- Home
- »
- Market Trend Reports
- »
-
Diabetes Clinical Trials: Emerging Trends And Pipeline Outlook
Report Overview
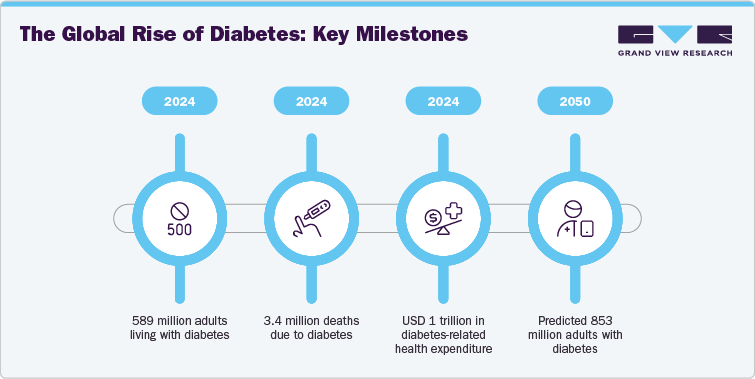
The diabetes clinical trials is experiencing robust growth driven by the increasing global burden of the disease, which is projected to affect over 853 million people by 2050. This dramatic rise is attributed to an aging global population, sedentary lifestyles, increasing rates of childhood and adult obesity, and urbanization-induced dietary shifts. As type 2 diabetes becomes more prevalent among younger populations and type 1 diabetes cases grow in many regions, pharmaceutical and biotech firms are under growing pressure to develop innovative therapeutic and management solutions. Governments and regulatory agencies are accelerating approval pathways for breakthrough therapies to combat diabetes-related morbidity, which spans cardiovascular complications, kidney disease, and limb amputations.
The current pipeline for diabetes therapeutics includes a mix of biologics, oral therapies, gene-based interventions, and digital-device combinations. A surge in interest around GLP-1 receptor agonists, SGLT2 inhibitors, and dual GIP/GLP-1 therapies is reshaping the late-stage trial landscape, with many candidates showing promise in simultaneously managing blood glucose levels and inducing weight loss. The recent expansion of indications for certain drugs to include cardiovascular protection and chronic kidney disease has made diabetes trials even more multidimensional in design.
Diabetes Clinical Trials: Emerging Trends and Pipeline Outlook Report Coverage
Market Outlook
Prevalence Trends Analysis
R&D Investment Analysis
Industry Ecosystem Analysis
Market Dynamics
Regulatory Framework
List of Top 50 Active Trials by Phase, Sponsor, and Indication
Emerging Clinical Trial Model Analysis
Global Diabetes Clinical Trials, by Phase & Study Design
Global Diabetes Clinical Trials, by key Indications, By Region
Furthermore, next-generation insulin delivery mechanisms, including automated insulin delivery systems, once-weekly injectables, and implantable pumps, are progressing through clinical phases, catering to patients seeking convenience and precision. There is also growing momentum around trials for disease-modifying therapies such as teplizumab to delay the onset of type 1 diabetes, as well as stem-cell-derived beta cell replacements that aim to restore insulin independence.
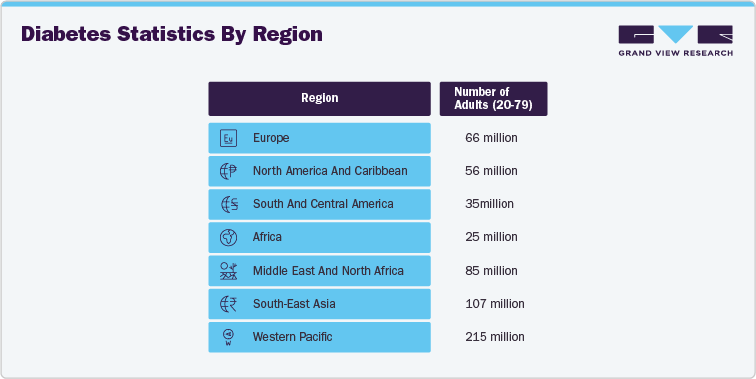
In addition, the global diabetes burden is highly uneven across regions, with developing economies accounting for the majority of diagnosed and undiagnosed cases. Rapid urbanization, shifting dietary patterns, and limited access to preventive care are fueling a silent diabetes epidemic in low- and middle-income countries. While high-income regions continue to report significant prevalence, the sharpest growth is being recorded in areas undergoing economic transitions and lifestyle westernization. This has created an urgent need for localized clinical trials that reflect regional genetic diversity, healthcare infrastructure, and comorbidity profiles. Consequently, sponsors are expanding trial footprints into Asia, Africa, and Latin America, not only to access untapped patient pools but also to ensure that new therapies are globally effective and inclusive.
