- Home
- »
- Medical Devices
- »
-
ADME Toxicology Testing Market Size & Share Report, 2030GVR Report cover
![ADME Toxicology Testing Market Size, Share & Trends Report]()
ADME Toxicology Testing Market Size, Share & Trends Analysis Report By Technology (Cell Culture, OMICS Tech), By Application (Neurotoxicity, Renal Toxicity), By Method (Cellular Assay, In-Silica), And Segment Forecasts, 2023 - 2030
- Report ID: GVR-2-68038-627-1
- Number of Report Pages: 120
- Format: PDF, Horizon Databook
- Historical Range: 2018 - 2021
- Forecast Period: 2023 - 2030
- Industry: Healthcare
Report Overview
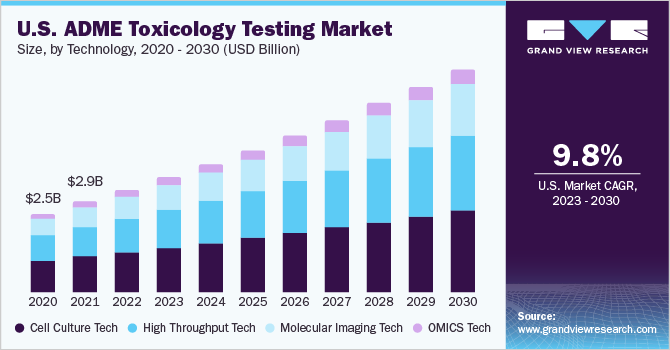
The global ADME toxicology testing market size was estimated at USD 8.7 billion in 2022 and is estimated to grow at a compound annual growth rate (CAGR) of 10.2% over the forecast period. A rising number of drug molecules have been failing in the final stages of clinical trials, resulting in the loss of valuable time and investments in drug discovery and development. This has resulted in increased investments in the discovery of newer technologies for toxicity testing, which is set to boost the market growth in the coming years. ADME toxicology testing has become an indispensable component of the drug development process as pharmaceutical companies continue to explore and create novel drugs. Regulatory bodies mandate the provision of comprehensive safety and toxicity data before approving clinical trials and subsequent commercialization of a new drug.
Furthermore, prioritizing the safety of drug candidates holds immense significance. ADME toxicology testing aids in the detection of potential toxic effects and the evaluation of the risk-benefit profile of emerging drugs. It facilitates the identification and elimination of drug candidates that could potentially induce harmful effects in humans, thereby diminishing the likelihood of drug failures or safety concerns during clinical trials. ADME toxicology testing is mainly used for conducting various genetic, chemical, and pharmacological tests that aid the drug discovery process, starting from drug design to drug trials and other drug interactions. This process involves control software, various devices to handle liquids, and other detectors that help rapidly identify active compounds, genetic interactions, and other bio molecular interactions.
However, most of these techniques need skilled expertise and software that can handle and store multiple sets of data at a rapid pace. This could limit market growth to an extent. ADME toxicology testing is now done throughout drug design and development to prevent any late-stage failure and to ensure systematic evaluation to prevent the potential loss of a molecule or investment. Most companies follow good laboratory practice (GLP) and non-GLP testing procedures to meet regulatory standards. Regulatory bodies are being pushed to establish tighter quality control standards to achieve integrity and accuracy of obtained and stored data. Automation and efficient liquid handling systems are boosting the market growth.
For instance, Tecan, a provider of laboratory instruments and solutions for clinical diagnostics, introduced an automated screening system to support ADME for the early stages of drug discovery. Regulatory changes from governing bodies, such as the U.S. FDA, are expected to impact the market growth and the direction of innovations carried out. For instance, according to the National Institutes of Health (NIH), clinical trials are being performed using biochemical or cell-based, in-vitro testing instead of animal testing. The use of artificial intelligence for drug discovery that predicts chemical toxicity, molecular design, and rational drug design is also expected to boost market growth. Moreover, it is expected that the availability of numerous innovative approaches, such as in-silica simulated models and databases for analysis of extensive chemicals to comprehend and analyze the toxicity, would contribute to the market growth.
Technology Insights
The cell culture technology segment dominated the market with the largest market share in 2022. As cell cultures provide reproducible results to consistent samples, they serve as excellent models for toxicology assays. This further enables early-stage drug toxicity testing due to which this technology is gaining popularity & extensive application. Moreover, cell culturing technology is adopted in comparison to animal testing by healthcare academic institutions, biotechnology industries, and pharmaceutical manufacturers across the globe. While evaluating the natural environment and undergoing other cell-related modifications, three-dimensional cells exhibit more lifelike characteristics that have benefited drug design and discovery. The OMICS technology segment is expected to grow at the fastest CAGR during the forecast period from 2023 to 2030. OMICS technology supports a thorough investigation of genomics, proteins (proteomics), and RNA molecules, which are then applied to the identification and development of new drugs. The single-cell OMICS technology is expected to significantly revolutionize over the coming years.
