- Home
- »
- Reports
- »
-
Contract Research Organization Market Intelligence Report,2025
![Contract Research Organization Market Intelligence Report,2025]()
Contract Research Organization Market Procurement Intelligence, Supplier Intelligence, Supplier Ranking, Pricing & Cost Structure Intelligence, Best Practices, Engagement Model, Low & Best Cost Country, Day One Analysis Report, 2020 - 2025
- Published Date: Sep, 2021
- Base Year for Estimate: 2020
- Report ID: GVR-P-10510
- Format: Electronic (PDF)
- Historical Data: 2018-2019
- Number of Pages: 60
Category Overview
“Increasing outsourcing of clinical research by pharmaceutical companies to reduce R&D expenditure is aiding to the growth of CRO market”
The global CRO market is expected to witness sustainable growth (CAGR of 6.4%) from 2020 to 2025 due to the increasing global need for drug development and investments by pharma giants in R&D, owing to the patent expiration of blockbuster drugs.
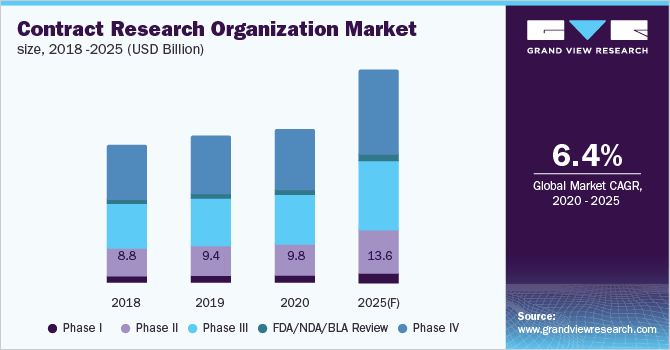
Another reason behind the increasing demand for CROs in the pharmaceutical industry is their diversified expertise as compared to pharma companies in terms of conducting clinical trials across multiple geographies and developing drugs in specific therapeutic areas. To efficiently cater to the rising demand, CROs are focusing on innovative sourcing and procurement methods for optimizing the cost of clinical trials and improving profitability.
Supplier Intelligence
“The global CRO market is moving toward consolidation, with many large corporations striking strategic partnerships with mid-sized or small-sized companies to leverage their regional clinical research capabilities.”
Mergers and acquisitions have increased over time in the CRO industry, thereby leading the market toward consolidation. Moreover, CROs are focused on combining capabilities to improve their service quality.
For instance, in January 2021, Parexel collaborated with Signify Health to provide customers with new solutions that enable access to a more diverse and traditionally underrepresented population for their clinical trials. In October 2020, Covance acquired SnapIoT to improve Covance’s mobile-connected self-service platform solutions.
Pricing and Cost Intelligence
“Though the Cost Transparency is lower, the Cost Plus Pricing Model is offered by the suppliers to maximize profits with fewer management complexities.”
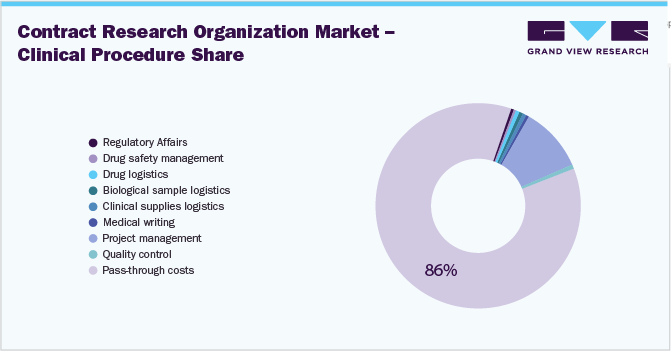
Clinical procedure is the biggest cost component (35%) of a clinical trial, followed by data management, site recruitment, and site management. The clinical procedure includes project management, regulatory affairs, drug safety management, drug logistics, biological supplies, clinical supplies logistics, medical writing, and quality control.
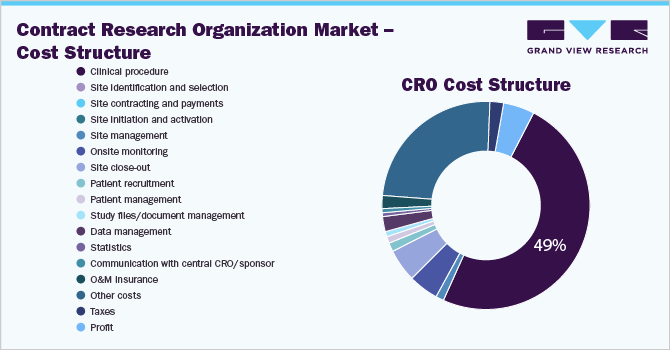
The contract research organization market procurement intelligence report provides a detailed analysis of the cost structure of the service along with the prevalent pricing models that prominent suppliers in the market opt for.
