar global asset, reflecting its potential to capture significant market share. Market expansion will be supported by continued investment in R&D, potential combination therapies, and global regulatory approvals. Pricing strategies, reimbursement alignment, and strategic partnerships will be critical to maximizing penetration and accessibility. By addressing unmet clinical needs and offering a precision-based solution, Aficamten is positioned to establish a leadership role in the HCM market. The drug’s long-term impact will be measured by improved patient outcomes, enhanced quality of life, and its contribution to shaping the future of cardiology therapeutics worldwide.

Analyst Perspective
Industry analysts view Aficamten as a game-changing therapy in hypertrophic cardiomyopathy (HCM) treatment due to its targeted mechanism, safety profile, and potential to address unmet clinical needs. Experts highlight that the drug’s ability to modulate cardiac myosin provides a distinct advantage over traditional therapies, positioning it as a first-line option in both obstructive and non-obstructive HCM. Analysts also emphasize the importance of precision dosing and patient-specific titration, which enhances clinical adoption and improves outcomes. The growing emphasis on early diagnosis, improved awareness, and patient-centered care is expected to further accelerate market uptake. Overall, Aficamten is considered a transformative asset likely to reshape the competitive landscape in HCM therapeutics.
From a commercial perspective, analysts note that Aficamten’s market potential is substantial due to rising prevalence, increasing diagnosis rates, and the limited availability of disease-modifying therapies. Strategic partnerships and global commercialization efforts by Cytokinetics are expected to support market penetration across key regions. Analysts also highlight the potential for combination approaches and expanded indications, which could further enhance revenue streams. Pricing and reimbursement strategies will be critical to sustaining adoption while maintaining accessibility. The predictable pharmacokinetics and favorable safety profile make it easier for physicians to adopt the therapy confidently, which is expected to drive long-term growth and strengthen its competitive positioning.
Looking ahead, industry experts anticipate that Aficamten will play a leadership role in shaping the future of precision cardiology. Analysts underscore the importance of continued R&D investments, post-marketing studies, and real-world evidence to sustain physician confidence and optimize patient outcomes. Market growth is expected to be supported by strategic regulatory approvals, expanding awareness, and evolving clinical guidelines emphasizing targeted therapy. Analysts believe that Aficamten’s combination of efficacy, safety, and global commercialization readiness positions it as a multi-billion-dollar asset with strong long-term prospects. Its impact is projected to extend beyond symptomatic relief, offering potential disease-modifying benefits and improved quality of life for HCM patients.
Case Study (Recent Engagement): GLP-1 Receptor Agonist Market Opportunity Assessment
Project Objective
A leading global life sciences client approached us to assess the market potential and commercialization strategy for GLP-1 receptor agonist therapies across type 2 diabetes and obesity indications. The project aimed to support strategic planning for a novel, oral GLP-1 pipeline candidate, with a focus on launch timing, competitive positioning, and regional expansion.
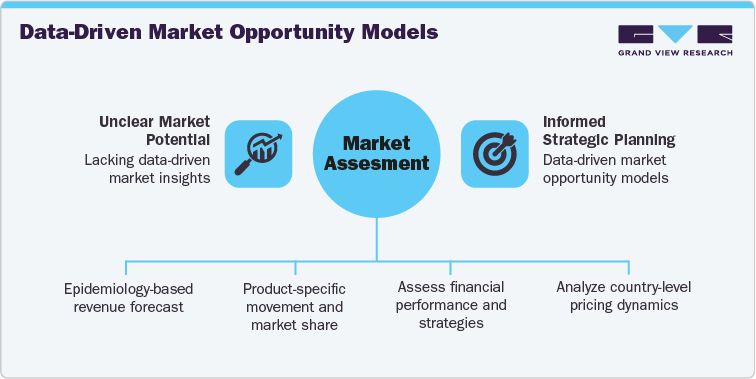
GVR Solution
-
Conducted an epidemiology-based revenue forecast (2021-2036) using patient flow and analogue modeling approaches across North America, Europe, Asia Pacific, and the Middle East.
-
Delivered product-specific movement and market share analysis for:
-
Aficamten - used as a reference analogue for uptake modeling
-
Orforglipron (pipeline)- projected using analogue-based scenarios from comparable oral GLP-1 launches
-
-
Benchmarked key players such as Eli Lilly and Novo Nordisk across financial performance, product pipeline, and global rollout strategies.
-
Assessed country-level pricing, regulatory, and reimbursement dynamics, supported by a custom launch timeline and uptake forecast for Orforglipron, modeled analogously to prior GLP-1 innovations.
-
Provided outputs (Excel, PPT, dashboard) and ongoing strategic support tailored to the client’s internal planning and commercialization team needs.
Impact for Client
-
Created market models for launch planning and portfolio prioritization.
-
Guided product strategy with pricing, uptake, and competitor insights.
-
Identified growth markets and shaped regulatory and launch plans.
Why this Matters
-
Build analogue-based forecasts for emerging therapies
-
Provide product insights for pipeline drugs with no historical sales
-
Offer strategic guidance on market entry, launch, and clinical-commercial integration
We bring the same level of analytical rigor, therapeutic market expertise, and consultative flexibility to your assessment of the pancreatic cancer microbubble-based therapy market.
Share this report with your colleague or friend.
Pricing & Purchase Options
Service Guarantee
-
Insured Buying
This report has a service guarantee. We stand by our report quality.
-
Confidentiality
We are in compliance with GDPR & CCPA norms. All interactions are confidential.
-
Custom research service
Design an exclusive study to serve your research needs.
-
24/5 Research support
Get your queries resolved from an industry expert.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