Application Insights
The systemic toxicity segment dominated the market with the largest market share of 67.4% in 2022. The demand for ADME toxicology testing services is substantial, particularly concerning the assessment of systemic toxicity owing to various factors, such as its relevance in drug development, compliance with regulatory standards, prioritization of patient safety, risk management considerations, and advancements in testing methodologies. Since the systemic circulation of the molecule is the most preferred method of drug distribution, it is also one of the major causes of toxic reactions, which may trigger multiple organs to fail.
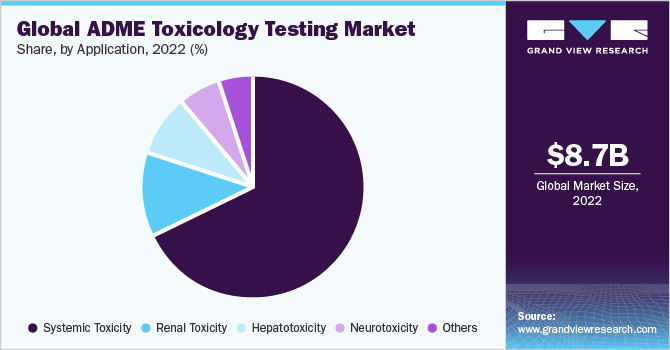
As a result, this segment has enjoyed a significant share of the market. The others segment is expected to grow at the fastest CAGR of 12.2% during the forecast period. As per the data from the National Institute of Health (NIH), acute kidney injury (AKI) accounted for 7% of hospital entries and around 30% of ICU entries in 2022. It is a progressive condition and affects 10% of the population worldwide. It is highly prevalent in older people and people with diabetes. Research initiatives are being carried out extensively that will drive market growth.
Method Insights
The cell assays segment dominated the market with the largest revenue share in 2022 owing to factors, such as highly detailed, accurate, and near-natural results of the cell assay method. Cell-based assays for physiological parameters are considered to be a bridge between the laboratory and clinic for drug candidates. These assays do not necessarily eliminate clinical trials but rather help make human experimentation less of a gamble. They also limit the number of animals required for experimentation. This is slated to be an important factor amidst the increasing sensitivity to animal rights. Simple cell-based assays have limited physiological relevance, leading to multicellular cell-based assays.
Charles River Laboratories, a provider of services and products to aid in the safe manufacturing, early-stage development, and discovery of therapeutics and new drugs, announced the development of human primary cell-based assays using cells derived from tissue, differentiated stem cells, and blood for high-content analysis. These high-throughput assays expedite the process of finding new drugs by providing the most pertinent biology. These factors will most likely fuel the growth of the ADME toxicology testing market.
Regional Insights
North America dominated the market with the largest revenue share of 46.4% in 2022. The ongoing technological advancements in the field, particularly in the U.S. drug discovery techniques using cell cultures have especially gained popularity among companies competing to reach the market with a new drug molecule. The market will be driven by collaborations and acquisitions in toxicology testing companies. In March 2023, Agilent Technologies, Inc., a provider of consumables, services, software, and instruments for laboratories, announced the acquisition of e-MSion, the developer of ExD cell technology.
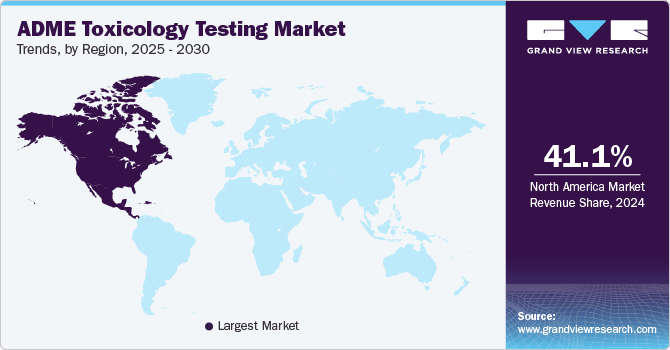
The ExD cell also known as electron capture dissociation (ECD) is a small instrument for mass spectrometers that allows biological researchers to generate biotherapeutic compounds for illness treatment more quickly. The acquisition will enable Agilent Technologies, Inc.'s LC-QTOF instrumentation to be seamlessly integrated with e-MSion's visualization tools, data analysis, and ExD technology to more thoroughly analyze speed biopharma and macromolecule treatment development and discovery. Asia Pacific is expected to grow at the fastest CAGR of 11.3% during the forecast period due to increased efforts for new pharmaceutical molecules and the adoption of newer technologies to improve the standard of the medicine being supplied.
Key Companies & Market Share Insights
The competitive landscape for the market is growing rapidly on a global scale. Companies are undertaking various business strategies, such as the introduction of newer technologies, product launches, and acquisitions, to gain higher market shares. For instance, in May 2021, CELLINK announced the acquisition of Visikol, Inc. Through this acquisition, CELLINK will leverage Visikol's capabilities to enhance various aspects of the drug discovery process. Some of the prominent players operating in the global ADME toxicology testing market include:
-
Thermo Fisher Scientific Inc.
-
Promega Corporation
-
Agilent Technologies, Inc.
-
Curia Global, Inc.
-
Dassault Systèmes
-
Beckman Coulter, Inc., (Danaher)
-
Catalent, Inc
-
Charles River Laboratories
-
Labcorp Drug Development
-
Eurofins Scientific
-
GE HealthCare
-
Miltenyi Biotec
-
IQVIA Inc.
