- Home
- »
- Market Trend Reports
- »
-
Cell Culture Industry Outlook: Competitive Market Shares, Innovation Trends, And Growth Opportunities
Report Summary
The cell culture industry is undergoing a competitive reshaping driven by the advance of cell and gene therapies, deeper adoption of chemically defined specialty formulations, and increasing emphasis on regulatory-grade traceability. While traditional cell lines and classical media remain foundational for upstream bioprocessing, these segments have stabilized competitively and now differentiate primarily on supply chain reliability, batch consistency, and cGMP alignment.
The most active competitive battlegrounds lie in stem cells & blood-derived cells and specialty media products, where suppliers are using Custom formulation co-development with therapy developers, closed, automated workflow packaging, transition support from research to clinical manufacturing.
In gene therapy and cell therapy manufacturing applications, competitive concentration is noticeably higher. Players with integrated offerings across media + cell lines + viral vector reagents + bioreactor & consumables ecosystems have gained share through:
-
End-to-end workflow standardization
-
Direct biopharma partnerships
-
Regionalized manufacturing footprints
-
Licensing & supply agreements with CDMOs
-
Across categories, the competitive environment is shaped by:
-
Vendor shift from catalog-based supply → strategic account-based technical support models
-
Increased private-label and OEM manufacturing relationships
-
Regional localization strategies to mitigate supply chain risk
-
Future growth opportunities lie in:
-
CGT-focused closed-system cell expansion workflows
-
iPSC and MSC scalable media platforms
-
Digital/AI-driven media optimization programs
-
Vendor–CDMO co-development partnerships
-
For instance, in April 2025, Sartorius and Mabion announced a strategic cooperation, in which Sartorius will provide lab-scale development leveraging its proprietary cell culture media plus cell line/UP process optimisation; Mabion will take on further upstream develop, downstream optimisation, analytics, scale-up & manufacturing.
-
This is a clear example of a media/bioprocess vendor (Sartorius) working together with a CDMO-type partner (Mabion) to create an integrated offering from development through scale-up/manufacturing. It shows vendors are not just selling “off-the-shelf media” but are embedding into CDMO workflows, thereby increasing their share and “stickiness” with customers.
-
Innovation Trends & Growth Opportunities
Trend
Opportunity
Impact
Shift Toward Chemically Defined, Serum-Free, and Xeno-Free Media
There is accelerated industry movement away from serum-containing and animal-derived media due to:
- Regulatory scrutiny over variability and contamination risk
- Lot-to-lot reproducibility requirements in CGT manufacturing
- Global supply chain reliability concerns
Strategic Advantage: Vendors offering chemically defined CGT-grade formulations with documented lineage and compliance traceability are gaining share in clinical and commercial manufacturing workflows.
Integration of Media with Closed & Automated Systems
Companies are bundling media + consumables + bioreactor + QC kits into turnkey workflows, particularly for:
- CAR-T and TCR-T manufacturing
- Viral vector production platforms
This reduces operator variability and facility footprint.
Growth Play: Suppliers offering closed-system compatible media and pre-validated protocol workflows create strong lock-in at the CDMO and therapy developer level.
Custom Media Co-Development for Pipeline-Specific Cell Lines
Top biopharma and CDMOs are increasingly engaging media suppliers early in drug development to:
- Optimize growth, viability, and productivity for proprietary CHO/HEK lines
- Enhance transduction efficiency in gene therapy workflows
- Improve differentiation fidelity in iPSC programs
Growth Opportunity: Suppliers with technical consultancy teams + rapid formulation prototyping capability are winning strategic multi-year supply agreements.
Competitive Landscape By Product Category
The global cell culture industry is undergoing strong expansion driven by the rapid scale-up of biologics, cell & gene therapies (CGT), and precision medicine. Advances in stem cell science, increased adoption of high-performance specialty media formulations, and improvements in upstream process intensification are reshaping both research and biomanufacturing workflows.
Traditional cell lines (CHO, HEK293, Vero, BHK-21) continue to be the foundation for monoclonal antibody and recombinant protein production, with major vendors competing on consistency, stability, and regulatory traceability. Meanwhile, classical media (DMEM, RPMI, MEM, Ham’s F-12) has matured into a price-competitive segment with differentiation largely based on cGMP quality grades and serum-free transition support.
Growth is strongest in the Stem Cells & Blood-Derived Cells and Specialty Media categories. These segments benefit from accelerating therapeutic pipelines, particularly in CAR-T, iPSC-derived therapies, regenerative medicine, and ex-vivo gene editing workflows, where requirements for chemical definition, serum-free/xeno-free formulations, and high-cell-density performance are critical.
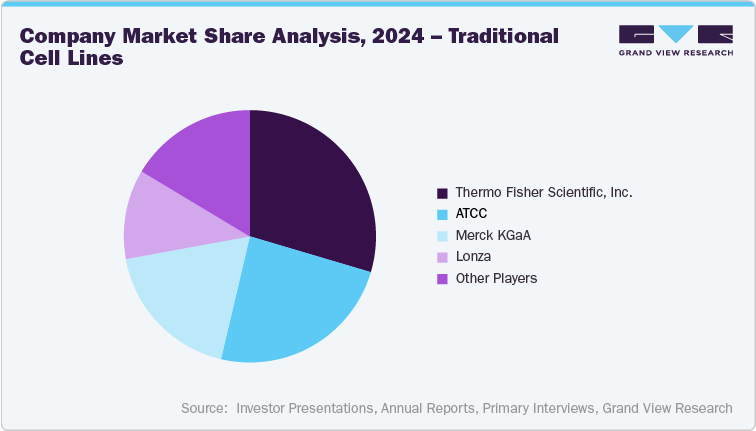
Traditional Cell Lines Competitive Landscape
Traditional cell lines such as CHO, HEK293, Vero and BHK-21 continue to form the foundational platforms for biologics, vaccine, and recombinant protein production, and represent one of the most mature and widely adopted segments in the cell culture industry. Competition in this category is shaped less by product novelty and more by cell line stability, regulatory traceability, IP/licensing structures, and technical support models that enable reliable scale-up from research to GMP manufacturing. While the overall market is relatively consolidated, differentiation persists in terms of proven clinical lineage, documentation readiness for regulatory submissions, and integration with upstream process optimization toolkits. As a result, market share leadership in this segment is typically held by suppliers that offer end-to-end workflow compatibility, global regulatory support, and validated performance data across commercial manufacturing environments.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
-
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