R&D Investment and Funding Analysis
R&D investment in diabetes clinical trials owing to increasing cases of diabetes globally. Several pharmaceutical companies like Novo Nordisk and Eli Lilly have significantly increased their R&D spending-Novo Nordisk allocated over $5 billion in 2023, much of it toward expanding its GLP-1 and insulin innovation portfolio. Eli Lilly has similarly invested in late-stage trials of tirzepatide, a dual GIP/GLP-1 receptor agonist that has shown superior efficacy in blood sugar and weight control. On the biotech front, Vertex Pharmaceuticals partnered with CRISPR Therapeutics to advance its gene-editing based diabetes cure (VX-880) into early-phase trials, backed by strong venture and institutional funding. Meanwhile, digital therapeutics companies like Virta Health and Tandem Diabetes Care are securing capital to scale up device-enabled clinical trials targeting behavior modification and closed-loop insulin delivery, respectively.
Furthermore, public agencies are also bolstering funding; the National Institutes of Health (NIH) allocated nearly $1.2 billion to diabetes research in 2024 alone. Across Europe, IMI2 and Horizon Europe programs are co-funding multi-site trials aimed at pediatric diabetes and beta-cell preservation. These combined efforts reflect a broad, multi-stakeholder strategy to diversify therapeutic approaches and accelerate regulatory approvals through well-funded, high-quality trials.
Ecosystem Analysis of the Diabetes Clinical Trials Market
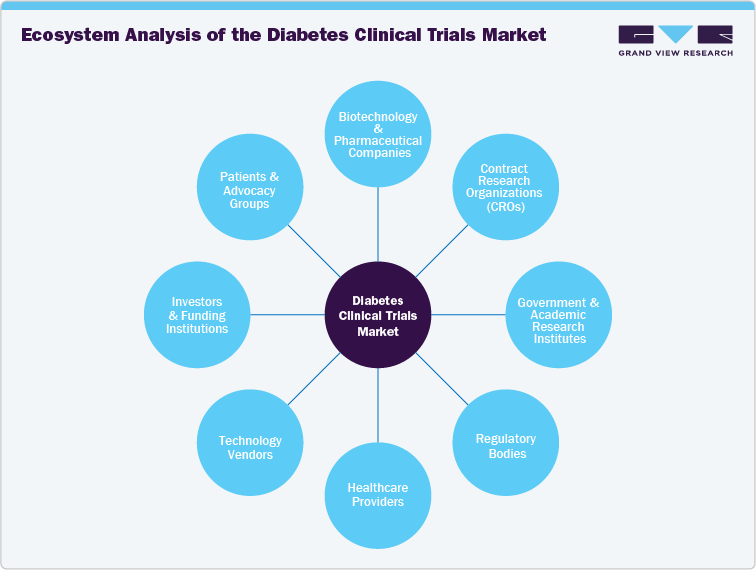
The diabetes clinical trials ecosystem comprises numerous stakeholders, including pharmaceutical and biotechnological companies, CROs, regulatory bodies, research institutions, healthcare providers, technology vendors, investors and funding institutions, and patients and advocacy groups. Pharmaceutical organizations and biotech firms are at the forefront of clinical trial sponsorship, investing heavily in research to develop innovative therapies for age-related disorders.
CROs, acting as intermediaries between sponsors and clinical trial sites, play a critical role in ensuring trial efficiency, regulatory compliance, and data management. Research institutions and universities often collaborate with industry players to advance novel treatment approaches.
Regulatory bodies like the U.S. FDA and EMA establish the framework within which clinical trials operate, influencing study design, recruitment strategies, and approval processes. The ecosystem also includes technology vendors providing clinical trial management solutions, data analytics tools, and advanced diagnostic technologies like OCT and AI-driven imaging. These entities' convergence fosters innovation, making the diabetes clinical trials market highly collaborative and competitive.