ADME Toxicology Testing Market Report Scope
Report Attribute
Details
Market size value in 2023
USD 9.73 billion
Revenue forecast 2030
USD 19.21 billion
Growth rate
CAGR of 10.2% from 2023 to 2030
Base year for estimation
2022
Historical data
2018 - 2021
Forecast period
2023 - 2030
Report updated
September 2023
Quantitative units
Revenue in USD million and CAGR from 2023 to 2030
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Technology, application, method, region
Regional scope
North America; Europe; Asia Pacific; Latin America; MEA
Country scope
U.S.; Canada; UK; Germany; France; Italy; Spain; Denmark; Sweden; Norway; Japan; China; India; Australia; South Korea; Thailand; Brazil; Mexico; Argentina; Saudi Arabia; South Africa; UAE; Kuwait
Key companies profiled
Thermo Fisher Scientific Inc.; Promega Corp.; Agilent Technologies, Inc.; Curia Global, Inc.; Dassault Systèmes; Beckman Coulter, Inc., (Danaher); Catalent, Inc.; Charles River Laboratories; Labcorp Drug Development; Eurofins Scientific; GE HealthCare; Miltenyi Biotec; IQVIA Inc.
Customization scope
Free report customization (equivalent up to 8 analyst’s working days) with purchase. Addition or alteration to country, regional & segment scope
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Global ADME Toxicology Testing Market Report Segmentation
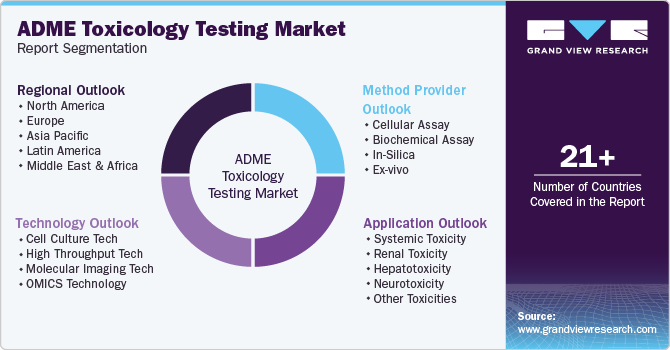
This report forecasts revenue growth at global, regional, and country levels and provides an analysis of the latest trends in each of the sub-segments from 2018 to 2030. For this study, Grand View Research has segmented the ADME toxicology testing market report on the basis of technology, application, method, and region:
-
Technology Outlook (Revenue, USD Million, 2018 - 2030)
-
Cell Culture
-
High Throughput
-
Molecular Imaging
-
OMICS Technology
-
-
Application Outlook (Revenue, USD Million, 2018 - 2030)
-
Systemic Toxicity
-
Renal Toxicity
-
Hepatotoxicity
-
Neurotoxicity
-
Other Toxicities
-
-
Method Outlook (Revenue, USD Million, 2018 - 2030)
-
Cellular Assay
-
Biochemical Assay
-
In-Silica
-
Ex-vivo
-
-
Regional Outlook (Revenue, USD Million, 2018 - 2030)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
UK
-
Germany
-
France
-
Italy
-
Spain
-
Denmark
-
Sweden
-
Norway
-
-
Asia Pacific
-
Japan
-
China
-
India
-
Australia
-
Thailand
-
South Korea
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
Kuwait
-
-
Frequently Asked Questions About This Report
b. The global ADME toxicology testing market size was estimated at USD 8.7 billion in 2022 and is expected to reach USD 9.73 billion in 2023.
b. The global ADME toxicology testing market is expected to grow at a compound annual growth rate of 10.2% from 2023 to 2030 to reach USD 19.21 billion by 2030.
b. North America dominated the ADME toxicology testing market with a share of 46.4% in 2022. This is attributable to ongoing technological advancements in the field, particularly in the U.S. Drug discovery techniques using cell cultures have especially gained popularity among companies competing to reach the market with a new drug molecule.
b. Some key players operating in the ADME toxicology testing market include Accelrys Inc.; Agilent Technologies, Inc.; Albany Molecular Research Inc.; Beckman Coulter Inc.; Catalent Inc.; Charles River Laboratories International Inc.; AbbVie; Actelion Pharmaceuticals; Amgen; Biocon; BioMarin; Boehringer Ingelheim; Bristol-Myers Squibb; Cipla; and CymaBay Therapeutics.
b. Key factors that are driving the ADME toxicology testing market growth include a high rate of late-stage drug failure, an increasing need for novel drug molecules, technological advancements.
Share this report with your colleague or friend.
![gvr icn]()
NEED A CUSTOM REPORT?
We can customize every report - free of charge - including purchasing stand-alone sections or country-level reports, as well as offer affordable discounts for start-ups & universities. Contact us now
![Certified Icon]()
We are GDPR and CCPA compliant! Your transaction & personal information is safe and secure. For more details, please read our privacy policy.
We are committed towards customer satisfaction, and quality service.
"The quality of research they have done for us has been excellent."