Sourcing Intelligence
“APAC is the leading sourcing destination of clinical research outsourcing services due to its dynamic population characteristics and low service cost.”
The demand for CROs is expected to increase in emerging markets such as China and India owing to the presence of huge growth opportunities coupled with reduced development time and R&D costs as well as the availability of a large, affordable, and skilled workforce. These countries offer clinical trials at significantly lower costs than the Western countries. They also have a greater population of clinically naive patients.
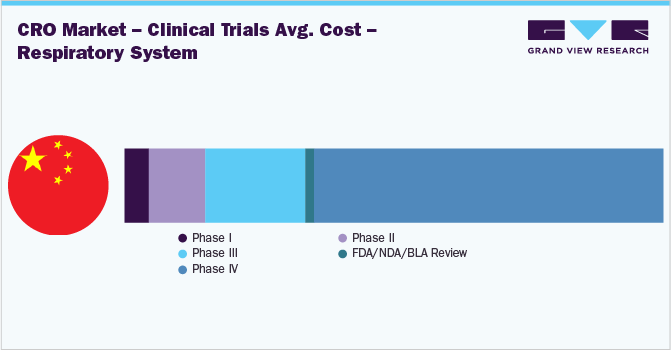
China is one of the most preferred destinations for outsourcing clinical trials, chiefly due to its large population and low patient recruitment cost. China’s position as the API manufacturing hub also favors pharmaceutical companies in outsourcing their clinical trials to China.
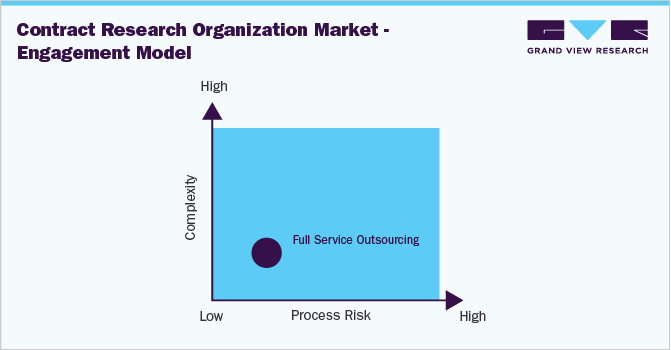
From the sourcing perspective, a majority of suppliers opt for a full services outsourcing engagement model as it reduces the complexities associated with in-house clinical trials.
Suppliers widely prefer approved provider operating models to reduce risks and improve the potential for value creation. As such, suppliers are expected to move from offering generic services to proprietary services in the future. The report also provides details regarding quick wins, portfolio analysis, key negotiation strategies of key suppliers, and low-cost/best-cost sourcing analysis.
Frequently Asked Questions About This Report
b. The global clinical trial CRO market size was valued at USD 30.3 billion in 2020 and is expected to reach USD 41.2 billion by 2025.
b. The global clinical trial CRO market is expected to record a CAGR of 6.4% from 2019 to 2025 and reach USD 41.2 billion by 2025.
b. Increasing outsourcing of clinical research by pharmaceutical companies to reduce R&D expenditure is one of the major driving factors of the clinical trial CRO market.
b. LCC/BCC sourcing analysis states China as one of the best sourcing destinations of clinical trials.
b. Some of the key CROs of clinical trials are IQVIA, Covance, PPD, and Parexel.
b. The clinical trial CRO market is majorly dominated by big CROs. However, there are many small players with niche service offerings.
b. Clinical procedures (project management, regulatory affairs, drug safety management, drug logistics, biological supplies logistics, and so on) account for 35% of the overall service cost of clinical trials.
b. Respiratory system, oncology clinical trials are the most expensive clinical trials.
b. The central nervous system, dermatology clinical trials are the least expensive clinical trials.
b. The prevalence of innovative sourcing practices, increasing spend transparency, migration to cloud-based procurement tools, and standardization of contract period are some of the best practices in the clinical trial CRO market.
Share this report with your colleague or friend.
GET A FREE SAMPLE
This FREE sample includes market data points, ranging from trend analyses to market estimates & forecasts. See for yourself...
Add-on Services
Should Cost Analysis
Component wise cost break down for better negotiation for the client, highlights the key cost drivers in the market with future price fluctuation for different materials (e.g.: steel, aluminum, etc.) used in the production process
Rate Benchmarking
Offering cost transparency for different products / services procured by the client. A typical report involves 2-3 case scenarios helping clients to select the best suited engagement with the supplier
Salary Benchmarking
Determining and forecasting salaries for specific skill set labor to make decision on outsourcing vs in-house.
Supplier Newsletter
A typical newsletter study by capturing latest information for specific suppliers related to: M&As, technological innovations, expansion, litigations, bankruptcy etc.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities.
Contact us now to get our best pricing.
![esomar icon]()
ESOMAR certified & member
![ISO]()
ISO Certified
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
Client Testimonials

"The quality of research they have done for us has been excellent..."
ISO Certified