Emerging Clinical Trial Designs
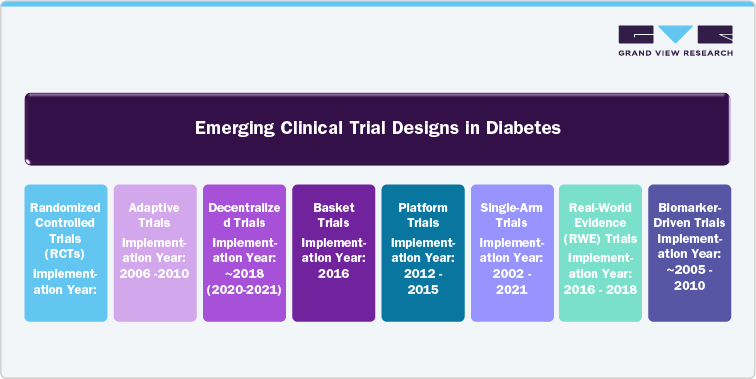
Emerging clinical trial models are transforming the way diabetes trials are designed and conducted. With the advent of decentralized trials, remote patient monitoring, and telemedicine, clinical trials are becoming more patient-centric, reducing geographic and logistical barriers. Virtual visits, wearables, and mobile health apps enable real-time data collection, making it easier for patients to participate from their homes, especially in rare or geographically dispersed conditions. Adaptive trial designs are also gaining traction, allowing modifications to the trial protocol based on interim results, thereby improving efficiency and reducing costs. Moreover, artificial intelligence (AI) and machine learning are enhancing recruitment, predictive modeling, and the identification of patient population that are most likely to benefit from specific therapies. These advancements are making clinical trials more efficient and allowing the collection of more granular data, thus providing deeper insights into treatment efficacy and safety. This report explores these emerging trial models, offering key insights into how these innovative approaches are reshaping rare disease research
U.S. Diabetes Active Clinical Trials, by Phase (2024)
Phase
Number of Trials
Key Sponsors
Top Indications
Phase I
85
The University of Texas, AstraZeneca
Type 2 Diabetes
Phase II
152
Eli Lilly and Company, Mayo Clinic
Type 1 Diabetes
Type 2 Diabetes
Phase III
69
Boehringer Ingelheim, Eli Lilly and Company
Obesity
Overweight
Type 2 Diabetes
Phase IV
61
Cellution Biologics Ohio State University
Gestational Diabetes Mellitus
Pregnancy, High Risk
Source: Clinicaltrials.gov, Grand View Research
This report’s detailed analysis of active clinical trials across phases provides stakeholders with a global view of ongoing research, helping identify trends, key players, and the most promising areas for investment. By offering insights into the number of trials, sponsors, and indications at each phase, this table serves as a vital resource for those looking to track the progress of the diabetes pipeline and make informed decisions about future investments and trial strategies.
List of Major Active Trials by Phase, Sponsor, and Indication, A Key Example
Clinical Trial Study Title
Nonhealing Diabetic Foot Ulcers Treated With Standard of Care (SOC) Alone or Standard of Care and Amnion-Intermediate-Chorion (AIC)
Study Status
Recruiting
Phase
Phase 4
Study Type
Interventional
Study Design
- Allocation: RANDOMIZED
- Intervention Model: PARALLEL
- Masking: NONE
- Primary Purpose: TREATMENT
Conditions
- Diabetic Foot Ulcer
Interventions
OTHER: Amnion-Intermediate-Chorion|PROCEDURE: Standard of Care (SOC)
Sponsor
Cellution Biologics
Number of Patients (Enrollment)
120
Start Date
22-06-2025
Primary Completion Date
30-06-2026
Completion Date
31-10-2026
Locations
004, Mission Viejo, California, 92691, United States|001, Jacksonville, Florida, 32216, United States|003, Chicago, Illinois, 60611, United States|002, Rocky Mount, North Carolina, 27804, United States
Other aspects that shall be analyzed will include the market overview, clinical trials by study design, by key indications, by region, and a list of key clinical trials, sponsors, among several other factors.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


